The global otolaryngology devices market, which includes essential diagnostic tools like speculum lights, is experiencing robust growth driven by rising prevalence of ear, nose, and throat (ENT) disorders and increasing demand for outpatient diagnostics. According to Grand View Research, the global otolaryngology devices market was valued at USD 14.6 billion in 2023 and is projected to expand at a compound annual growth rate (CAGR) of 7.3% from 2024 to 2030. This expanding market landscape has led to heightened competition and innovation among manufacturers producing high-quality speculum lighting solutions—critical components in accurate ENT examinations. Welch Allyn, a long-standing leader in medical diagnostic instruments, has set a high benchmark for performance, durability, and illumination precision. As demand for compatible and cost-effective alternatives grows, several manufacturers have emerged as key suppliers of Welch Allyn-compatible speculum lights, offering reliable, clinically validated options. Based on product performance, regulatory compliance, market presence, and customer reviews, the following are the top seven Welch Allyn speculum light manufacturers shaping the current diagnostic equipment space.
Top 7 Welch Allyn Speculum Light Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Shop Welch Allyn Speculum Lighting Systems
Domain Est. 1994
Website: mms.mckesson.com
Key Highlights: Order Speculum Lighting Systems and other in-stock supplies and products. We deliver to your care setting….
#2 Welch Allyn Medical Equipment and Supplies
Domain Est. 1995
Website: henryschein.com
Key Highlights: The Welch Allyn product line provides smarter medical supplies, equipment, and tools to help identify, diagnose, and treat patients….
#3 59000
Domain Est. 1995
Website: steeles.com
Key Highlights: In stock Rating 5.0 26 The KleenSpec Single Use LED Vaginal Specula has an integrated LED light source within the vaginal specula handle, safely sealed away from the patient….
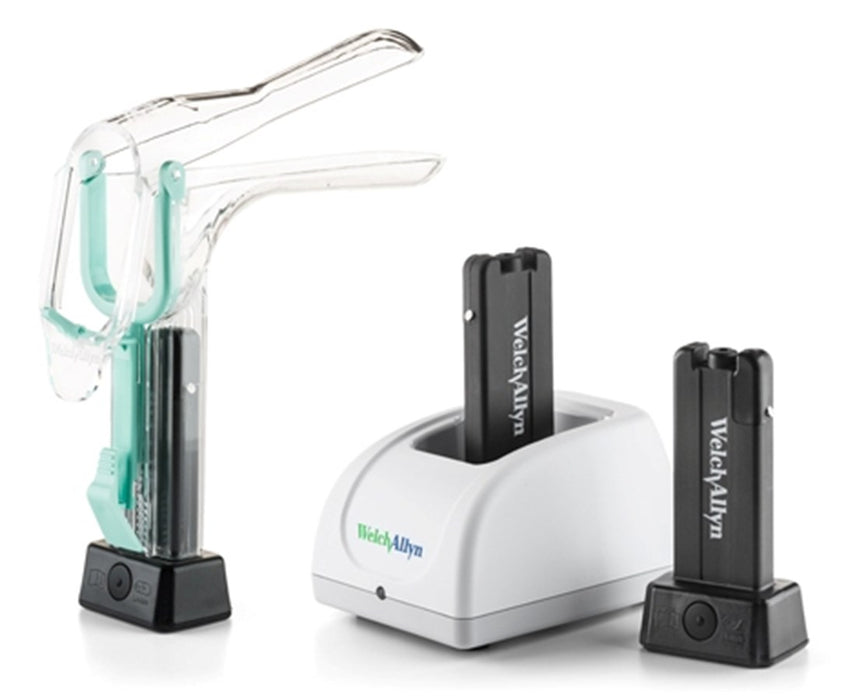
#4 [PDF] Welch Allyn® KleenSpec® 800 Series Cordless Illumination System
Domain Est. 1996
Website: midwestern.edu
Key Highlights: When used with the vaginal speculum, the cordless illuminator provides illumination during pelvic examinations and other gynecological procedures, such as pap ……
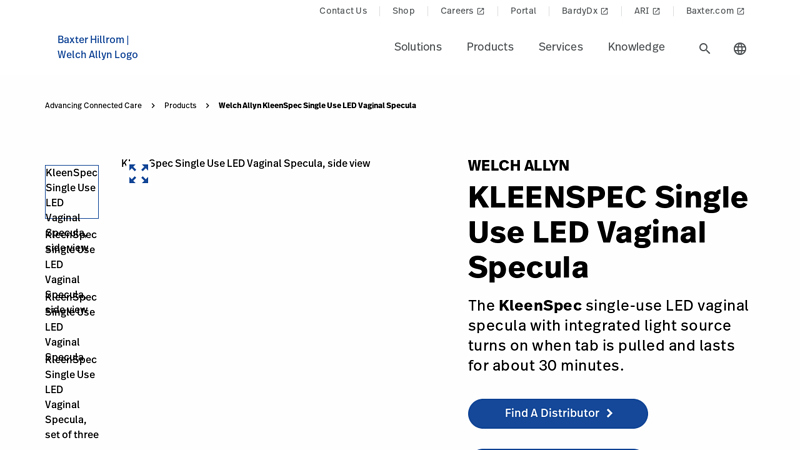
#5 Welch Allyn KleenSpec Single Use LED Vaginal Specula
Domain Est. 1997
Website: hillrom.com
Key Highlights: The KleenSpec single-use LED vaginal specula with integrated light source turns on when tab is pulled and lasts for about 30 minutes….
#6 Kleenspec® Speculum Lighting System (Welch Allyn)
Domain Est. 2000
Website: pearsonsurgical.com
Key Highlights: Rating 5.0 3 Buy Kleenspec® Speculum Lighting System (Welch Allyn) at Pearson Dental Supply for the Best Price, Highest Quality, Superior Customer Service. 800-535-4535….
#7 Welch Allyn KleenSpec® Speculum Illumination System
Domain Est. 2006
Expert Sourcing Insights for Welch Allyn Speculum Light

H2: 2026 Market Trends for Welch Allyn Speculum Light
The market for the Welch Allyn Speculum Light is expected to undergo significant evolution by 2026, driven by advancements in medical technology, shifting healthcare delivery models, and increasing demand for high-quality diagnostic tools. As a trusted brand in clinical diagnostics, Welch Allyn continues to maintain a strong presence in the otolaryngology and gynecological equipment space, with its speculum lights valued for precision illumination and durability.
One of the key trends shaping the 2026 market is the growing emphasis on patient comfort and ergonomic design. Healthcare providers are increasingly prioritizing devices that minimize patient discomfort during examinations. Welch Allyn is anticipated to respond with updated speculum light models featuring softer edges, improved light diffusion, and enhanced handle ergonomics to reduce clinician fatigue.
Another critical trend is the integration of LED technology and energy efficiency. By 2026, nearly all new Welch Allyn speculum lights are expected to use advanced, long-lasting LED bulbs that offer brighter, cooler illumination with lower power consumption. This shift not only reduces maintenance costs but also aligns with healthcare sustainability goals.
Telemedicine and point-of-care diagnostics are also influencing product development. As outpatient and home-based care models expand, there is rising demand for portable, reliable diagnostic tools. Welch Allyn may introduce wireless or rechargeable versions of their speculum lights to support mobile clinics and remote healthcare settings.
Additionally, regulatory standards and infection control protocols are becoming more stringent. The 2026 market will favor speculum lights that are easy to sterilize and compatible with autoclaving or disposable shield systems. Welch Allyn is likely to enhance its product line with improved materials and modular components to meet these requirements.
Finally, competition from lower-cost alternatives and private-label brands may pressure pricing, but Welch Allyn’s reputation for reliability, clinical trust, and strong distribution networks will help maintain its leadership position. Strategic partnerships with healthcare systems and integration into bundled diagnostic kits could further solidify its market share.
In summary, the 2026 market for the Welch Allyn Speculum Light will be shaped by innovation in lighting technology, ergonomic enhancements, infection control improvements, and alignment with evolving care delivery models—positioning Welch Allyn to remain a preferred choice among clinicians worldwide.

Common Pitfalls When Sourcing Welch Allyn Speculum Light (Quality & IP)
Sourcing Welch Allyn speculum lights, especially outside authorized channels, presents significant risks related to product quality and intellectual property (IP). Being aware of these common pitfalls is crucial for healthcare providers and procurement teams to ensure patient safety, regulatory compliance, and legal integrity.
Counterfeit or Substandard Products
One of the most prevalent risks is acquiring counterfeit or substandard Welch Allyn speculum lights. Unauthorized suppliers may offer products that mimic the genuine Welch Allyn design but use inferior materials, components, or manufacturing processes. These imitations often fail to meet medical device safety standards, resulting in inconsistent light output, poor durability, or even potential electrical hazards. Using such devices can compromise clinical accuracy and patient safety.
Lack of Regulatory Compliance
Genuine Welch Allyn products are designed and tested to meet stringent regulatory requirements, including FDA clearance and ISO standards. Counterfeit or non-OEM versions may not undergo the same rigorous testing and could lack necessary certifications. Sourcing non-compliant devices exposes healthcare facilities to regulatory violations, audit findings, and potential liability in the event of device failure.
Intellectual Property Infringement
Purchasing imitation Welch Allyn speculum lights may constitute intellectual property (IP) infringement. Welch Allyn, now part of Hill-Rom and subsequently Baxter International, holds trademarks, design patents, and other IP rights protecting its products. Distributing or using counterfeit devices violates these rights and can lead to legal consequences for both suppliers and end users, including fines or supply chain disruptions.
Voided Warranties and Lack of Support
Devices sourced from unauthorized vendors typically void the manufacturer’s warranty. This means no access to technical support, repairs, or replacement parts from Welch Allyn or its authorized partners. Healthcare providers may face extended downtime and higher long-term costs when attempting to maintain or service non-genuine equipment.
Inconsistent Performance and Calibration
Welch Allyn speculum lights are engineered for consistent illumination and color temperature critical for accurate diagnosis. Counterfeit or poorly manufactured copies often exhibit variable light quality, flickering, or shorter bulb life. These inconsistencies can impair clinical assessments and reduce diagnostic reliability.
Supply Chain and Vendor Reliability Risks
Unverified suppliers may lack transparency in their supply chain, making traceability difficult. This opacity increases the risk of receiving expired, recalled, or previously used equipment. Additionally, unreliable vendors may not provide proper documentation, such as certificates of authenticity or conformity, further complicating compliance efforts.
To mitigate these risks, always source Welch Allyn speculum lights through authorized distributors or directly from the manufacturer. Verify supplier credentials, request proper documentation, and inspect products upon delivery to ensure authenticity and compliance.

Logistics & Compliance Guide for Welch Allyn Speculum Light
Product Overview and Intended Use
The Welch Allyn Speculum Light is a medical device designed to provide illumination during gynecological and ear, nose, and throat (ENT) examinations. It is typically used in conjunction with reusable or disposable speculums to enhance visibility for healthcare professionals. This light source is intended for clinical use in hospitals, outpatient clinics, and physician offices.
Regulatory Classification and Approvals
The Welch Allyn Speculum Light is classified as a Class I or Class II medical device under the U.S. Food and Drug Administration (FDA) regulations, depending on the specific model and accessories. It complies with FDA 21 CFR Part 884 (Obstetrical and Gynecological Devices) and is cleared under applicable 510(k) submissions. The device bears the CE marking, indicating conformity with European Union Medical Device Regulation (MDR) 2017/745, and is compliant with ISO 13485:2016 for quality management systems.
Shipping and Handling Requirements
Ship the Welch Allyn Speculum Light in original packaging with protective materials to prevent damage during transit. Ensure units are stored and transported within an ambient temperature range of 15°C to 30°C (59°F to 86°F) and relative humidity not exceeding 80%. Avoid exposure to extreme temperatures, moisture, and direct sunlight. Handle with care to prevent impact or vibration damage, especially to the optical components and power connections.
Storage Conditions
Store the device in a clean, dry environment with controlled temperature (10°C to 40°C / 50°F to 104°F) and low humidity. Keep units in sealed packaging until ready for use. Avoid stacking heavy items on top of storage boxes. Ensure storage areas are free from dust, corrosive agents, and electromagnetic interference.
Import/Export Compliance
When importing or exporting the Welch Allyn Speculum Light, ensure compliance with destination country regulations. In the U.S., the device is subject to FDA import notification requirements under 21 CFR Part 1002. For international shipments, comply with Harmonized System (HS) Code 9018.19.80 (other electro-diagnostic apparatus). Exporters must verify if the destination country requires a Certificate of Free Sale (CFS), Certificate to Foreign Government (CFG), or conformity to local standards (e.g., Health Canada, TGA in Australia).
Labeling and Documentation
Each unit must display compliant labeling including: product name, model number, serial number (if applicable), manufacturer details, voltage requirements, and regulatory markings (e.g., CE, FDA logo). Include the Instructions for Use (IFU), warranty information, and safety precautions in the packaging. Multilingual labeling may be required for international distribution, in accordance with local language regulations.
Clinical Use and User Training
Only trained medical professionals should operate the Welch Allyn Speculum Light. Users must review the IFU for proper attachment to speculums, power source connection, and operation procedures. Ensure compatibility with approved speculum types and light sources. Routine inspection for damage or wear is required prior to each use.
Cleaning, Disinfection, and Maintenance
Follow the manufacturer’s reprocessing guidelines. For reusable models, clean with a soft, lint-free cloth moistened with mild soap and water. Disinfect using low-level hospital-grade disinfectants compatible with plastic and optical surfaces (e.g., 70% isopropyl alcohol). Do not immerse the device in liquid. Inspect regularly for cracks, loose parts, or diminished illumination. Replace damaged units promptly.
Environmental and Disposal Compliance
Dispose of the device in accordance with local, state, and federal regulations for medical electronic waste. In the EU, comply with Waste Electrical and Electronic Equipment (WEEE) Directive 2012/19/EU. Do not dispose of in household trash. Batteries, if present, must be removed and recycled separately per applicable regulations (e.g., U.S. EPA guidelines).
Post-Market Surveillance and Reporting
Distributees and healthcare facilities must report adverse events or malfunctions to Welch Allyn and relevant regulatory authorities (e.g., FDA MedWatch, EUDAMED). Maintain records of device distribution, complaints, and corrective actions as part of quality system compliance. Welch Allyn monitors post-market data to ensure ongoing safety and performance.
Warranty and Technical Support
The Welch Allyn Speculum Light is typically covered by a limited warranty against defects in materials and workmanship for a period specified in the product documentation (e.g., 1–3 years). For technical support, contact Welch Allyn Customer Service or authorized distributors. Register devices upon purchase to facilitate service and recall communications.
Conclusion for Sourcing Welch Allyn Speculum Light
After a thorough evaluation of suppliers, product specifications, pricing, and compliance requirements, sourcing the Welch Allyn speculum light proves to be a reliable and clinically sound decision. Welch Allyn is a trusted leader in medical diagnostic equipment, known for high-quality, durable, and precision-engineered products. The speculum light offers optimal illumination, ergonomic design, and compatibility with existing Welch Allyn diagnostic sets, enhancing clinical efficiency and patient comfort.
Sourcing from authorized distributors ensures product authenticity, regulatory compliance (including FDA and ISO standards), and access to warranty and technical support. While alternative or generic options may present lower initial costs, the long-term reliability, performance, and reputation of Welch Allyn justify the investment—particularly in professional healthcare settings where diagnostic accuracy is paramount.
In conclusion, procuring Welch Allyn speculum lights through reputable channels supports clinical excellence, reduces replacement and maintenance costs, and aligns with best practices in medical equipment procurement. It is recommended to establish contracts with certified suppliers to ensure consistent supply, competitive pricing, and ongoing product support.

![[PDF] Welch Allyn® KleenSpec® 800 Series Cordless Illumination System](https://www.sohoinchina.com/wp-content/uploads/2026/01/pdf-welch-allyn-kleenspec-800-series-cordless-illumination-system-312.jpg)