The global body jewelry market is experiencing steady expansion, driven by increasing consumer interest in body modifications and personalized adornments. According to Grand View Research, the global body jewelry market size was valued at USD 4.7 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 8.3% from 2023 to 2030. This growth is supported by rising disposable incomes, heightened social media influence, and growing acceptance of body piercings across diverse demographics. Within this expanding landscape, industrial piercings—particularly those utilizing durable, hypoallergenic materials like surgical steel and titanium—have gained notable popularity. As demand for high-quality, precision-engineered industrial barbells rises, a select group of manufacturers has emerged at the forefront of design, safety, and innovation. Based on production scale, material quality, and market presence, the following three manufacturers stand out in the sword industrial piercing segment.
Top 3 Sword Industrial Piercing Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
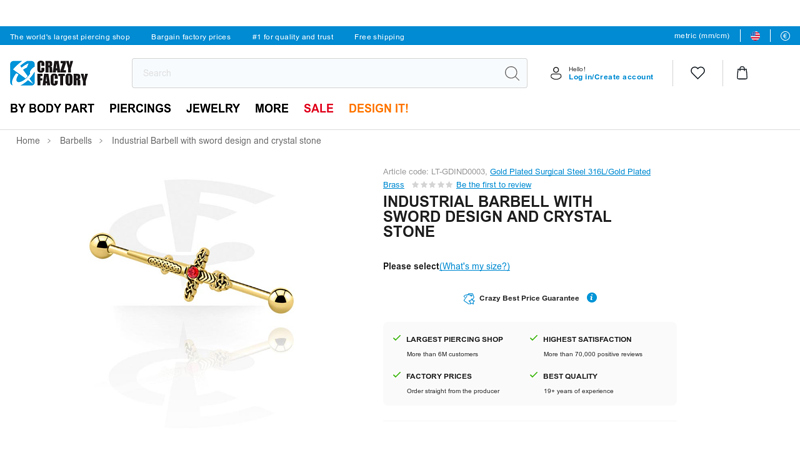
#1 Industrial Barbell with sword design and crystal stone
Domain Est. 2005
Website: crazy-factory.com
Key Highlights: In stock Rating 5.0 2 Order Industrial Barbell with sword design and crystal stone (Gold Plated Surgical Steel 316L/Gold Plated Brass) from the category Barbells online, now….
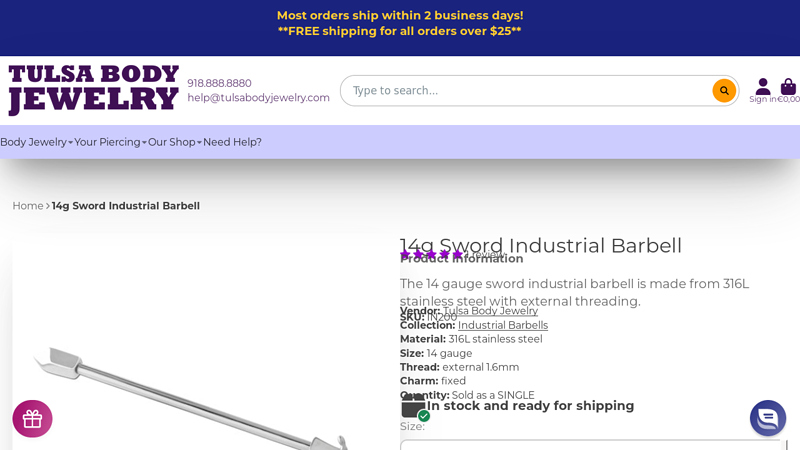
#2 14g Sword Industrial Barbell
Domain Est. 2003
Website: tulsabodyjewelry.com
Key Highlights: In stock Rating 5.0 1 The 14 gauge sword industrial barbell is made from 316L stainless steel with external threading. SKU: IN200. Vendor: Tulsa Body Jewelry….
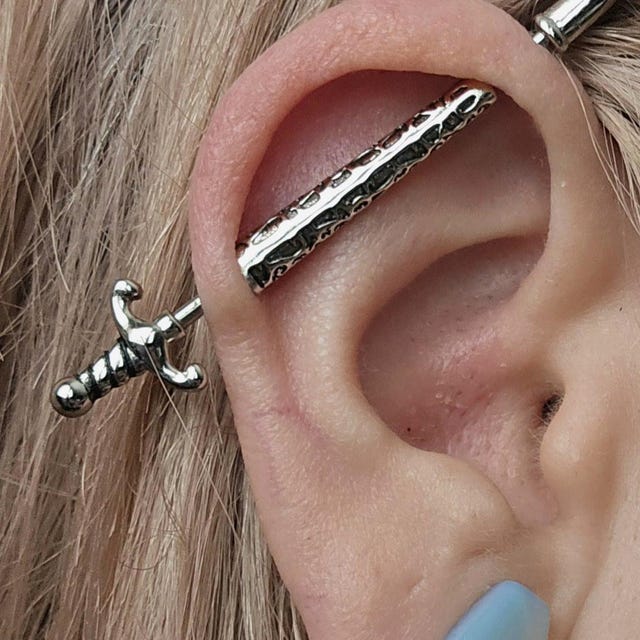
#3 Industrial Piercing illustrations
Domain Est. 2003
Website: shutterstock.com
Key Highlights: Piercing jewelry icons set. This set of icons offers a variety of piercing options, showcasing both the tools used and the body parts that can be adorned….
Expert Sourcing Insights for Sword Industrial Piercing

H2: 2026 Market Trends for Sword Industrial Piercing
As we approach 2026, the body modification industry—particularly industrial piercings—is experiencing a dynamic transformation influenced by cultural shifts, technological advancements, and evolving consumer preferences. While “Sword Industrial Piercing” does not refer to a widely recognized brand or standardized piercing technique, the term can be interpreted as either a stylized or thematic variation of the traditional industrial piercing (commonly placed through the upper ear cartilage), or as a niche trend within the broader piercing market. The following analysis explores key market trends shaping the industrial piercing landscape in 2026, with implications for practitioners, studios, and product manufacturers.
1. Rise of Personalized and Thematic Piercing Designs
By 2026, consumers are increasingly seeking body modifications that reflect individual identity and narrative. Inspired by fantasy, gothic, and cyberpunk aesthetics, “sword-themed” or “edged weapon-inspired” industrial piercings are emerging as symbolic expressions of strength, resilience, and rebellion. Studios are responding by offering custom barbell designs shaped like miniature swords, daggers, or rune-engraved metalwork, often crafted from titanium, surgical steel, or biocompatible gold alloys. This trend is especially popular among Gen Z and younger millennials engaged in alternative fashion communities.
2. Advancements in Piercing Jewelry Materials and Safety
In 2026, there is heightened emphasis on biocompatibility and hypoallergenic materials. The use of ASTM F136 titanium and Parylene-coated jewelry has become standard in premium piercing studios, reducing infection risks and supporting faster healing—critical for complex placements like industrial piercings. Innovations such as antimicrobial surface treatments and smart jewelry (embedded with healing trackers or NFC tags for aftercare information) are gaining traction, positioning high-end piercing studios as wellness-focused service providers.
3. Growth of Social Media-Driven Piercing Culture
Platforms like TikTok, Instagram, and YouTube continue to drive piercing trends. “Sword industrial” variations—featuring dramatic barbells, layered configurations, or dual industrial setups resembling crossed blades—are frequently shared as visual content. This virality has increased demand for unique, photogenic piercings, pushing studios to innovate in design and aftercare education. Influencer collaborations with piercing artists are now common marketing strategies.
4. Mainstream Acceptance and Corporate Evolution
Industrial piercings, once associated with counterculture, are increasingly normalized in professional and social settings. This shift has broadened the customer base to include older demographics and corporate professionals seeking subtle yet meaningful body art. As a result, piercing studios are adopting medical-grade environments and transparent pricing models to appeal to this audience. Chains like Claire’s and specialized urban piercing boutiques are expanding offerings to include premium industrial services.
5. Sustainability and Ethical Sourcing
By 2026, eco-conscious consumers are demanding sustainable practices in the piercing industry. Studios are responding by sourcing recycled metals, using biodegradable packaging, and partnering with ethical jewelers. Some brands are marketing “sword-inspired” pieces made from reclaimed steel or conflict-free materials, appealing to environmentally aware clients.
6. Regulatory and Standardization Developments
Health and safety regulations for body piercing are becoming more rigorous globally. In North America and the EU, certification requirements for piercers and standardized sterilization protocols are being enforced more strictly. This benefits reputable studios offering complex services like industrial piercings, enhancing consumer trust and reducing complications.
Conclusion
The 2026 market for industrial piercings—particularly niche variations like “Sword Industrial Piercing”—is characterized by aesthetic innovation, technological integration, and growing mainstream appeal. While the term may not denote a formal category, it reflects a broader trend toward symbolic, personalized body art. Studios and jewelers that embrace customization, safety, and digital engagement are best positioned to capitalize on this evolving landscape. The future of industrial piercing lies at the intersection of art, identity, and wellness, with themed designs serving as both fashion statements and personal emblems.

Common Pitfalls When Sourcing Industrial Piercings (Quality & Intellectual Property)
When sourcing industrial piercings—particularly from manufacturers in competitive markets—businesses and retailers often encounter challenges related to quality control and intellectual property (IP) protection. Understanding these pitfalls is essential to maintaining brand integrity, customer safety, and legal compliance.
Poor Material Quality and Safety Standards
One of the most frequent issues is receiving industrial piercings made from substandard materials. Many low-cost suppliers use metals that contain nickel or other allergens, or fail to meet ASTM F138 (surgical implant-grade stainless steel) or ISO 10993 biocompatibility standards. This can lead to customer health risks, product returns, and reputational damage.
Inconsistent Manufacturing Precision
Industrial piercings require precise bar lengths, gauge uniformity, and secure threading. Inconsistent machining can result in mismatched barbells, loose ends, or uneven curvature, increasing the risk of irritation or injury. Poor craftsmanship often stems from outdated equipment or lack of quality assurance protocols.
Misrepresentation of Material Claims
Some suppliers falsely advertise products as “surgical steel,” “titanium,” or “hypoallergenic” without providing certification or batch testing. Without third-party verification or material test reports (MTRs), buyers may unknowingly distribute non-compliant jewelry.
Lack of Intellectual Property Due Diligence
When sourcing unique or branded industrial piercing designs, there’s a risk of inadvertently purchasing counterfeit or IP-infringing products. Suppliers may replicate patented closure mechanisms, signature finishes, or proprietary styles without authorization, exposing the buyer to legal liability.
Inadequate Packaging and Labeling Compliance
Non-compliant packaging—missing sterility indicators, ingredient disclosures, or country-of-origin labeling—can violate regulations in markets like the EU (under the Medical Device Regulation) or the U.S. (via the FTC or FDA guidelines). This may result in customs holds or fines.
Absence of Supplier Audits and Traceability
Many buyers rely on supplier claims without conducting onsite audits or requesting supply chain transparency. Without traceability, it’s difficult to verify ethical sourcing, labor practices, or raw material origins—key concerns for conscious consumers and compliance frameworks.
Failure to Secure IP Rights in Contracts
Purchasers often overlook including IP ownership, confidentiality, and non-disclosure clauses in sourcing agreements. This can allow suppliers to replicate designs for competitors or sell identical products under different brands.
Conclusion
To mitigate these risks, buyers should conduct thorough due diligence, request material certifications, perform product testing, audit suppliers, and formalize IP protections in contracts. Partnering with reputable, transparent manufacturers ensures both product quality and legal safety in the competitive body jewelry market.
Logistics & Compliance Guide for Sword Industrial Piercing
This guide outlines the essential logistics and compliance considerations for the safe handling, transportation, and regulatory adherence related to Sword Industrial piercings—both for professionals performing the procedure and individuals managing aftercare.
Product Sourcing and Inventory Management
Ensure all Sword Industrial piercing jewelry is sourced from reputable, certified suppliers adhering to ASTM F138 or ISO 5832-1 standards for implant-grade titanium. Maintain a documented inventory log including batch numbers, sterilization dates, and expiration (if applicable). Store sterile jewelry in sealed, tamper-evident packaging in a clean, dry, temperature-controlled environment to prevent contamination.
Sterilization and Infection Control Protocols
All piercing tools and jewelry must be sterilized using an FDA-cleared autoclave following CDC and APIC guidelines. Use chemical indicators and biological monitoring weekly to validate sterilization effectiveness. Implement a strict single-use policy for needles, gloves, and disposable barriers. Surfaces must be disinfected with hospital-grade EPA-registered disinfectants between clients.
Client Consent and Documentation
Obtain written informed consent prior to any piercing procedure. The consent form must detail risks (e.g., infection, migration, nerve damage), aftercare instructions, healing timelines (typically 6–12 months), and jewelry specifications. Retain client records for a minimum of seven years in compliance with HIPAA and state privacy regulations, ensuring data is securely stored and access is restricted.
Aftercare Product Compliance
Distribute aftercare materials (e.g., saline spray, antiseptic wipes) that are FDA-compliant and clearly labeled with ingredients, usage instructions, and expiration dates. Avoid recommending unapproved home remedies. Provide written and verbal aftercare guidance, emphasizing cleanliness, avoidance of manipulation, and signs of complications requiring medical attention.
Transportation and Travel Considerations
When traveling with piercing tools or jewelry, use lockable, sterile instrument cases that comply with TSA and IATA regulations for medical devices. Carry documentation certifying sterilization and professional use. For pierced individuals, advise avoiding prolonged pressure on the area during air travel and ensuring aftercare supplies are TSA-approved in carry-ons.
Regulatory and Industry Standards
Adhere to all local, state, and federal regulations governing body art establishments, including licensing, zoning, and waste disposal. Follow OSHA Bloodborne Pathogens Standard (29 CFR 1910.1030) for employee safety, including vaccination and exposure control plans. Stay current with APP (Association of Professional Piercers) recommended practices for ethical and safe piercing procedures.
Incident Reporting and Risk Management
Establish a protocol for documenting and reporting adverse events (e.g., infections, allergic reactions). Maintain liability insurance specific to body piercing services. Conduct regular staff training on emergency response, first aid, and compliance updates to minimize risk and ensure consistent operational standards.
Conclusion for Sourcing Industrial Piercing Swords:
Sourcing industrial piercing swords requires a strategic approach focused on quality, safety, compliance, and reliable supply. Successful procurement involves identifying reputable manufacturers or suppliers that adhere to medical-grade standards and use biocompatible materials such as implant-grade titanium (ASTM F136 or F67). It is essential to verify sterilization practices, certifications, and regulatory compliance to ensure the safety of clients in piercing studios.
Building relationships with suppliers who offer consistency in product quality, transparent labeling, and excellent customer support helps maintain professionalism and trust in the body modification industry. Additionally, considering ethical sourcing and sustainable practices can enhance brand reputation and customer loyalty.
In summary, carefully vetting suppliers, prioritizing health and safety standards, and maintaining stringent quality control are critical to successfully sourcing industrial piercing swords. Investing time in due diligence ensures that practitioners deliver safe, high-quality services, ultimately protecting both client well-being and business integrity.