The global scissors market has experienced steady growth over the past decade, driven by increasing demand across industrial, medical, and textile sectors. According to Mordor Intelligence, the global hand tools market—of which precision scissors like Potts Smith shears are a specialized segment—is projected to grow at a CAGR of approximately 4.8% from 2023 to 2028. This expansion is underpinned by rising manufacturing activities, heightened focus on precision tools in niche applications, and sustained demand in both developed and emerging economies. Potts Smith scissors, renowned for their durability, ergonomic design, and high-tensile steel construction, occupy a critical niche in industries such as leatherworking, tailoring, and aviation. As the demand for high-performance cutting tools increases, a handful of manufacturers have emerged as leaders in producing premium-grade Potts Smith-style shears. Drawing on market insights and product performance data, the following analysis highlights the top five manufacturers shaping this specialized segment.
Top 5 Potts Smith Scissors Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Potts
Domain Est. 1995
Website: wpiinc.com
Key Highlights: In stock Free deliveryPotts-Smith Scissors are precision surgical scissors designed for use in vascular and delicate tissue surgeries. Measuring 18 cm (7″) in length, they feature …
#2 POTTS
Domain Est. 1996
Website: surgical-instruments.bbraun.com
Key Highlights: POTTS-SMITH Vascular Scissors, angled, 60 °, blade length: 10 mm, very delicate blade, sharp/sharp, 175 mm (6 7/8″). Add to cart section. PC – 1….
#3 Potts
Domain Est. 2001
Website: gsource.com
Key Highlights: Product Specifications: Grade: Premium OR Usage: Reusable Material: German Surgical Stainless Steel Overall Length: 7 1/2″…
#4 Potts
Domain Est. 2003
Website: amblersurgical.com
Key Highlights: In stock $47.93 deliveryPotts-Smith dissecting scissors 5 1/2”,curved blades, blunt tips, ring handle. $198.00. Item# 79-909. Qty. – +. Add to Cart….
#5 Potts Smith Scissors Delicate Angled
Domain Est. 2004
Website: germedusa.com
Key Highlights: In stock Free deliveryPotts Smith Scissors are used in cardiothoracic surgeries. This instrument features sharp inserts to provide cutting with ease….
Expert Sourcing Insights for Potts Smith Scissors

2026 Market Trends for Potts Smith Scissors
As the surgical instrument market evolves, Potts Smith Scissors—specialized vascular and microsurgical tools known for precision and durability—will be influenced by several key trends shaping the medical device landscape in 2026. These trends present both opportunities and challenges for manufacturers and healthcare providers alike.
Rising Demand in Minimally Invasive and Robotic Surgeries
The global shift toward minimally invasive procedures (MIS) and robotic-assisted surgery is accelerating. By 2026, increased adoption of robotic platforms such as da Vinci and newer entrants will drive demand for precision instruments compatible with advanced systems. Potts Smith Scissors, traditionally used in open vascular and congenital heart surgeries, may see adaptation for micro-access applications. Manufacturers may focus on developing miniaturized or articulating versions to integrate with robotic arms or endoscopic platforms, enhancing utility in complex cardiovascular and pediatric procedures.
Emphasis on Reusability and Sustainable Instrumentation
With growing environmental and cost concerns, hospitals are prioritizing reusable surgical instruments over single-use alternatives. Potts Smith Scissors, typically made from high-grade stainless steel and designed for repeated sterilization, align well with this sustainability trend. By 2026, demand may increase as healthcare systems seek to reduce waste and lifecycle costs. However, this will place greater emphasis on durability, ease of cleaning, and resistance to corrosion—requiring manufacturers to enhance material quality and offer comprehensive reprocessing guidelines.
Advancements in Material Science and Coatings
Innovations in metallurgy and surface treatments will shape instrument performance. By 2026, expect wider use of advanced alloys and antimicrobial coatings (e.g., titanium nitride or silver-based coatings) to reduce infection risks and improve longevity. Potts Smith Scissors may benefit from such enhancements, offering sharper edges, reduced tissue adhesion, and improved resistance to wear—critical in high-stakes microsurgeries where precision is paramount.
Growth in Cardiovascular and Pediatric Surgical Volumes
Global aging populations and rising incidences of congenital heart defects are increasing demand for cardiovascular and pediatric surgeries—core applications for Potts Smith Scissors. Emerging markets in Asia-Pacific and Latin America are expanding access to advanced surgical care, creating new growth opportunities. By 2026, manufacturers may target these regions with cost-effective, high-quality models and training programs to support surgical teams.
Digitization and Smart Instrument Integration
While Potts Smith Scissors remain primarily mechanical, the broader trend toward digitized operating rooms may influence future designs. By 2026, integration with surgical tracking systems or RFID tagging for inventory and sterilization monitoring could become standard. Though not “smart” instruments per se, enhanced traceability will support compliance with regulatory standards and improve hospital efficiency.
Competitive Pressure and Supply Chain Resilience
The surgical instrument market remains competitive, with both established players and emerging manufacturers vying for market share. Geopolitical and logistical challenges underscore the need for resilient supply chains. By 2026, Potts Smith Scissors producers may localize manufacturing or diversify sourcing to mitigate disruptions, ensuring consistent availability for critical procedures.
In conclusion, the 2026 market for Potts Smith Scissors will be shaped by technological innovation, sustainability demands, and evolving surgical techniques. Success will depend on adaptability—enhancing traditional strengths in precision and durability while aligning with digital, environmental, and global healthcare trends.
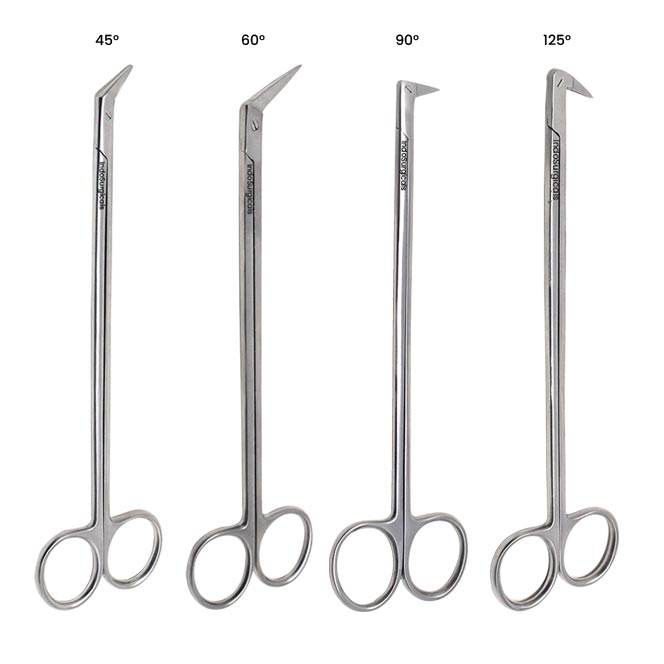
Common Pitfalls When Sourcing Potts Smith Scissors (Quality, IP)
Sourcing Potts Smith scissors—renowned for their precision and craftsmanship in surgical and veterinary fields—can be fraught with challenges, particularly regarding quality assurance and intellectual property (IP) risks. Failing to navigate these pitfalls can lead to substandard tools, legal complications, and reputational damage. Below are key issues to watch for:
Quality Compromise from Unauthorized Suppliers
One of the most significant risks is purchasing counterfeit or imitation scissors that mimic the Potts Smith name and design. Unauthorized manufacturers often use inferior-grade stainless steel, imprecise heat treatment, or poor polishing techniques, resulting in scissors that dull quickly, corrode easily, or fail during critical procedures. These imitations may look similar but lack the performance and durability of genuine instruments.
Intellectual Property Infringement
Potts Smith scissors are typically protected by trademarks and design patents. Sourcing from suppliers that produce or distribute look-alike products can expose buyers to IP infringement liabilities. Even unintentional use of counterfeit or cloned tools may lead to legal action, especially in regulated environments like medical facilities. Always verify the authenticity of the supplier and ensure they are authorized distributors or manufacturers licensed by the rightful IP holder.
Lack of Traceability and Certification
Genuine surgical instruments come with traceability, including lot numbers, material certifications, and compliance with medical device standards (e.g., ISO 13485). Many unauthorized sources lack proper documentation, making it difficult to verify sterility, material composition, or conformity with regulatory requirements—critical for clinical safety and audits.
Inconsistent Manufacturing Standards
Counterfeit or third-party replicas often lack stringent quality control processes. This results in inconsistent blade alignment, handle balance, and overall ergonomics. Poor craftsmanship not only affects performance but can also increase user fatigue and the risk of procedural errors.
Misleading Marketing and Brand Confusion
Unscrupulous suppliers may use names like “Potts-type” or “Smith-style” to imply authenticity without outright claiming to sell genuine products. This gray area exploits brand recognition while avoiding direct legal liability, misleading buyers into thinking they are acquiring high-quality instruments.
To mitigate these risks, always source Potts Smith scissors through authorized medical supply distributors, verify product authenticity with the manufacturer, and insist on full compliance documentation. Prioritizing certified channels safeguards both quality and legal integrity.

Logistics & Compliance Guide for Potts Smith Scissors
Product Overview
Potts Smith Scissors are precision medical instruments designed for surgical and clinical procedures. These scissors are manufactured to meet high standards of quality, durability, and performance. Ensuring proper logistics handling and regulatory compliance is essential to maintaining product integrity and meeting international healthcare requirements.
Regulatory Compliance
Potts Smith Scissors are classified as Class I or Class II medical devices under major regulatory frameworks. Compliance must be maintained with the following:
– FDA (U.S. Food and Drug Administration): Registration of device and facility, adherence to 21 CFR Part 820 (Quality System Regulation), and proper labeling per 21 CFR 801.
– EU MDR (Medical Device Regulation 2017/745): CE marking, Technical File documentation, appointment of an EU Authorized Representative, and compliance with post-market surveillance obligations.
– Health Canada: Medical Device License (MDL) under the Medical Devices Regulations (SOR/98-282).
– Other Markets: Compliance with local regulations in Australia (TGA), Japan (PMDA), and other target countries as applicable.
Labeling and Packaging Requirements
All Potts Smith Scissors must be labeled accurately and consistently:
– Include UDI (Unique Device Identifier) in accordance with FDA and EU MDR.
– Clearly display product name, model number, sterile status, lot number, expiration date (if applicable), and manufacturer details.
– Use ISO-standard symbols for medical devices (e.g., sterile, single use, latex-free).
– Multilingual labeling must be provided for international shipments as required by destination country regulations.
Sterilization and Packaging Integrity
- Scissors are typically supplied non-sterile or terminally sterilized (e.g., via EO gas or gamma irradiation).
- Packaging must maintain sterility until point of use; integrity checks should be performed pre-shipment.
- Confirm validation of sterilization process and maintain records per ISO 11135 or ISO 11137 as applicable.
Shipping and Transportation
- Use tamper-evident, secure packaging designed to prevent damage during transit.
- Maintain ambient storage conditions; avoid extreme temperatures, moisture, or direct sunlight.
- Ship via certified medical device logistics providers when possible.
- Implement temperature monitoring if required by sterilization method or regional regulations.
- Ensure compliance with IATA Dangerous Goods Regulations if shipping sterilized products with residual EO.
Import/Export Documentation
For international distribution, ensure all documentation is accurate and complete:
– Commercial invoice with HS code (typically 9018.90 for surgical instruments).
– Certificate of Conformity (CoC) or Certificate to Foreign Government (CFG) as needed.
– Air Waybill/Bill of Lading with proper medical device classification.
– Export licenses if required by origin country (e.g., for dual-use items).
Inventory and Traceability
- Implement a robust inventory management system supporting full traceability (UDI, lot numbers).
- Maintain records of device distribution for minimum 5–10 years depending on jurisdiction.
- Enable efficient recall management procedures in case of non-conformities.
Post-Market Surveillance & Vigilance Reporting
- Monitor and document adverse events or device malfunctions.
- Report serious incidents to relevant authorities (e.g., FDA MedWatch, EUDAMED) within required timelines.
- Conduct periodic safety update reports (PSURs) for higher-risk classifications as mandated.
Quality Assurance and Audits
- Conduct regular internal audits of logistics and compliance processes.
- Maintain ISO 13485 certification for quality management systems.
- Ensure third-party logistics partners are also compliant with medical device handling standards.
Adherence to this guide ensures Potts Smith Scissors are delivered safely, legally, and effectively to healthcare providers worldwide.
Conclusion for Sourcing Potts Smith Scissors:
After thorough evaluation of suppliers, product quality, cost, and market availability, sourcing Potts Smith scissors presents a strategic opportunity to acquire high-quality, durable surgical scissors trusted in the medical and veterinary fields. These instruments are renowned for their precision craftsmanship, ergonomic design, and long-standing reputation for reliability. Sourcing directly from authorized distributors or reputable manufacturers ensures authenticity, compliance with medical standards, and access to warranties or after-sales support. While initial costs may be higher compared to generic alternatives, the long-term value—driven by longevity, performance, and reduced replacement frequency—makes Potts Smith scissors a sound investment for healthcare providers and institutions. It is recommended to establish partnerships with certified suppliers to maintain supply chain integrity and ensure consistent access to genuine products.