The global private label vitamins market is experiencing robust growth, driven by rising consumer demand for affordable, high-quality nutritional supplements and the expansion of retail and e-commerce distribution channels. According to Mordor Intelligence, the global dietary supplements market was valued at USD 151.9 billion in 2023 and is projected to grow at a CAGR of 9.7% through 2029, with private label brands capturing an increasingly significant share. This surge is fueled by heightened health awareness, personalized nutrition trends, and the growing preference for store-brand supplements among price-conscious consumers. As retailers and startups seek reliable manufacturing partners, the need for transparent, scalable, and compliant private label vitamin manufacturers has never been greater. In this evolving landscape, identifying manufacturers with proven track records in quality, innovation, and regulatory adherence is critical for brands aiming to compete effectively.
Top 7 Private Label Vitamins Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Private Label Supplement Manufacturer
Domain Est. 1998
Website: davincilabs.com
Key Highlights: We are a private label supplement manufacturer with 50 years of success at delivering fast, easy growth solutions for businesses and practices….
#2 Eagle Labs Inc
Domain Est. 2005
Website: eaglelabsinc.com
Key Highlights: Eagle Labs Inc. manufactures the highest quality and most rigorously tested nutritional supplements and skin care products in the USA….
#3 Private Label Skincare & Dietary Supplements Manufacturer
Domain Est. 2012
Website: tci-bio.com
Key Highlights: TCI is a global leader in private label supplements and beauty CDMO+ services, offering skincare and functional health food solutions….
#4 SMP Nutra
Domain Est. 2019
Website: smpnutra.com
Key Highlights: SMP Nutra is a nutraceutical dietary supplement & vitamin manufacturer based in the USA. We are your full turn-key partner for supplements….
#5 Vitalabs
Domain Est. 1997
Website: vitalabs.com
Key Highlights: Vitalabs offers private label vitamins, weight loss, dietary supplements, and single serving packets of vitamins, minerals, protein powder, and herbs, ……
#6 IVC Private Label Program
Domain Est. 1997
Website: ivcinc.com
Key Highlights: Build your brand with IVC’s private label program—science-backed supplements trusted by top U.S. retailers and global consumers….
#7 NOW Foods Private Label
Domain Est. 2004
Website: nowprivatelabel.com
Key Highlights: For more than 50 years, companies have trusted NOW to provide superior quality supplements, sports nutrition, essential oils, sweeteners and more as private ……
Expert Sourcing Insights for Private Label Vitamins
2026 Market Trends for Private Label Vitamins
Rising Consumer Demand for Personalization and Wellness
By 2026, the private label vitamins market is expected to be significantly driven by heightened consumer focus on personalized wellness. Shoppers are increasingly seeking tailored nutrition solutions based on lifestyle, age, gender, and health goals. Private label brands are responding by offering customizable vitamin packs, gender-specific formulations, and targeted supplements for immunity, cognitive function, and gut health. This shift reflects a broader trend toward proactive health management, with consumers viewing vitamins not just as supplements but as essential components of a holistic wellness routine.
Expansion of E-Commerce and Direct-to-Consumer Models
The digital transformation of retail continues to accelerate, with e-commerce platforms becoming primary channels for private label vitamin sales. By 2026, brands are expected to leverage subscription models, mobile apps, and AI-driven recommendations to enhance customer engagement and retention. Direct-to-consumer (DTC) strategies allow private label manufacturers to bypass traditional retail markups, offering competitive pricing while collecting valuable consumer data. This data enables more precise product development and targeted marketing, further strengthening brand loyalty.
Emphasis on Clean Label and Transparency
Consumers in 2026 are more informed and skeptical than ever, demanding full transparency in ingredient sourcing, manufacturing practices, and labeling. Private label vitamin brands are responding by emphasizing clean labels—free from artificial additives, GMOs, and common allergens. Certifications such as Non-GMO Project Verified, USDA Organic, and third-party testing seals are becoming standard. Traceability through QR codes that reveal supply chain details is emerging as a competitive advantage, reinforcing consumer trust.
Growth of Niche and Functional Formulations
The market is witnessing a surge in demand for specialized and functional formulations beyond basic multivitamins. Private label brands are introducing products targeting specific demographics—such as seniors, athletes, vegans, and pregnant women—as well as functional benefits like stress relief (adaptogens), sleep support (melatonin blends), and energy optimization (B-complex with natural stimulants). These niche offerings allow retailers to differentiate their private label lines in a crowded marketplace.
Sustainability and Eco-Conscious Packaging
Environmental sustainability is a key differentiator by 2026. Private label vitamin brands are adopting biodegradable, recyclable, or refillable packaging to meet consumer expectations for eco-friendly practices. Brands are also minimizing plastic use and partnering with sustainable suppliers. Retailers are increasingly aligning their private label strategies with corporate sustainability goals, recognizing that environmentally responsible products resonate strongly with younger, values-driven consumers.
Increased Competition and Retailer Differentiation
As the barrier to entry lowers due to advancements in contract manufacturing and formulation expertise, competition in the private label vitamin space is intensifying. To stand out, retailers are investing in premium branding, clinically backed formulations, and exclusive ingredient partnerships. Some are collaborating with nutritionists or wellness influencers to co-develop products, adding credibility and market appeal. Retailer-owned brands are no longer just cost-effective alternatives but are positioning themselves as premium, trustworthy options.
Regulatory Scrutiny and Quality Assurance
With the market expanding rapidly, regulatory bodies are expected to increase oversight of supplement claims and product safety by 2026. Private label brands will need to comply with stricter labeling requirements and substantiate health claims with scientific evidence. Investment in quality assurance, including Good Manufacturing Practices (GMP) certification and routine third-party testing, will become essential to maintain compliance and consumer confidence.
Conclusion
The 2026 private label vitamins market is poised for dynamic growth, shaped by digital innovation, personalization, sustainability, and heightened consumer awareness. Retailers and manufacturers that prioritize transparency, quality, and targeted wellness solutions will be best positioned to capture market share and build lasting consumer trust in an increasingly competitive landscape.

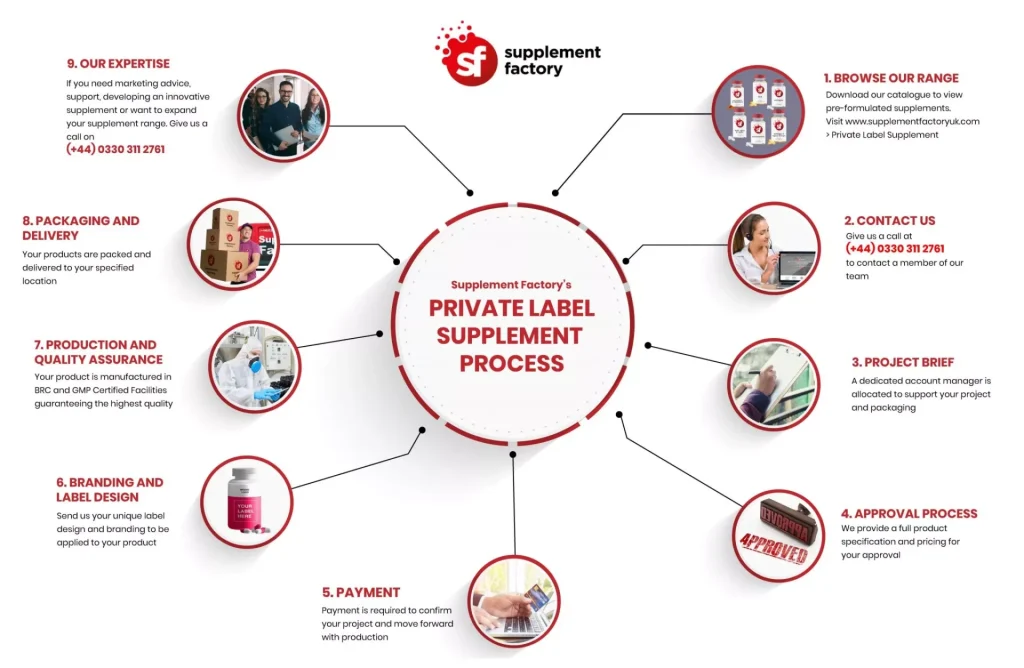
Common Pitfalls Sourcing Private Label Vitamins: Quality and Intellectual Property Concerns
Sourcing private label vitamins can be a lucrative business strategy, but it comes with significant risks if not managed carefully. Two of the most critical areas where companies stumble are ensuring product quality and protecting intellectual property (IP). Failing to address these pitfalls can lead to regulatory issues, consumer harm, reputational damage, and legal disputes.
Quality Control and Ingredient Sourcing
One of the biggest challenges in private label vitamin production is maintaining consistent product quality. Many manufacturers may source low-cost raw materials from unreliable suppliers, leading to inconsistencies in potency, purity, and safety. Contaminants such as heavy metals, pesticides, or undeclared allergens may be present if proper testing protocols are not enforced. Additionally, some suppliers may use fillers or substandard forms of active ingredients to cut costs, resulting in ineffective products that fail to meet label claims. Without rigorous third-party testing and transparent supply chain oversight, brands risk distributing products that do not comply with regulatory standards like those set by the FDA or NSF International.
Manufacturing Standards and Compliance
Another quality-related pitfall is partnering with manufacturers that do not adhere to Good Manufacturing Practices (GMPs). Facilities that are not GMP-certified may lack proper sanitation, quality control procedures, and documentation practices, increasing the risk of contamination and mislabeling. Brands must verify that their suppliers are compliant with current regulations and undergo regular audits. Assuming a manufacturer is trustworthy without independent verification can lead to recalls, legal liability, and loss of consumer trust.
Intellectual Property Ownership and Formulation Rights
A frequently overlooked issue in private label arrangements is the ownership of formulations and branding elements. Many private label suppliers offer “off-the-shelf” formulas that are sold to multiple brands. While cost-effective, this means your product is not unique and may be indistinguishable from competitors’. Worse, you may have no exclusive rights to the formula, allowing the manufacturer or other clients to sell identical products under different labels. This undermines your brand differentiation and marketing efforts.
Lack of Legal Protections and Supplier Agreements
Failing to establish clear contractual terms around IP rights can result in disputes over ownership of custom-developed formulations or proprietary blends. Without a formal agreement specifying that your brand owns the exclusive rights to a customized formula, the manufacturer may retain rights or reuse the formulation for others. Additionally, trademarks, logos, and packaging designs may not be fully protected if not properly registered or if the supplier claims partial ownership. Always ensure contracts explicitly define IP ownership and include non-compete or exclusivity clauses where applicable.
Conclusion
To avoid these pitfalls, brands must conduct due diligence on potential manufacturers, demand transparency in ingredient sourcing, require third-party testing, and secure comprehensive legal agreements that protect their intellectual property. Investing time and resources upfront in vetting partners and establishing clear terms can prevent costly issues down the line and ensure a high-quality, differentiated product in the competitive supplement market.

Logistics & Compliance Guide for Private Label Vitamins
Understanding Regulatory Requirements
Before launching a private label vitamin line, it is essential to understand the regulatory framework governing dietary supplements. In the United States, the Food and Drug Administration (FDA) regulates vitamins under the Dietary Supplement Health and Education Act (DSHEA) of 1994. This means that while supplements do not require pre-market approval, manufacturers and distributors are responsible for ensuring product safety, accurate labeling, and adherence to Good Manufacturing Practices (GMPs).
Key compliance obligations include:
– Registering your facility with the FDA.
– Complying with Current Good Manufacturing Practice (cGMP) regulations (21 CFR Part 111) for quality control.
– Ensuring accurate and compliant labeling, including Supplement Facts panel, ingredient list, and appropriate disclaimers (e.g., “This statement has not been evaluated by the FDA…”).
– Reporting any serious adverse events to the FDA within 15 business days.
Selecting a GMP-Certified Manufacturing Partner
Partnering with a reputable, GMP-certified manufacturer is critical for compliance and product quality. Look for facilities certified under NSF International, UL, or the FDA’s cGMP standards. Verify that the manufacturer conducts third-party testing for contaminants (e.g., heavy metals, microbes) and validates label claims through potency testing.
When evaluating manufacturers:
– Request documentation of certifications and testing protocols.
– Confirm they provide a Certificate of Analysis (CoA) for each batch.
– Ensure they support your brand with formulation expertise and packaging options.
A reliable manufacturer reduces compliance risk and ensures consistent product quality.
Product Labeling and Claims Compliance
Labeling is a major compliance focus. All private label vitamins must adhere to FDA labeling requirements, including:
– A Supplement Facts panel with serving size, active ingredients, and % Daily Value.
– Full disclosure of all ingredients, including excipients.
– The product name and net quantity.
– Name and place of business of the manufacturer, packer, or distributor.
– A required disclaimer: “These statements have not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure, or prevent any disease.”
Avoid structure/function claims that imply disease treatment (e.g., “cures arthritis”). Instead, use compliant language such as “supports joint health.” All claims must be truthful, not misleading, and substantiated by scientific evidence.
Supply Chain and Inventory Management
Efficient logistics begin with a transparent and reliable supply chain. Work closely with your manufacturer to establish lead times, minimum order quantities (MOQs), and reorder points. Consider:
– Safety stock levels to prevent stockouts.
– Shelf-life management—most vitamins have a 2–3 year shelf life; implement a first-expired, first-out (FEFO) inventory system.
– Climate-controlled storage to preserve ingredient integrity.
Use inventory management software to track stock levels, expiration dates, and order history, ensuring smooth operations and reducing waste.
Shipping, Storage, and Distribution
Vitamins are sensitive to heat, moisture, and light. Ensure storage and shipping conditions maintain product stability:
– Store in a cool, dry place with controlled humidity.
– Use insulated packaging if shipping in extreme temperatures.
– Partner with carriers experienced in handling health products.
For international distribution, research import regulations in target markets. The EU, Canada, Australia, and other regions have their own supplement regulations and require specific labeling, licensing, or product notifications.
Adverse Event Reporting and Product Recalls
As the brand owner, you are legally responsible for monitoring and reporting serious adverse events related to your products. Establish a system to receive, document, and evaluate consumer complaints. If a product issue arises, you must:
– Report serious adverse events to the FDA within 15 business days.
– Be prepared to initiate a recall if a product is found to be adulterated or misbranded.
– Maintain records of all complaints and actions taken for at least 3 years.
Having a recall plan in place minimizes risk and protects your brand reputation.
Third-Party Testing and Quality Assurance
While not required by law, third-party testing enhances credibility and compliance. Consider using independent labs to:
– Verify label accuracy and ingredient potency.
– Test for contaminants such as lead, arsenic, and microbes.
– Certify products with seals like USP, NSF, or ConsumerLab.
These certifications can boost consumer trust and differentiate your brand in a competitive market.
Final Steps: Launching with Confidence
Before launching, conduct a final compliance review:
– Audit labels for FDA compliance.
– Confirm facility registration and GMP adherence.
– Ensure all documentation (CoAs, test results, adverse event procedures) is in order.
By prioritizing logistics and compliance from the start, private label vitamin brands can build trust, reduce legal risk, and deliver safe, high-quality products to consumers.
Conclusion: Sourcing Private Label Vitamins
Sourcing private label vitamins presents a strategic opportunity for entrepreneurs, retailers, and health-focused brands to enter or expand within the growing wellness market. By partnering with a reputable manufacturer, businesses can offer high-quality, customized supplements under their own brand name—without the need for extensive R&D, production infrastructure, or regulatory expertise.
Key benefits include rapid time-to-market, cost-effective product development, and the ability to maintain brand consistency across a range of formulations. However, success depends on selecting a reliable manufacturer that adheres to strict quality standards (such as cGMP and third-party testing), offers transparency in sourcing ingredients, and provides flexible customization options.
Ultimately, with careful due diligence, clear branding, and a focus on product quality and consumer trust, private labeling vitamins can be a profitable and sustainable venture in the expanding health and wellness industry.