The global surgical retractors market is experiencing steady growth, driven by rising surgical volumes, technological advancements, and increasing demand for minimally invasive procedures. According to Grand View Research, the global surgical instruments market was valued at USD 19.2 billion in 2022 and is expected to expand at a compound annual growth rate (CAGR) of 7.8% from 2023 to 2030. A key segment within this market is Sims retractors—widely used in gynecological and abdominal surgeries for their effective tissue retraction capabilities. With expanding healthcare infrastructure and heightened focus on surgical precision, demand for high-quality Sims retractors is on the rise. As providers seek reliable, durable, and ergonomically designed instruments, a handful of manufacturers have emerged as leaders in innovation, production quality, and global reach. Based on market presence, product quality, and industry reputation, here are the top 5 Sims retractor manufacturers shaping the future of surgical care.
Top 5 Sims Retractor Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
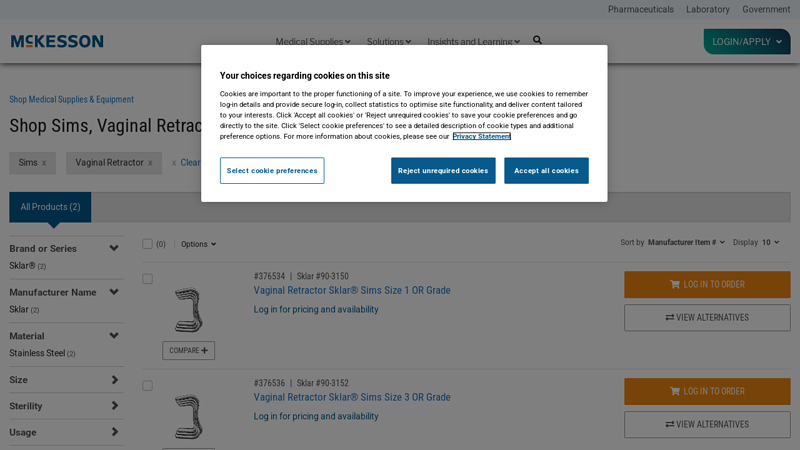
#1 Shop Sims, Vaginal Retractor
Domain Est. 1994
Website: mms.mckesson.com
Key Highlights: Shop Sims, Vaginal Retractor · Manufacturer Name. Sklar (2) · Brand or Series. Sklar® (2) · Material. Stainless Steel (2) · Size. Size 1 (1) · Size 3 (1) · Sterility….
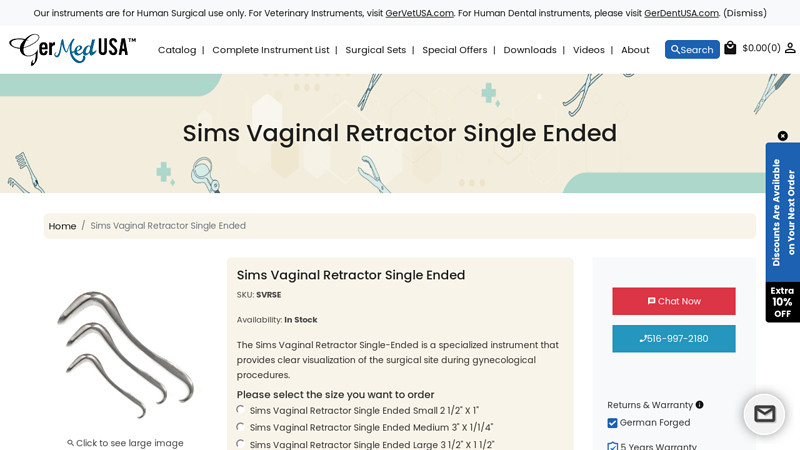
#2 Sims Vaginal Retractor Single-Ended
Domain Est. 2004
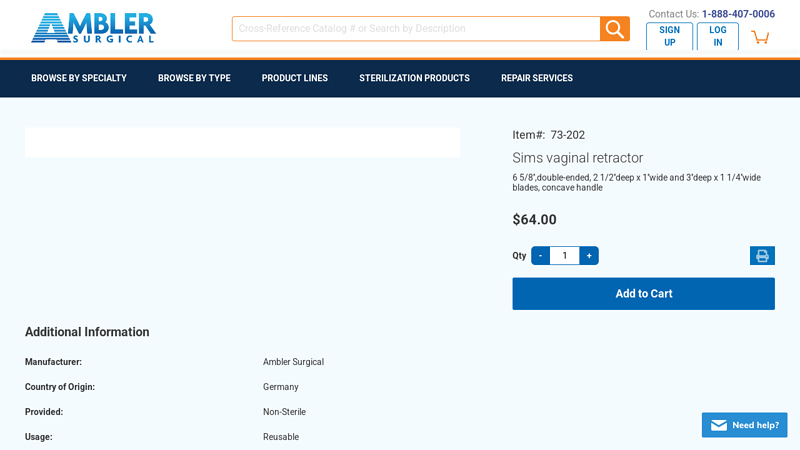
#3 Sims vaginal retractor, 6 5/8” 73
Domain Est. 2003
Website: amblersurgical.com
Key Highlights: In stock $47.93 deliverySims vaginal retractor 6 5/8”,double-ended, 2 1/2”deep x 1”wide and 3”deep x 1 1/4”wide blades, concave handle. $64.00. Item# 73-202….
#4 Sims Vaginal Retractor
Domain Est. 2006
Website: artisanmed.com
Key Highlights: Sims Vaginal Retractor Blade Small 2.5″,7.25″ Sims Vaginal Retractor Blade Small 2.5′′,7.25′′ RET-001 Read more Sims Vaginal Retractor Double End Large 5.5″…
#5 Sims Vaginal Retractor, Grip Handle
Domain Est. 2006
Expert Sourcing Insights for Sims Retractor

H2: Projected Market Trends for Sims Vaginal Retractors in 2026
Based on current healthcare dynamics, technological evolution, and industry trajectories, the market for Sims Vaginal Retractors is expected to exhibit several key trends by 2026:
1. Growth Driven by Rising Gynecological & Surgical Volumes: The primary driver will be the increasing global prevalence of gynecological conditions (e.g., fibroids, incontinence, pelvic organ prolapse) and age-related urological needs. An aging population, particularly in developed economies, will lead to higher demand for diagnostic procedures (like colposcopy) and surgical interventions (such as hysterectomies and incontinence repairs), directly increasing the need for reliable retraction tools like the Sims retractor.
2. Shift Towards Reusable Instruments Amidst Sustainability Pressures: While single-use plastics offer convenience, significant pressure from hospitals and governments to reduce healthcare waste and carbon footprints will accelerate the shift back to high-quality, durable reusable instruments. By 2026, robust, autoclavable stainless steel Sims retractors from established manufacturers are expected to regain market share, supported by improved sterilization protocols and lifecycle cost analysis favoring reusables.
3. Integration with Minimally Invasive & Ambulatory Settings: The trend towards minimally invasive gynecological surgery (MIS) and outpatient procedures will influence retractor design and usage. Sims retractors will remain essential in procedures like colposcopy and certain laparoscopic-assisted vaginal surgeries performed in ambulatory surgery centers (ASCs). Demand will grow in the ASC sector as more gynecological procedures shift out of traditional hospitals.
4. Focus on Ergonomics and Operator Comfort: Surgeon fatigue and repetitive strain injuries are growing concerns. Manufacturers will increasingly emphasize ergonomic design in Sims retractors by 2026, focusing on optimized handle shape, weight distribution, and non-slip grips. This enhances surgical precision, reduces operator fatigue during prolonged procedures, and improves overall user satisfaction.
5. Regional Market Divergence: Market growth will be uneven. North America and Europe will see steady growth driven by advanced healthcare systems, high procedure volumes, and stringent regulatory standards emphasizing reusable instruments and sterilization. The Asia-Pacific region, particularly countries like India and China, will experience the fastest growth due to expanding healthcare access, rising disposable incomes, increasing awareness of women’s health, and growing numbers of trained gynecologists, albeit with a mix of reusable and single-use product adoption.
6. Supply Chain Resilience and Localized Manufacturing: Lessons from recent global disruptions will lead to a push for more resilient supply chains. By 2026, there may be increased regional manufacturing or sourcing of surgical instruments, including Sims retractors, to mitigate risks and ensure consistent supply, potentially benefiting local manufacturers in key markets.
7. Impact of Value-Based Healthcare: As healthcare systems increasingly adopt value-based models, purchasing decisions will focus on total cost of ownership and long-term value. This favors high-quality reusable Sims retractors with proven durability and lower lifetime costs compared to single-use alternatives, despite a higher initial price point.
In conclusion, the Sims retractor market in 2026 will be characterized by steady growth fueled by demographic and procedural trends, a strong counter-movement towards sustainable reusables, innovation in ergonomics, and significant regional variations shaped by healthcare infrastructure and economic factors.

Common Pitfalls When Sourcing Sims Retractors (Quality, IP)
Sourcing Sims retractors—especially from international or non-traditional suppliers—can present significant risks related to both product quality and intellectual property (IP). Being aware of these pitfalls is essential for medical device distributors, hospitals, and procurement teams to ensure patient safety and legal compliance.
Poor Manufacturing Quality and Material Standards
One of the most frequent issues when sourcing Sims retractors is encountering substandard manufacturing quality. Low-cost suppliers may use inferior stainless steel grades that corrode easily, lack proper heat treatment, or have rough edges and burrs that increase the risk of tissue trauma. Poorly finished instruments can also harbor bacteria, compromising sterility and patient safety. Additionally, inconsistent dimensions or weak spring mechanisms in double-ended models can impair functionality during surgical procedures.
Non-Compliance with Medical Device Regulations
Many off-market Sims retractors fail to meet international regulatory standards such as FDA 510(k) clearance, CE marking under the EU MDR, or ISO 13485 certification. Sourcing non-compliant devices exposes healthcare providers and distributors to legal liability, product recalls, and potential bans. Devices lacking proper documentation may also be detained at customs, disrupting supply chains.
Risk of Intellectual Property Infringement
Sims retractors, particularly those bearing the names or logos of established medical brands (e.g., “Zimmer,” “Aesculap,” or “B. Braun”), may be counterfeit or unauthorized copies. Distributing or using such instruments can lead to IP infringement claims, including trademark and design patent violations. Even if the device functions adequately, unauthorized use of protected branding or design features can result in lawsuits, fines, and reputational damage.
Inadequate Supplier Verification and Traceability
Working with suppliers who lack transparency in their manufacturing processes increases the risk of receiving counterfeit or reconditioned instruments labeled as new. Without robust supplier vetting—including on-site audits, quality certifications, and supply chain traceability—it’s difficult to ensure authenticity and consistency. Unverified suppliers may also provide falsified documentation, further complicating compliance efforts.
Lack of Sterility and Repackaging Risks
Some sourced Sims retractors are sold as “pre-sterilized” but come from facilities without validated sterilization processes. Others may be reprocessed or repackaged without proper controls, increasing infection risks. Ensuring instruments are sterile and packaged according to ISO 11135 or ISO 11137 standards is critical, particularly for single-use or single-patient devices.
Conclusion
To mitigate these pitfalls, organizations should prioritize sourcing from reputable, certified suppliers, verify regulatory compliance and IP status, and conduct regular quality audits. Investing in due diligence protects patient outcomes, maintains regulatory standing, and safeguards against legal and financial risks associated with substandard or infringing medical devices.

Logistics & Compliance Guide for Sims Retractor
Product Overview
The Sims Retractor is a medical surgical instrument designed for use in gynecological and rectal procedures to provide optimal exposure by retracting tissue. It is typically made from high-grade stainless steel and reusable after proper sterilization. Understanding logistics and compliance requirements ensures safe handling, distribution, and clinical use.
Regulatory Classification
The Sims Retractor is classified as a Class I medical device under the U.S. Food and Drug Administration (FDA) regulations, as defined in 21 CFR 878.4800 (Gynecologic Surgical Instruments). This classification indicates a low to moderate risk and typically requires general controls such as registration, labeling, and adherence to Good Manufacturing Practices (GMP). Similar classifications apply under the EU Medical Device Regulation (MDR) (Regulation (EU) 2017/745), where it may fall under Class I non-sterile or Class Is (sterile) depending on supply status.
Labeling Requirements
All packaging and labeling must comply with FDA 21 CFR Part 801 and EU MDR Annex I. Required information includes:
– Device name (“Sims Vaginal Speculum” or equivalent)
– Manufacturer name and address
– Unique Device Identifier (UDI) for traceability (mandatory in U.S. and EU)
– Intended use statement
– Reusable device symbol (if applicable)
– Sterility status (if supplied sterile)
– Instructions for use (IFU) including reprocessing guidelines
Packaging and Shipping
Sims Retractors must be packaged to prevent contamination and physical damage during transit. Use tamper-evident, sterile barrier systems if supplied sterile. For non-sterile units, ensure packaging protects against moisture and corrosion. Ship using validated transport methods that maintain product integrity. Include appropriate hazard labels if applicable (e.g., sharp instruments).
Import/Export Regulations
When shipping internationally, ensure compliance with destination country regulations. Key considerations include:
– U.S. exports: Comply with FDA export requirements under 21 CFR Part 807 and U.S. Customs and Border Protection (CBP) rules
– EU imports: Ensure CE marking and EU Representative registration (if applicable under MDR)
– Other regions: Verify local medical device registration (e.g., Health Canada, TGA in Australia, NMPA in China)
Reprocessing and Reuse Compliance
As a reusable device, Sims Retractors must be cleaned, disinfected, and sterilized according to validated protocols prior to each use. Follow manufacturer IFUs and comply with:
– CDC and AAMI ST79 guidelines for steam sterilization
– OSHA Bloodborne Pathogens Standard (29 CFR 1910.1030) for handling contaminated devices
– Joint Commission standards for reprocessing in healthcare facilities
Storage Conditions
Store Sims Retractors in a clean, dry, and temperature-controlled environment. Avoid exposure to corrosive chemicals or high humidity to prevent rust and degradation. Keep in original packaging until ready for reprocessing or use.
Documentation and Traceability
Maintain detailed records including:
– Device UDI and lot numbers
– Distribution logs
– Certificate of Conformance (CoC)
– Regulatory approvals (e.g., FDA listing, CE Certificate)
– Reprocessing validation documentation
These records support compliance audits, recalls, and adverse event reporting.
Adverse Event Reporting
Manufacturers and user facilities must report device-related adverse events:
– In the U.S.: Report to FDA via MedWatch (Form 3500A) under Medical Device Reporting (MDR) regulations (21 CFR Part 803)
– In the EU: Report via Eudamed under MDR vigilance procedures (Article 87)
Training and Staff Compliance
Ensure staff involved in handling, reprocessing, or using Sims Retractors are trained on:
– Infection control protocols
– Reprocessing procedures
– Regulatory requirements
– Incident reporting processes
Regular training updates are essential to maintain compliance and patient safety.
Conclusion
Proper logistics and compliance management for the Sims Retractor ensures patient safety, regulatory adherence, and efficient clinical use. Always verify current regulations in your jurisdiction and update procedures accordingly.
Conclusion for Sourcing SIMS Retractor:
After a thorough evaluation of suppliers, product quality, cost-effectiveness, and compliance with medical device standards, sourcing the SIMS retractor from a reputable and certified manufacturer ensures reliability, durability, and patient safety. It is recommended to establish a long-term partnership with a supplier that adheres to ISO 13485 standards, offers consistent product quality, and provides responsive customer support. Additionally, considering factors such as lead times, scalability, and regulatory documentation will support seamless integration into clinical workflows. Ultimately, strategic sourcing of the SIMS retractor enhances surgical efficiency and contributes to improved patient outcomes in gynecological and abdominal procedures.