The global laminar flow hood market is experiencing steady expansion, driven by increasing demand for contamination-controlled environments across pharmaceutical, biotechnology, and healthcare sectors. According to Grand View Research, the global cleanroom technology market—of which laminar flow hoods are a critical component—was valued at USD 5.5 billion in 2023 and is projected to grow at a CAGR of 5.8% from 2024 to 2030. Vertical laminar flow hoods, in particular, are gaining traction due to their space-efficient design and effective unidirectional airflow, making them ideal for lab and manufacturing applications requiring ISO Class 5 (Class 100) cleanroom standards. With stringent regulatory requirements and increased R&D investments in life sciences, the demand for high-performance, reliable flow hoods is intensifying. As a result, several manufacturers have emerged as leaders, leveraging advanced filtration systems, energy-efficient blowers, and ergonomic designs to capture market share. Based on performance metrics, innovation, and global footprint, these top 8 vertical laminar flow hood manufacturers are shaping the future of clean airflow technology.
Top 8 Vertical Laminar Flow Hood Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Laminar Airflow Workstations
Domain Est. 1996
Website: nuaire.com
Key Highlights: AireGard® Laminar Airflow Workstations (LAFW) also known as laminar flow hoods/clean benches provide ISO Class 5 product protection for USP <797> sterile, ……
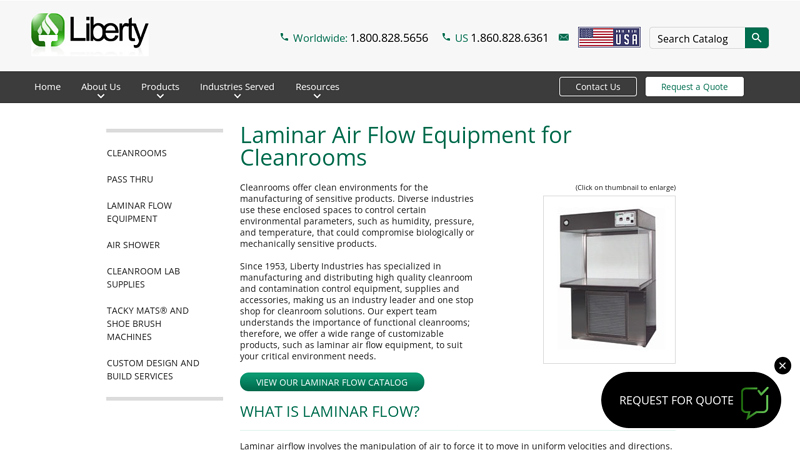
#2 Laminar Air Flow Equipment for Cleanrooms
Domain Est. 1996
Website: liberty-ind.com
Key Highlights: At Liberty Industries, we offer the following equipment to suit the needs of cleanrooms: Horizontal laminar flow hoods; Vertical laminar flow hoods; Fume hoods ……
#3 Germfree Vertical Laminar Flow Workstation Brochure
Domain Est. 1996
Website: germfree.com
Key Highlights: The BVBI series is Germfree’s advanced line of Vertical Laminar Flow Hoods. These USP <797> compliant units provide a high degree of localized control for ……
#4 Laminar Flow Hood
Domain Est. 1998
Website: airscience.com
Key Highlights: Air Science Laminar Flow Hoods are a series of high efficiency products designed to protect equipment and other contents of the work zone from particulates….
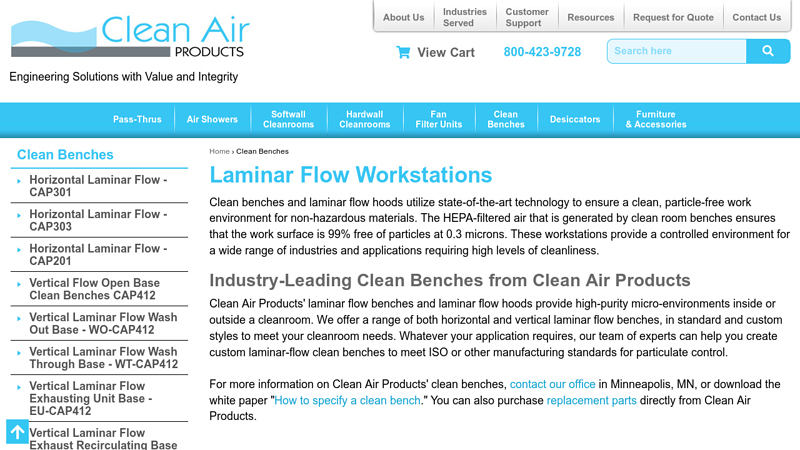
#5 Laminar Flow Clean Benches
Domain Est. 1998
Website: cleanairproducts.com
Key Highlights: We offer a range of both horizontal and vertical laminar flow benches, in standard and custom styles, to meet your cleanroom needs….
#6 Vertical Laminar Flow Cabinet
Domain Est. 2008
Website: lorderan.com
Key Highlights: Free delivery 30-day returns3 years warranty. Standard with base stand. HEPA Filter. ISO Class 5 work zone. Low noise <65dB. Stainless steel worktop. Available sizes: 4.3ft and 6ft...
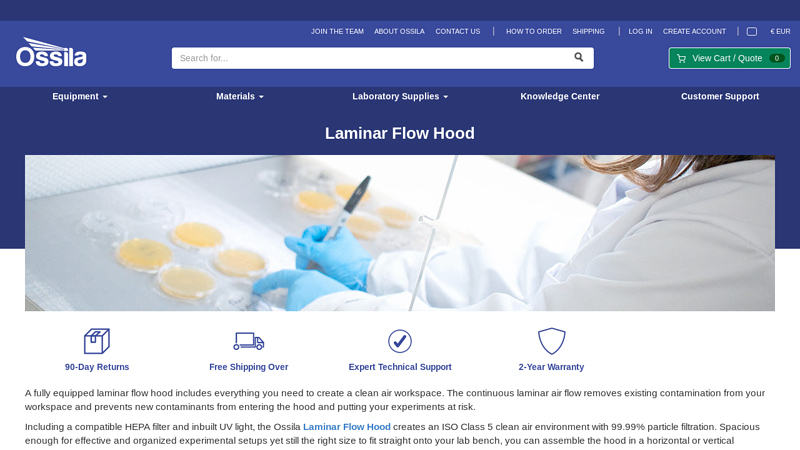
#7 Laminar Flow Hoods
Domain Est. 2009
Website: ossila.com
Key Highlights: Horizontal and Vertical Laminar Flow Hoods. Two main configurations, vertical and horizontal, are available. The difference between them is the direction of air ……
#8 Baker CleanAir Laminar Flow Cabinets & Hoods
Website: labman.be
Key Highlights: CleanAir by Baker horizontal and vertical laminar flow hoods. Laboratory hoods providing a clean work space. Official distributor Belgium….
Expert Sourcing Insights for Vertical Laminar Flow Hood
2026 Market Trends for Vertical Laminar Flow Hood
The global market for Vertical Laminar Flow Hoods (VLHs) is poised for significant evolution by 2026, driven by converging forces in healthcare, biotechnology, and advanced manufacturing. Key trends shaping the landscape include:
Accelerated Adoption in Emerging Biotech and Pharma Hubs
Emerging economies in Asia-Pacific (particularly India, South Korea, and Southeast Asia) and Latin America are witnessing rapid expansion in pharmaceutical manufacturing, biologics development, and contract research organizations (CROs). This growth is fueling substantial demand for affordable, reliable contamination control solutions like VLHs. Local manufacturers are increasingly offering cost-competitive models, making VLHs more accessible and driving market penetration beyond traditional Western markets.
Integration of Smart Technologies and IoT Connectivity
By 2026, VLHs are increasingly incorporating smart features. Integration with Internet of Things (IoT) platforms enables real-time monitoring of critical parameters such as airflow velocity, filter integrity, temperature, and humidity. Remote alerts for maintenance or deviations, coupled with data logging for compliance (e.g., GMP, ISO 14644), enhance operational efficiency and regulatory adherence. Touchscreen interfaces and connectivity to laboratory information management systems (LIMS) are becoming standard in premium models.
Heightened Focus on Energy Efficiency and Sustainability
Growing environmental regulations and corporate sustainability goals are pushing manufacturers to develop energy-efficient VLHs. Innovations include high-efficiency particulate air (HEPA) filters with lower pressure drops, variable frequency drives (VFDs) for blowers to modulate airflow based on usage, and improved cabinet designs minimizing energy consumption. Demand for units with reduced noise emissions and recyclable materials is also rising.
Demand for Enhanced Ergonomics and Flexibility
Laboratory workflows demand more adaptable equipment. The 2026 market shows a clear trend towards VLHs with adjustable-height work surfaces, improved ergonomic designs to reduce user fatigue, and modular configurations. Increased demand for portable or benchtop models suitable for smaller labs, point-of-care testing facilities, and educational institutions is evident, offering flexibility without compromising sterility.
Stringent Regulatory Scrutiny and Quality Assurance
As biopharmaceutical production (especially cell and gene therapies) becomes more prevalent, regulatory bodies enforce stricter contamination control standards. This drives demand for VLHs with robust validation support packages, superior filter performance (including ULPA options), and seamless integration into cleanroom validation protocols. Traceability, calibration services, and comprehensive documentation are becoming critical purchasing factors.
Competition from Horizontal Laminar Flow and Advanced Alternatives
While VLHs remain popular, competition is intensifying. Horizontal Laminar Flow Hoods (HLFHs) are regaining favor in certain applications due to superior ergonomics and lower risk of operator contamination of the work. Furthermore, advancements in isolator technology and restricted access barrier systems (RABS) for high-risk processes are displacing VLHs in some sterile manufacturing applications, pushing VLH manufacturers to innovate and differentiate.
Common Pitfalls When Sourcing a Vertical Laminar Flow Hood (Quality & IP)
Sourcing a Vertical Laminar Flow Hood (VLFH) requires careful attention to both quality and intellectual property (IP) considerations. Overlooking key factors can lead to performance issues, contamination risks, compliance failures, or legal complications. Below are common pitfalls to avoid:
Poor Build Quality and Material Selection
Many low-cost VLFHs use substandard materials such as non-corrosion-resistant metals or low-grade plastics that degrade under frequent disinfection. Inferior welding, poor sealing, or inadequate filtration housing can compromise airflow integrity and create contamination hotspots. Always verify construction materials (e.g., 304 or 316 stainless steel) and inspect workmanship before purchase.
Inadequate or Uncertified HEPA Filtration
A critical function of a VLFH is maintaining ISO Class 5 (Class 100) clean air via HEPA filtration. Some suppliers offer units with uncertified or counterfeit HEPA filters that do not meet H13 or H14 standards. Ensure filters are individually tested and certified with a valid test report (e.g., IEST or EN 1822 compliance) and confirm proper filter installation and sealing.
Insufficient Airflow Velocity and Uniformity
VLFHs must maintain a consistent downward airflow velocity (typically 0.3–0.5 m/s) with minimal turbulence. Poorly designed hoods exhibit uneven airflow, dead zones, or recirculation due to improper fan placement or baffle design. Demand airflow velocity maps and certifications (e.g., ISO 14644-3) to validate performance.
Lack of Regulatory Compliance Documentation
Units intended for pharmaceutical, medical, or research use must comply with regional standards (e.g., USP <797>, ISO 14644, or GMP guidelines). Some suppliers fail to provide traceable compliance documentation or valid calibration records. Always request a Declaration of Conformity and verify alignment with applicable regulations.
Counterfeit or Infringed Intellectual Property
Reputable VLFH designs often involve patented airflow systems, control interfaces, or safety features. Sourcing from unverified suppliers increases the risk of purchasing counterfeit units that infringe on IP rights. This can expose your organization to legal liability, especially in regulated industries. Procure equipment only from authorized distributors or manufacturers with verifiable IP ownership.
Incomplete or Missing Design Documentation
Proper validation and maintenance require access to technical drawings, schematics, and user manuals. Some suppliers provide incomplete documentation or omit critical details like filter part numbers or electrical specifications. Ensure full technical documentation is included and that software/firmware (if applicable) is properly licensed.
Hidden Costs from Poor Design
Units with inefficient fans or oversized enclosures lead to higher energy consumption and noise levels. Others may lack ergonomic features or easy access for maintenance, increasing long-term operational costs. Evaluate total cost of ownership, including power usage, filter replacement frequency, and service requirements.
Inadequate After-Sales Support and Service
Some suppliers, especially offshore manufacturers, offer limited technical support or spare parts availability. This can lead to extended downtime or forced replacement. Confirm warranty terms, service network accessibility, and availability of consumables before finalizing procurement.
Avoiding these pitfalls requires due diligence in supplier vetting, verification of certifications, and attention to both technical specifications and legal compliance. Engaging with reputable, established manufacturers ensures long-term reliability and regulatory safety.
Logistics & Compliance Guide for Vertical Laminar Flow Hood
Receiving and Transportation
Ensure the vertical laminar flow hood is shipped via a freight carrier experienced in handling sensitive laboratory equipment. Use packaging with sufficient cushioning and a rigid outer crate to prevent damage during transit. Upon delivery, inspect the shipment for visible damage before signing the delivery receipt. If damage is observed, document it with photos and notify the supplier and carrier immediately. Store the unit in a clean, dry, temperature-controlled environment until installation.
Installation and Site Preparation
Install the laminar flow hood on a stable, level surface away from foot traffic, doors, and ventilation sources that could disrupt airflow. Ensure adequate clearance around the unit (minimum 6 inches on all sides) for proper airflow and maintenance access. Verify that the electrical supply matches the unit’s requirements (typically 110–120V, 60 Hz, grounded outlet). Avoid daisy-chaining power strips; plug directly into a dedicated circuit if possible. Perform initial setup according to the manufacturer’s instructions, including filter installation and pre-operation checks.
Regulatory and Safety Compliance
Comply with relevant standards such as ISO 14644-1 (cleanroom classifications), NSF/ANSI 49 (if applicable for biosafety comparisons), and local occupational health and safety regulations. Although vertical laminar flow hoods are not designed for hazardous biological materials (unlike biosafety cabinets), they must be used in accordance with OSHA and institutional safety policies. Conduct a risk assessment prior to use to confirm suitability for the intended application (e.g., sterile compounding, electronics assembly).
Operational Qualification (OQ) and Performance Verification
Perform operational qualification upon installation and at regular intervals (typically annually or per institutional policy). Key tests include:
- Airflow velocity: Measure face velocity using a calibrated anemometer; typical range is 90–100 ft/min.
- HEPA filter integrity testing (DOP/PAO testing): Verify no leaks in the filter system.
- Airflow visualization (smoke test): Confirm unidirectional, laminar airflow across the work surface.
- Particle count testing: Validate cleanliness levels per ISO 14644-1, usually Class 100 (ISO 5).
Document all test results and maintain a compliance log.
Routine Maintenance and Cleaning
Establish a routine maintenance schedule per manufacturer guidelines. Clean the work surface and interior surfaces daily with 70% isopropyl alcohol or approved disinfectant. Replace pre-filters every 3–6 months depending on usage and environment. Schedule HEPA filter replacement based on pressure drop readings or at 3–5 year intervals. Keep a maintenance log including dates, personnel, and actions performed.
Personnel Training and Documentation
Ensure all users are trained on proper use, limitations, and safety procedures. Training should cover avoiding disruptive movements, correct material placement, and contamination prevention. Maintain training records and standard operating procedures (SOPs) that align with regulatory requirements such as cGMP (for pharmaceutical applications) or GLP (for research). Document all operations, maintenance, and performance tests for audit readiness.
Decommissioning and Disposal
When decommissioning, decontaminate the unit per institutional protocols. Safely remove and dispose of HEPA filters in accordance with local hazardous waste regulations if contaminated. Retain all compliance and maintenance records for the required retention period (typically 5–7 years). Notify EHS (Environmental Health and Safety) for proper disposal of electronic components and housing.
Conclusion for Sourcing a Vertical Laminar Flow Hood
Sourcing a vertical laminar flow hood is a critical decision that directly impacts the integrity, safety, and efficiency of clean working environments in laboratories, pharmaceutical settings, microbiology, and other contamination-sensitive applications. After evaluating key factors such as airflow efficiency, HEPA filter quality, construction materials, certifications (e.g., ISO 14644, NSF), noise levels, energy consumption, and after-sales support, it becomes evident that selecting the right unit requires a balance between performance, reliability, and cost-effectiveness.
Vertical laminar flow hoods provide unidirectional airflow from the top down, effectively sweeping away particulates and maintaining a sterile workspace. While they are not suitable for handling hazardous materials (unlike biosafety cabinets), they are ideal for protecting products during processes such as media preparation, sterile sampling, and aseptic assembly.
To ensure optimal performance and compliance, it is essential to source from reputable manufacturers or suppliers who provide proper documentation, calibration, and validation support. Additionally, considering future scalability, ease of maintenance, and compatibility with existing laboratory infrastructure will contribute to long-term operational success.
In conclusion, investing in a high-quality vertical laminar flow hood from a trusted supplier ensures a controlled, particle-free environment, enhances process reliability, and supports adherence to regulatory standards—making it a vital component in maintaining sterility and precision in critical laboratory workflows.