The global audit equipment manufacturing market has experienced robust expansion in recent years, driven by increasing regulatory scrutiny, rising demand for quality assurance across industries, and the adoption of automated inspection technologies. According to a 2023 report by Mordor Intelligence, the non-destructive testing (NDT) and inspection equipment market—central to audit and quality control processes—is projected to grow at a CAGR of 6.8% from 2023 to 2028, reaching an estimated value of USD 22.5 billion by the end of the forecast period. This growth is further propelled by stringent safety standards in aerospace, automotive, and energy sectors, along with digital transformation initiatives integrating IoT and AI into audit systems. As industries prioritize precision and compliance, leading audit manufacturers are innovating rapidly to deliver advanced measurement, calibration, and inspection solutions. Based on market presence, technological capability, and product breadth, the following nine manufacturers stand out as key players shaping the future of industrial and commercial audit systems.
Top 9 Audit Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Factory Audit Inspection Services
Domain Est. 2001
Website: tuvsud.com
Key Highlights: A factory audit is a tailor-made program aimed at assessing a facility’s quality systems, workplace environment and capabilities….
#2 Supplier Audit
Domain Est. 2019
Website: gqc.io
Key Highlights: Factory Audit is the first stage of any supply chain. Aimed to provide an unbiased image of your supply chain. For Factory Audit call us today….
#3 Supplier Audit: Definition, Types & How To Conduct One
Domain Est. 1994
Website: ramp.com
Key Highlights: A supplier audit evaluates a vendor’s facilities, operations, and processes to confirm they meet your quality, contractual, and regulatory ……
#4 Industry Resources on Third
Domain Est. 2000
Website: fda.gov
Key Highlights: We are providing templates that may be used to help receiving facilities, importers, and other stakeholders (eg, audit programs) compare the standards used in ……
#5 apexanalytix
Domain Est. 2001
Website: apexanalytix.com
Key Highlights: Minimize supplier risk with touchless onboarding and risk management. Discover how Enterprise Risk Resolution can help protect your business….
#6 A Supplier Audit Report Template for the FDA
Domain Est. 2007
Website: thefdagroup.com
Key Highlights: Discover best practices for creating effective supplier audit reports in FDA-regulated industries, ensuring quality and compliance with ……
#7 AuditComply
Domain Est. 2014
Website: auditcomply.com
Key Highlights: Designed to streamline operations, unlock deep insights and drive digital transformation, AuditComply enhances efficiency, resilience and safety….
#8 Taking part in a PSCI Audit
Domain Est. 2015
Website: pscinitiative.org
Key Highlights: Suppliers cam make their own request to be audited according to our Audit Framework by asking a member to sponsor their audit or by nominating themselves to the ……
#9 Supplier Audit and Assessment (SAA)
Website: pdrep.csd.disa.mil
Key Highlights: The Supplier Audit and Assessment (SAA) was designed to ensure consistent oversight of the supplier base that provides critical material to the Navy’s ……
Expert Sourcing Insights for Audit
2026 Market Trends in the Audit Industry
The audit profession is undergoing a significant transformation as it approaches 2026, driven by technological innovation, regulatory shifts, and evolving stakeholder expectations. Firms that adapt proactively will be best positioned to thrive in this dynamic environment.
1. Accelerated Adoption of AI and Advanced Analytics
By 2026, artificial intelligence (AI) and machine learning will be deeply embedded in audit workflows. Audit firms will leverage AI for real-time risk assessment, anomaly detection, and predictive analytics, moving beyond sampling to analyze entire datasets. Natural language processing (NLP) will automate document review and contract analysis, significantly improving efficiency and accuracy. Firms investing in proprietary AI audit platforms will gain a competitive edge in delivering faster, more insightful audits.
2. Expansion of Assurance Beyond Financial Statements
Demand for assurance on non-financial information will surge, reflecting growing stakeholder interest in ESG (Environmental, Social, and Governance) performance. By 2026, audit firms will offer standardized assurance services on carbon emissions, diversity metrics, supply chain ethics, and cybersecurity readiness. Regulatory frameworks like the EU’s CSRD (Corporate Sustainability Reporting Directive) will drive compliance-related audits, creating new revenue streams and requiring auditors to develop multidisciplinary expertise.
3. Increased Regulatory Scrutiny and Standardization
Global regulators will intensify oversight of audit quality and independence. In 2026, we expect stricter enforcement of audit rotation rules, enhanced transparency requirements (e.g., audit firm disclosures), and the adoption of international sustainability assurance standards (e.g., ISSB). The U.S. PCAOB and similar bodies worldwide will push for real-time audit data sharing and greater accountability, compelling firms to upgrade their governance and documentation systems.
4. Talent Transformation and Upskilling Imperatives
The auditor of 2026 will need a hybrid skill set combining accounting expertise with data science, technology literacy, and ESG knowledge. Audit firms will prioritize upskilling existing staff and recruiting talent from non-traditional backgrounds. Remote and hybrid work models will persist, enabling access to a global talent pool but requiring new approaches to team collaboration, mentorship, and quality control.
5. Consolidation and Specialization Among Audit Firms
Market pressures, technological costs, and regulatory complexity will drive further consolidation, particularly among mid-tier and regional firms. At the same time, niche audit providers specializing in sectors like fintech, healthcare, or renewable energy will emerge. The Big Four will continue expanding their advisory offerings, blurring the lines between audit and consulting—albeit under tighter regulatory constraints.
6. Blockchain and Distributed Ledger Technology Integration
While still emerging, blockchain will begin to impact audit processes by 2026. Companies using blockchain for transaction recording will enable auditors to verify data immutability and traceability in near real time. Pilot programs for smart contract audits and decentralized finance (DeFi) assurance will gain traction, particularly in tech-forward industries.
In conclusion, the 2026 audit landscape will be defined by technology integration, expanded assurance domains, and heightened accountability. Success will depend on agility, innovation, and the ability to deliver credible, value-added insights in an increasingly complex business environment.
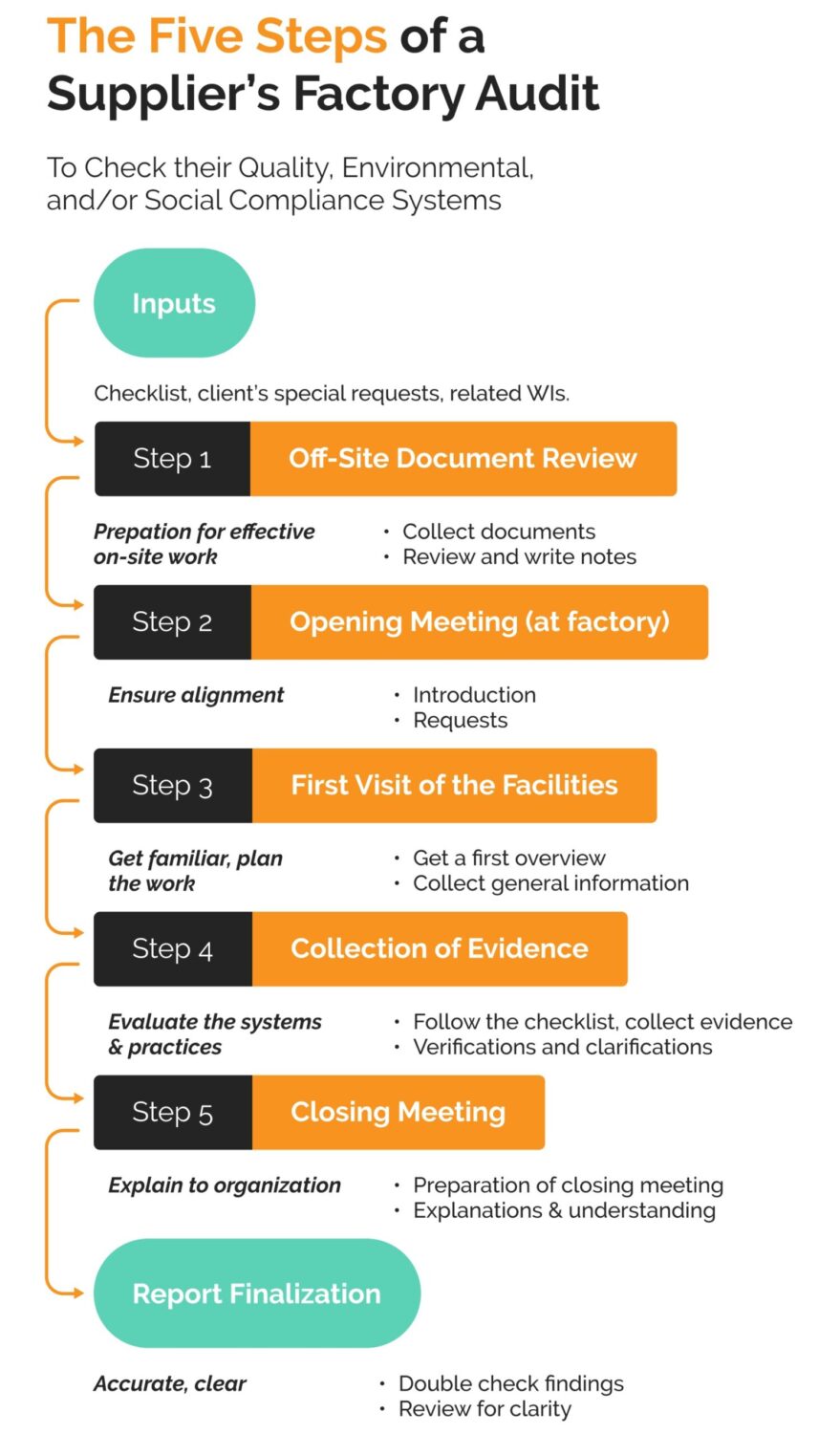
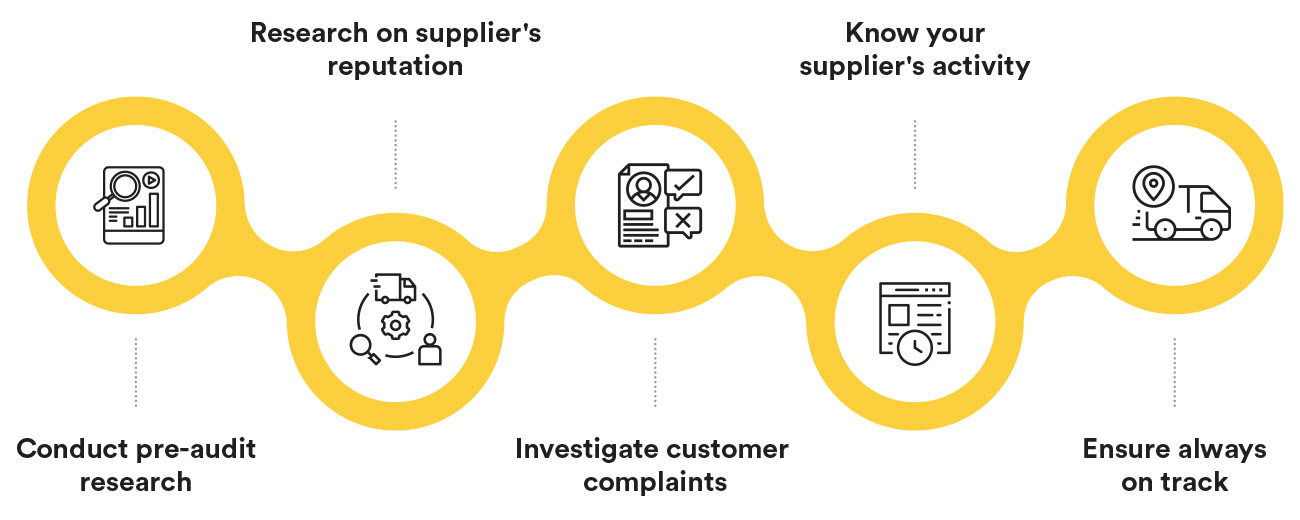
Common Pitfalls in Sourcing Audit (Quality, IP)
Sourcing audits—especially those evaluating quality and intellectual property (IP) compliance—are critical for mitigating risk and ensuring supply chain integrity. However, organizations often encounter recurring pitfalls that undermine audit effectiveness. Recognizing these issues is key to improving oversight and supplier accountability.
Inadequate Scope Definition
Audits may fail to cover all relevant aspects of quality systems or IP protection due to poorly defined objectives. This can result in critical gaps, such as overlooking subcontractors, unapproved production sites, or digital IP handling procedures.
Overreliance on Documentation
Many audits focus excessively on reviewing paperwork without validating actual practices. Suppliers may present pristine records while operational processes deviate significantly, leading to a false sense of compliance.
Infrequent or One-Time Audits
Conducting audits only at onboarding or sporadically allows non-compliance to go undetected for long periods. Continuous or risk-based audit scheduling is often neglected, increasing exposure to quality failures or IP leakage.
Lack of Qualified Auditors
Using auditors without specific expertise in quality standards (e.g., ISO 9001) or IP protection (e.g., trade secret handling, licensing compliance) can result in superficial findings and missed red flags.
Insufficient Follow-Up on Non-Conformances
Identifying issues is only the first step. A common failure is not enforcing corrective action plans (CAPAs) or verifying their implementation, allowing problems to persist.
Cultural or Language Barriers
Global sourcing often involves auditors and suppliers from different regions, leading to miscommunication or reluctance to disclose sensitive information, especially around IP practices.
Supplier Coaching or Audit Preparation
Some suppliers “stage” their facilities or train employees to give scripted responses prior to audits, masking real-world deficiencies in quality control or IP safeguards.
Failure to Audit Sub-Tier Suppliers
Organizations often audit only direct suppliers, ignoring sub-tier vendors who may introduce counterfeit components or mishandle proprietary designs, creating blind spots in the supply chain.
Not Aligning Audits with Risk Profiles
Applying a one-size-fits-all audit approach fails to prioritize high-risk suppliers—such as those handling core technology or in regions with weak IP enforcement—leading to inefficient resource allocation.
Poor Integration with Broader Compliance Programs
Audit findings are sometimes siloed and not shared with procurement, legal, or R&D teams, limiting cross-functional risk mitigation and strategic decision-making.
Avoiding these pitfalls requires a proactive, risk-based audit strategy with trained personnel, robust follow-up mechanisms, and integration across quality, legal, and supply chain functions.

Logistics & Compliance Guide for Audit
This guide outlines the essential logistics and compliance requirements to ensure a smooth and successful audit process. Adherence to these protocols helps maintain data integrity, regulatory compliance, and operational efficiency throughout the audit lifecycle.
Pre-Audit Preparation
- Document Organization: Gather and organize all required records (financial statements, contracts, invoices, policies, and procedures) in a secure, accessible location—physical or digital.
- Audit Scope Confirmation: Verify the audit scope, timeline, and objectives with the auditing team. Ensure all departments involved are aware and prepared.
- Designate Audit Coordinator: Appoint a primary point of contact responsible for facilitating communication, scheduling meetings, and coordinating document requests.
- Compliance Check: Conduct an internal review to confirm adherence to relevant regulations (e.g., SOX, GDPR, HIPAA) and internal policies prior to the audit.
Access & Security Protocols
- Controlled Access: Restrict physical and digital access to audit-related materials to authorized personnel only.
- Data Protection: Ensure sensitive information is encrypted and stored in compliance with data privacy regulations.
- Visitor Management: Register all auditors upon arrival; issue temporary badges and accompany them during site visits as needed.
- Confidentiality Agreements: Confirm that all auditors have signed non-disclosure agreements (NDAs) before accessing proprietary information.
During the Audit
- Scheduled Meetings: Provide timely access to key personnel for interviews and discussions. Prepare staff with talking points and relevant documentation.
- Document Requests: Respond promptly to document requests. Maintain a log of all submissions for tracking and accountability.
- Real-Time Support: The audit coordinator should be available to address questions, clarify processes, and facilitate information flow.
- Compliance Demonstration: Be prepared to demonstrate compliance through process walkthroughs, system access, and policy references.
Post-Audit Procedures
- Findings Review: Schedule a closing meeting to review preliminary findings and clarify any discrepancies.
- Response Documentation: Prepare formal responses to audit observations, including corrective action plans and timelines.
- Evidence Retention: Archive all audit-related communications, reports, and supporting documentation per retention policies.
- Follow-Up Actions: Implement recommended corrective actions and conduct follow-up reviews to ensure compliance.
Regulatory & Internal Compliance
- Regulatory Alignment: Ensure all practices align with applicable legal and industry standards.
- Internal Controls: Maintain robust internal controls and routinely test their effectiveness.
- Audit Trail Maintenance: Keep detailed logs of transactions, access, and changes to critical systems to support traceability.
- Training & Awareness: Provide regular compliance training to staff to reinforce audit readiness and regulatory obligations.
Adhering to this logistics and compliance guide ensures preparedness, minimizes disruptions, and supports a transparent, efficient audit process.
Conclusion for Supplier Audit
The supplier audit has been conducted in accordance with established quality, compliance, and operational standards. Based on the findings, [Supplier Name] demonstrates a strong commitment to quality management, regulatory compliance, and continuous improvement. Key processes are well-documented, and the facility meets the necessary requirements in areas such as manufacturing practices, traceability, health and safety, and environmental management.
While minor non-conformances were identified, they are considered manageable and do not impact the overall integrity of supplied products. Corrective actions have been proposed by the supplier, and a follow-up timeline has been agreed upon to ensure timely resolution.
Overall, [Supplier Name] is deemed a reliable and capable supplier, aligned with our organization’s standards and expectations. Continued collaboration, supported by periodic audits and performance monitoring, will further strengthen this partnership and ensure sustained compliance and product quality.
Recommendation: Approved for continued supply, subject to confirmation of corrective actions within the agreed timeframe.