The global private label supplements market is experiencing robust expansion, driven by increasing consumer demand for personalized wellness solutions and the growing popularity of retail and e-commerce platforms offering customized health products. According to Grand View Research, the global dietary supplements market was valued at USD 151.9 billion in 2022 and is expected to grow at a compound annual growth rate (CAGR) of 8.9% from 2023 to 2030. A significant portion of this growth is attributed to private label manufacturing, as more brands seek cost-effective, scalable partnerships to bring specialized supplement formulations to market quickly. Mordor Intelligence further supports this trend, projecting a CAGR of over 7.5% for the dietary supplements sector through 2028, with private label providers playing a pivotal role in meeting demand across vitamins, sports nutrition, and herbal supplements. As competition intensifies and consumers prioritize transparency and quality, selecting the right private label manufacturer has become a strategic imperative for brands aiming to capture market share in this dynamic industry.
Top 10 Private Labeling Supplements Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Private Label Supplement Manufacturer
Domain Est. 1998
Website: davincilabs.com
Key Highlights: We are a private label supplement manufacturer with 50 years of success at delivering fast, easy growth solutions for businesses and practices….
#2 Private Label Skincare & Dietary Supplements Manufacturer
Domain Est. 2012
Website: tci-bio.com
Key Highlights: TCI is a global leader in private label supplements and beauty CDMO+ services, offering skincare and functional health food solutions….
#3 Garmon Corp.
Domain Est. 2017
Website: garmoncorp.com
Key Highlights: Garmon Corporation has been the industry leader in manufacturing private-label pet supplements for over 25 years. Contact us today about your project!…
#4 BL Bio Lab
Domain Est. 2018
Website: blbiolab.com
Key Highlights: BL Bio Lab is a manufacturer of private label supplements in Florida, USA providing capsules, tablets, powders, liquids and other products….
#5 SMP Nutra
Domain Est. 2019
Website: smpnutra.com
Key Highlights: SMP Nutra is a nutraceutical dietary supplement & vitamin manufacturer based in the USA. We are your full turn-key partner for supplements….
#6 Enhanced Labs
Domain Est. 2024
Website: enhanced-labs.com
Key Highlights: As a dietary supplement manufacturer, we create premium consumable products, deliver expert private label manufacturing, and build impactful ……
#7 IVC Private Label Program
Domain Est. 1997
Website: ivcinc.com
Key Highlights: Build your brand with IVC’s private label program—science-backed supplements trusted by top U.S. retailers and global consumers….
#8 Vitalabs
Domain Est. 1997
Website: vitalabs.com
Key Highlights: Vitalabs is a private labeler of dietary supplements including of stock and custom formulations. Vitalabs does not sell any products directly to the consuming ……
#9 NOW Foods Private Label
Domain Est. 2004
Website: nowprivatelabel.com
Key Highlights: For more than 50 years, companies have trusted NOW to provide superior quality supplements, sports nutrition, essential oils, sweeteners and more as private ……
#10 Eagle Labs Inc
Domain Est. 2005
Website: eaglelabsinc.com
Key Highlights: We provide complete, flexible formulating and manufacturing services for private label lines. We also specialize in working with businesses who are interested ……
Expert Sourcing Insights for Private Labeling Supplements
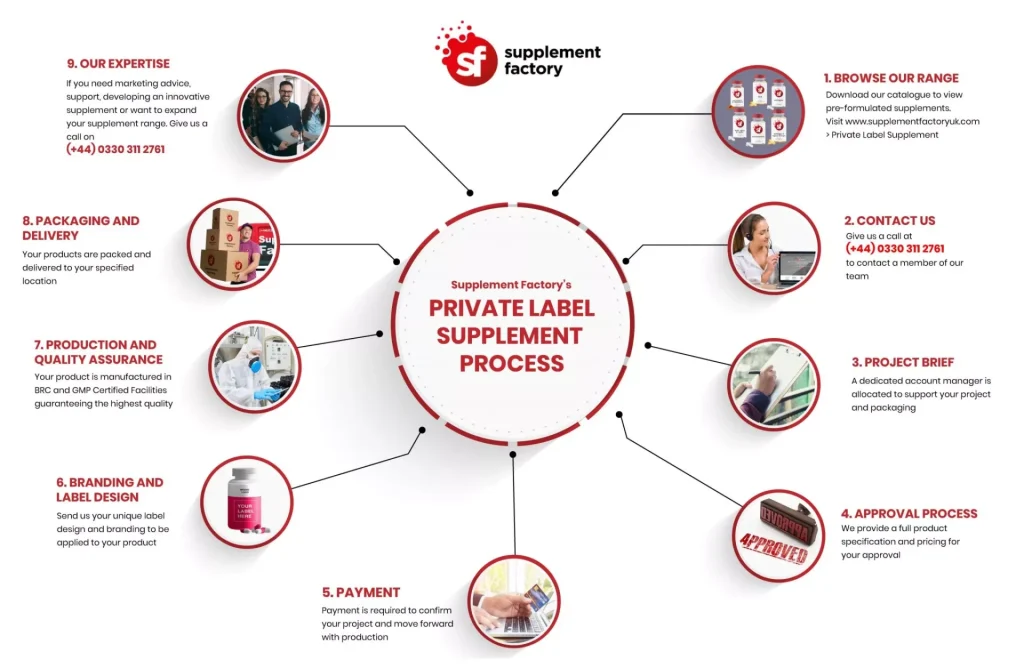
H2: 2026 Market Trends for Private Labeling Supplements
The private label supplements market is poised for significant transformation by 2026, driven by shifting consumer preferences, technological advancements, and evolving regulatory landscapes. As demand for personalized health solutions grows, private labeling in the supplement industry is becoming a strategic advantage for retailers, e-commerce brands, and wellness startups. The following key trends are expected to shape the private labeling supplements market in 2026:
1. Personalization and Custom Formulations
By 2026, consumers will increasingly seek supplements tailored to their individual health needs, genetic profiles, and lifestyle goals. Private label manufacturers are responding with scalable customization options, including gender-specific, age-targeted, and condition-focused formulations. Brands leveraging AI-driven assessments and at-home testing kits will gain a competitive edge by offering hyper-personalized supplement regimens under their own labels.
2. Clean Label and Transparency Demand
Consumers are demanding greater transparency in ingredient sourcing, manufacturing practices, and labeling. In 2026, successful private label supplements will emphasize clean, non-GMO, vegan, and allergen-free formulations. QR codes on packaging that trace ingredient origins and provide third-party testing results will become standard, enhancing trust and brand loyalty.
3. Rise of E-commerce and DTC (Direct-to-Consumer) Models
The shift toward online shopping will continue to accelerate, with DTC brands dominating the private label supplement space. By 2026, these brands will leverage social media, influencer marketing, and subscription models to drive customer acquisition and retention. Private label manufacturers will need to support seamless integration with e-commerce platforms and offer white-label digital marketing support.
4. Expansion of Niche and Functional Categories
Beyond traditional multivitamins, private label growth will be driven by niche categories such as adaptogens, nootropics, gut health (probiotics and prebiotics), immune support, and sleep enhancement. These functional supplements align with holistic wellness trends and allow brands to differentiate themselves in a crowded market.
5. Sustainability and Eco-Conscious Packaging
Environmental concerns will play a larger role in purchasing decisions. By 2026, private label supplement brands that adopt sustainable packaging—such as biodegradable, refillable, or recyclable materials—will appeal to eco-conscious consumers. Manufacturers offering green certifications and carbon-neutral production will be preferred partners.
6. Regulatory and Compliance Challenges
As regulatory scrutiny increases globally, particularly from the FDA and EFSA, private label brands must ensure strict compliance with labeling, claims, and ingredient regulations. Investment in regulatory consulting and adherence to GMP (Good Manufacturing Practices) will be essential to avoid recalls and legal issues.
7. Global Market Expansion and Emerging Markets
While North America and Europe remain strong markets, growth in Asia-Pacific, Latin America, and the Middle East will present new opportunities for private label expansion. Localized formulations that cater to regional health concerns and cultural preferences will be critical for success.
8. Advancements in Manufacturing Technology
Innovations in encapsulation, nanoemulsions, and bioavailability enhancement will enable private label brands to offer more effective and rapidly absorbed supplements. Contract manufacturers investing in R&D and automation will attract premium clients seeking differentiation.
In conclusion, the 2026 private label supplements market will be defined by personalization, transparency, digital engagement, and sustainability. Brands that partner with agile, compliant, and innovative manufacturers—and stay attuned to consumer wellness trends—will be best positioned for long-term growth and market leadership.

Common Pitfalls in Sourcing Private Label Supplements: Quality and Intellectual Property Risks
Sourcing private label supplements offers a fast track to launching your brand, but it comes with significant risks—particularly in quality control and intellectual property (IP) protection. Overlooking these areas can damage your reputation, lead to legal issues, or result in product failures. Here are the most common pitfalls to avoid:
Quality Control Challenges
One of the biggest risks in private labeling is inconsistent or subpar product quality, often due to lack of oversight and transparency in the supply chain.
1. Inadequate Manufacturer Vetting
Choosing a manufacturer based solely on low pricing or quick turnaround without verifying certifications (such as cGMP—Current Good Manufacturing Practices) increases the risk of contamination, mislabeling, or ineffective formulations. Always audit facilities, request third-party lab results, and ensure compliance with FDA or regional regulatory standards.
2. Poor Ingredient Sourcing and Transparency
Many private label suppliers source raw materials from unknown or unverified vendors. Without full ingredient traceability, brands risk using low-grade, adulterated, or allergenic components. Demand detailed Certificates of Analysis (CoAs) and insist on transparency about ingredient origins.
3. Inconsistent Batch-to-Batch Quality
Without strict quality assurance protocols, you may experience variations in potency, dosage, or appearance across product batches. This inconsistency damages consumer trust. Ensure your supplier conducts routine in-process and finished product testing.
4. Labeling and Regulatory Non-Compliance
Misleading claims, incorrect Supplement Facts panels, or failure to list allergens can trigger FDA warnings or product recalls. Verify that your supplier follows labeling regulations in your target market and review all packaging content carefully.
Intellectual Property Risks
When private labeling, your brand identity and product uniqueness are at risk if IP protections are not properly managed.
1. Lack of Formulation Ownership
Many private label manufacturers use “stock” formulas that are sold to multiple brands. If you don’t own or license the exclusive rights to a formulation, competitors can sell the identical product under different labels—eroding your differentiation.
2. Shared or Copycat Packaging and Branding
Some suppliers reuse molds, labels, or packaging designs across clients. This can result in your product looking identical to others on the market. Always ensure your packaging design is custom and protected through trademarks or design rights.
3. Weak Contractual Protections
Generic supplier agreements may not include clauses that protect your IP, such as exclusivity, confidentiality, or ownership of custom formulations. Work with legal counsel to draft contracts that clearly assign IP rights to your brand.
4. Risk of Idea Theft
Sharing unique product concepts with unvetted manufacturers could lead to your ideas being replicated or sold to others. Use Non-Disclosure Agreements (NDAs) before discussing proprietary formulations or branding strategies.
Conclusion
To mitigate these pitfalls, conduct thorough due diligence on manufacturers, demand transparency in ingredients and testing, and secure strong legal protections for your brand and formulations. Investing time upfront in vetting and contracts helps safeguard your brand’s reputation, ensures product quality, and protects your intellectual property in the competitive supplement market.

Logistics & Compliance Guide for Private Labeling Supplements
Understanding Private Labeling in the Supplement Industry
Private labeling supplements involves branding and selling products manufactured by a third-party supplier under your company’s name. This business model allows entrepreneurs and companies to enter the dietary supplement market quickly without investing in production facilities. However, it comes with significant logistical and regulatory responsibilities to ensure product quality, safety, and compliance.
Regulatory Framework: FDA and Dietary Supplements
The U.S. Food and Drug Administration (FDA) regulates dietary supplements under the Dietary Supplement Health and Education Act (DSHEA) of 1994. As a private labeler, you are considered a “responsible party” and must comply with current Good Manufacturing Practices (cGMPs) for dietary supplements (21 CFR Part 111). Key requirements include:
– Ensuring product identity, purity, strength, and composition.
– Verifying ingredient safety and proper labeling.
– Maintaining records of manufacturing, packaging, and distribution.
– Registering your facility with the FDA and listing your products.
Choosing a Compliant Contract Manufacturer
Selecting the right manufacturer is critical. Look for:
– FDA-registered facilities that follow cGMPs.
– Third-party certifications (e.g., NSF, UL, or USP verification).
– Experience in producing your type of supplement (capsules, powders, gummies, etc.).
– Transparent documentation and testing protocols.
Ensure your contract clearly outlines quality control, labeling responsibilities, and ownership of formulations.
Ingredient Sourcing and Safety
All ingredients must be Generally Recognized as Safe (GRAS) or comply with New Dietary Ingredient (NDI) notification requirements if applicable. You must:
– Verify the safety and legality of each ingredient.
– Obtain Certificates of Analysis (CoA) from suppliers.
– Avoid banned substances or unapproved drug ingredients (e.g., ephedra, DMAA).
– Monitor the FDA’s list of tainted supplement products.
Labeling Compliance and Requirements
Supplement labels must meet strict FDA guidelines. Required elements include:
– Statement of identity (e.g., “Dietary Supplement”).
– Net quantity of contents.
– Supplement Facts panel with serving size, ingredients, and amounts.
– Proprietary blend disclosure (if applicable).
– Manufacturer/distributor name and address.
– Daily Value percentages (if established).
Avoid making unauthorized disease claims (e.g., “cures arthritis”)—only structure/function claims are allowed (e.g., “supports joint health”), and they must include a disclaimer:
“These statements have not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure, or prevent any disease.”
Product Testing and Quality Assurance
To ensure safety and label accuracy, implement routine testing for:
– Potency and ingredient verification.
– Contaminants (heavy metals, microbes, pesticides).
– Stability and shelf-life.
Work with an independent laboratory to conduct batch testing, and maintain records for FDA inspections.
Packaging and Storage Logistics
Proper packaging protects product integrity and ensures compliance:
– Use tamper-evident seals and child-resistant packaging if required.
– Store products in a clean, temperature-controlled environment.
– Track inventory using FIFO (First In, First Out) to prevent expired stock.
– Ensure packaging materials comply with food-grade safety standards.
Import and Export Considerations
If sourcing or selling internationally:
– Comply with import regulations of the destination country (e.g., Health Canada, EFSA in Europe).
– Verify foreign manufacturers meet U.S. cGMP standards.
– Use FDA Prior Notice for imported supplements.
– Be aware of restricted ingredients in target markets.
Adverse Event Reporting and Recall Preparedness
As a private labeler, you must:
– Establish a system to receive and document consumer complaints.
– Report serious adverse events to the FDA within 15 days.
– Develop a recall plan in case of contamination, mislabeling, or safety issues.
– Notify the FDA and relevant parties promptly during a recall.
Intellectual Property and Brand Protection
Protect your brand by:
– Trademarking your product name and logo.
– Ensuring your formulations are unique or properly licensed.
– Using contracts to prevent manufacturer misuse of your branding.
Conclusion
Successfully private labeling supplements requires a balance of smart logistics and rigorous compliance. Partnering with reputable manufacturers, adhering to FDA regulations, and maintaining high-quality standards are essential to building a trustworthy and sustainable brand in the competitive supplement market.
In conclusion, sourcing private label supplements presents a significant opportunity for entrepreneurs and businesses looking to enter or expand within the health and wellness industry efficiently and cost-effectively. By partnering with a reputable manufacturer, brands can leverage pre-developed formulations, streamlined production processes, and regulatory compliance support—reducing both time-to-market and upfront investment. However, success hinges on thorough due diligence, including evaluating manufacturing standards (e.g., GMP certification), product quality, ingredient transparency, and the flexibility of branding and packaging options. Building strong relationships with trustworthy suppliers and maintaining clear communication ensures consistent product quality and customer satisfaction. Ultimately, private labeling empowers businesses to establish a unique market presence while focusing on marketing, customer acquisition, and brand growth, making it a strategic pathway to long-term success in the booming supplement industry.