The global demand for portable Automated External Defibrillators (AEDs) has surged in recent years, driven by rising awareness of sudden cardiac arrest (SCA) and increased adoption of emergency medical devices in public access defibrillation programs. According to Grand View Research, the global AED market was valued at USD 1.48 billion in 2023 and is expected to expand at a compound annual growth rate (CAGR) of 9.3% from 2024 to 2030. Similarly, Mordor Intelligence projects a CAGR of over 9% during the forecast period of 2024–2029, citing expanding healthcare infrastructure, regulatory support, and growing integration of AEDs in schools, airports, and commercial facilities. This accelerating market trajectory, combined with advancements in device portability, ease of use, and real-time connectivity, has intensified competition among manufacturers. As accessibility and response time become critical factors in SCA survival, innovation in compact, user-friendly AEDs has positioned leading companies at the forefront of emergency cardiac care. The following analysis highlights the top 10 manufacturers shaping this dynamic landscape through technological leadership, global reach, and robust clinical performance.
Top 10 Portable Automated External Defibrillator Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 AED Professionals
Domain Est. 2005
Website: aedprofessionals.com
Key Highlights: Free delivery over $99 30-day returnsShop AED devices and accessories, EMS supplies, first aid kits and other medical supplies from AED Professionals.Missing: portable manufacture…
#2 Avive AED
Website: avive.life
Key Highlights: At just 2.1 lbs, the Avive Connect AED is the lightest and smallest AED on the market. Easy to transport and take with you on-the-go….
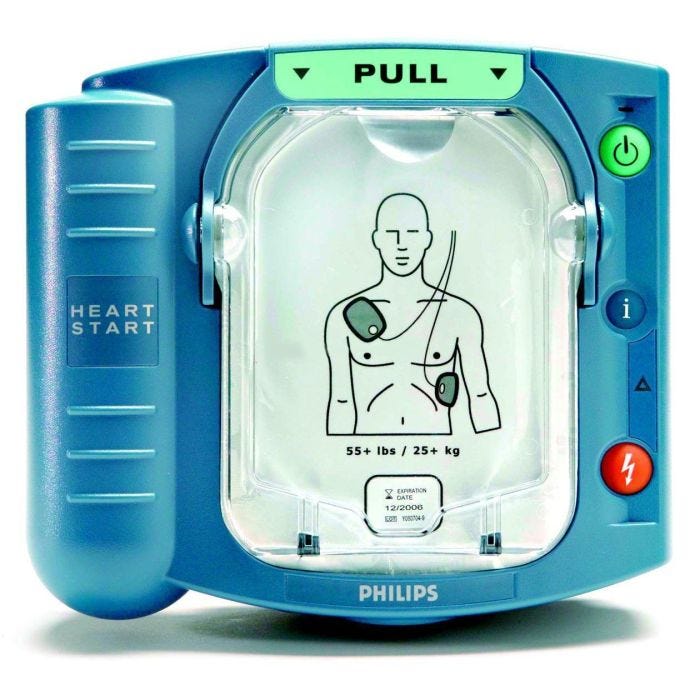
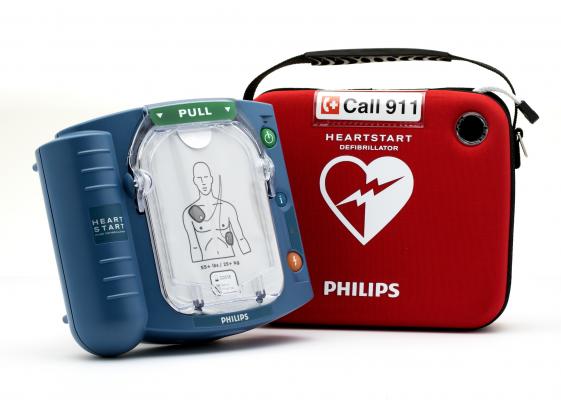
#3 Philips
Domain Est. 1987
Website: aedshop.philips.com
Key Highlights: Free delivery · 30-day returnsPhilips HeartStart automated external defibrillators (AEDs) are designed for every use type, whether inside or outside, in varying environments or at…
#4 Automated External Defibrillators (AEDs)
Domain Est. 1995
Website: zoll.com
Key Highlights: ZOLL provides intuitive and intelligent AEDs for sudden cardiac arrest emergencies. Learn about innovative features like real-time CPR feedback….
#5 Shop AEDs and Accessories
Domain Est. 2000
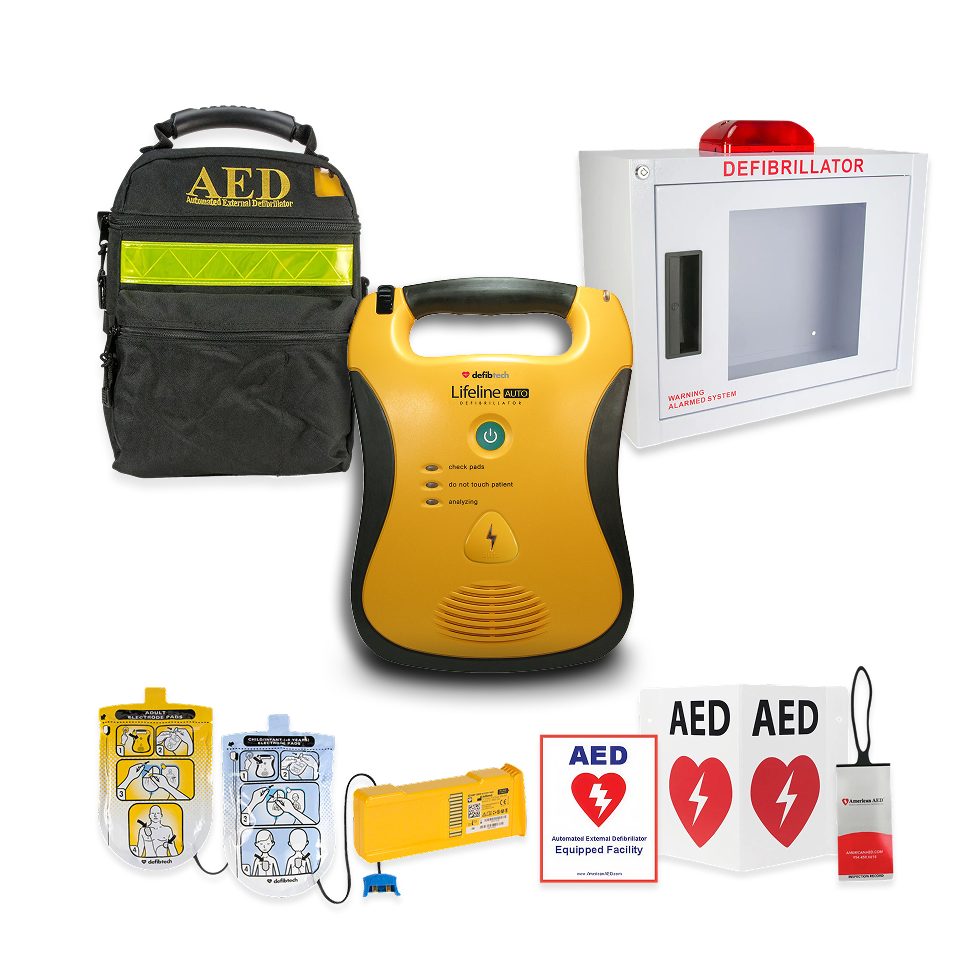
#6 Lifeline AED
Domain Est. 2000
Website: defibtech.com
Key Highlights: The original Lifeline AED includes all mission critical features necessary to provide the most advanced treatment for Sudden Cardiac Arrest emergencies….
#7 American AED
Domain Est. 2003
Website: americanaed.com
Key Highlights: Free delivery · 30-day returnsAmerican AED – The most trusted online seller of the latest AED machines, Automated External Defibrillators. Shop for your right AED Package….
#8 AED Brands
Domain Est. 2008
#9 AED USA: AEDs & Accessories for Life
Domain Est. 2015
Website: aedusa.com
Key Highlights: Free delivery over $99 30-day returnsWe supply new, top quality Automated External Defibrillators (AEDs) and accessories at affordable prices to organizations and individuals throu…
#10 Cardiangel
Domain Est. 2020
Website: cardiangel.com
Key Highlights: Cardiangel automated external defibrillator is a medical device designed for quick and simple use during sudden cardiac arrests….
Expert Sourcing Insights for Portable Automated External Defibrillator

H2: 2026 Market Trends for Portable Automated External Defibrillators (AEDs)
The global market for Portable Automated External Defibrillators (AEDs) is poised for significant evolution by 2026, driven by technological innovation, increased public awareness, supportive regulatory environments, and growing healthcare accessibility. The following key trends are expected to shape the landscape of the portable AED market in 2026:
-
Increased Integration of Smart Technology and IoT Connectivity
By 2026, portable AEDs will increasingly feature integrated Internet of Things (IoT) capabilities, enabling real-time monitoring, remote diagnostics, and GPS tracking. These smart AEDs will communicate with emergency medical services (EMS) and public response networks, reducing response times. Cloud-based platforms will allow organizations to manage device readiness, battery status, and electrode expiration, enhancing maintenance and reliability. -
Expansion in Public Access Defibrillation (PAD) Programs
Governments and health organizations are expected to expand PAD initiatives, placing AEDs in public spaces such as airports, schools, sports venues, and workplaces. Regulatory mandates in regions like North America and Europe will continue to support widespread AED deployment, fueled by evidence linking early defibrillation to improved survival rates from sudden cardiac arrest (SCA). -
Miniaturization and Enhanced Portability
Advancements in battery technology and component design will lead to even more compact, lightweight, and durable AEDs. These improvements will increase adoption in non-traditional settings such as homes, fitness centers, and personal use, making life-saving devices more accessible to individuals at high risk of SCA. -
AI-Powered Voice and Visual Guidance
Artificial intelligence will enhance user interfaces with adaptive voice prompts and visual instructions tailored to the rescuer’s actions. In 2026, AEDs are expected to leverage AI to assess user competence in real time, adjusting guidance complexity to improve layperson effectiveness during emergencies. -
Growth in Emerging Markets
Asia-Pacific, Latin America, and parts of Africa will see accelerated AED adoption due to rising healthcare infrastructure investment, urbanization, and growing awareness of cardiovascular diseases. Local manufacturing and partnerships with global AED providers will reduce costs and improve accessibility. -
Focus on Training and Public Awareness
Digital training platforms, including virtual reality (VR) and mobile apps, will become standard companion tools to AEDs. In 2026, integration of CPR and AED training into school curricula and corporate wellness programs will further empower bystander intervention. -
Regulatory and Reimbursement Support
Regulatory bodies such as the FDA and EMA are expected to streamline approval processes for innovative AED models. Additionally, expanded insurance coverage and public funding will lower financial barriers for institutions and individuals purchasing AEDs. -
Sustainability and Eco-Friendly Design
As environmental concerns grow, manufacturers will prioritize recyclable materials, longer-life components, and energy-efficient designs. Reusable electrode pads and modular AED systems will reduce electronic waste and operational costs.
In conclusion, the 2026 portable AED market will be defined by smarter, more accessible, and user-centric devices, supported by robust public health strategies and technological innovation. These trends collectively aim to improve survival outcomes from sudden cardiac arrest and mark a transformative phase in emergency cardiovascular care.

Common Pitfalls When Sourcing Portable Automated External Defibrillators (Quality, IP)
Sourcing Portable Automated External Defibrillators (AEDs) requires careful attention to both product quality and intellectual property (IP) considerations. Overlooking these aspects can lead to significant risks, including device failure during emergencies, legal liabilities, and reputational damage. Below are key pitfalls to avoid:
Poor Device Quality and Reliability
One of the most critical pitfalls is selecting an AED based solely on cost, without verifying its reliability and performance. Low-quality devices may fail during life-threatening situations due to substandard components, inadequate battery life, or poor durability. It is essential to source AEDs that meet international standards such as the FDA (U.S.), CE marking (Europe), or other regional regulatory approvals. Devices should also undergo rigorous environmental testing (e.g., temperature, humidity, shock resistance) to ensure functionality in real-world conditions.
Lack of Regulatory Compliance
Sourcing AEDs that are not certified by recognized regulatory bodies is a major risk. Non-compliant devices may not have undergone proper safety and performance testing, potentially rendering them illegal to use or distribute. Always verify that the manufacturer holds valid certifications and that each unit is traceable to an approved model. Purchasing non-compliant AEDs may also invalidate insurance coverage or liability protections.
Inadequate Software and Firmware Security
Modern AEDs often include software for self-diagnostics, data recording, and firmware updates. Sourcing devices with poorly secured or outdated software can expose organizations to cybersecurity risks. Additionally, unauthorized firmware modifications may compromise device functionality. Ensure that the manufacturer provides regular, secure updates and maintains control over software integrity.
Intellectual Property Infringement
Procuring AEDs from manufacturers or suppliers that infringe on existing patents or trademarks can lead to legal disputes, product seizures, or financial penalties. Counterfeit or clone devices often mimic reputable brands but lack proper IP licensing. Conduct due diligence on the supplier’s IP standing—request documentation showing licensed technology and verify patents associated with core features such as electrode design, algorithms, or user interface.
Insufficient Training and Support Materials
AEDs are only effective when used correctly. Sourcing devices without comprehensive, multilingual training materials, clear visual instructions, or accessible technical support diminishes their utility. Poorly designed user interfaces or lack of ongoing training programs can delay response times and increase user error. Ensure the supplier provides robust educational resources and post-purchase support.
Hidden Costs and Maintenance Requirements
Low upfront pricing can be misleading if ongoing costs for electrode replacements, battery swaps, or software licenses are excessive or poorly disclosed. Some AEDs require proprietary consumables that are expensive or difficult to source. Evaluate the total cost of ownership and confirm long-term availability of parts and service before committing.
Supply Chain and Warranty Risks
Sourcing from unknown or unstable suppliers increases the risk of interrupted support, voided warranties, or lack of spare parts. Ensure the supplier has a proven track record, offers a comprehensive warranty, and maintains an accessible service network. Devices without reliable manufacturer backing may become obsolete or unsupported over time.
By recognizing and addressing these quality and IP-related pitfalls, organizations can ensure they deploy safe, effective, and legally compliant AEDs, ultimately enhancing emergency preparedness and public safety.

Logistics & Compliance Guide for Portable Automated External Defibrillator (AED)
Portable Automated External Defibrillators (AEDs) are life-saving medical devices that must be properly managed to ensure readiness in emergencies. This guide outlines key logistics and compliance considerations for the deployment, maintenance, and regulatory adherence of AEDs in workplaces, public spaces, and organizations.
Regulatory Compliance
AED programs must comply with local, state, and federal regulations to ensure legal protection and operational effectiveness. Key compliance areas include:
- FDA Clearance: Ensure all AEDs are cleared by the U.S. Food and Drug Administration (or equivalent regulatory body in your country) for public use.
- Good Samaritan Laws: Familiarize yourself with local Good Samaritan laws, which protect individuals who use AEDs in good faith during emergencies. Compliance often requires visible signage, proper training, and device maintenance.
- OSHA Guidelines: While OSHA does not specifically mandate AEDs, it encourages their availability under the General Duty Clause. Providing AEDs and training can reduce workplace injury and demonstrate due diligence.
- State and Local Regulations: Some jurisdictions require AEDs in specific facilities (e.g., schools, fitness centers, government buildings). Verify requirements with local health or emergency medical services (EMS) authorities.
- Registration with Local EMS: Many regions require AEDs to be registered with the local emergency medical services system to facilitate rapid response coordination.
Device Acquisition and Deployment
Strategic planning is essential when acquiring and placing AEDs:
- Risk Assessment: Conduct a site evaluation to identify high-risk areas (e.g., gyms, airports, schools) and determine the number and placement of AEDs.
- Accessibility: Install AEDs in visible, unlocked, and easily accessible locations. Follow the “3-5 minute” rule—devices should be reachable within 3 to 5 minutes of collapse.
- Environmental Protection: Use weatherproof cabinets for outdoor units and ensure indoor units are protected from extreme temperatures and humidity.
- Signage: Install standardized AED signage per American National Standards Institute (ANSI) guidelines to aid quick identification.
Maintenance and Inspection
Regular maintenance ensures AEDs are operational when needed:
- Scheduled Inspections: Perform visual and operational checks monthly. Verify battery status, pad expiration dates, and device self-test indicators.
- Replace Consumables: Electrode pads and batteries have expiration dates. Replace them proactively, typically every 2–5 years depending on manufacturer specifications.
- Service Records: Maintain a log of all inspections, maintenance, and repairs. Retain records for at least 3–5 years for audit and compliance purposes.
- Software Updates: Periodically check for firmware or software updates from the manufacturer to ensure optimal performance.
Training and Readiness
An effective AED program includes trained personnel and clear emergency procedures:
- CPR and AED Training: Train employees and designated responders in CPR and AED use through accredited programs (e.g., American Heart Association, Red Cross). Retrain every two years.
- Drills and Simulations: Conduct regular emergency response drills to reinforce readiness and coordination with local EMS.
- Public Awareness: Post instructions near AEDs and provide basic public education on how and when to use the device.
Documentation and Liability Management
Proper documentation supports compliance and reduces legal risk:
- AED Program Policy: Develop a written policy outlining responsibilities, maintenance schedules, training protocols, and emergency response procedures.
- Incident Reporting: Document any AED use, including the outcome and device performance. Report incidents to medical directors or regulatory bodies as required.
- Liability Protection: Ensure your organization has appropriate liability insurance. Compliance with training, maintenance, and registration requirements strengthens legal protection.
Vendor and Support Management
Partnering with reliable suppliers ensures long-term success:
- Certified Suppliers: Purchase AEDs and supplies from authorized distributors to ensure authenticity and regulatory compliance.
- Technical Support: Confirm access to 24/7 technical assistance and warranty services from the manufacturer or vendor.
- Replacement Planning: Track device lifespan (typically 8–10 years) and budget for timely replacement.
By following this logistics and compliance guide, organizations can maintain effective AED programs that enhance public safety, meet regulatory standards, and support a culture of preparedness.
Conclusion:
Sourcing portable automated external defibrillators (AEDs) is a critical step in enhancing emergency response capabilities and promoting public safety. AEDs play a vital role in improving survival rates during sudden cardiac arrest, especially when administered within the first few minutes of an incident. When sourcing these life-saving devices, organizations must consider key factors such as device reliability, ease of use, maintenance requirements, training support, warranty, and compliance with regulatory standards.
Additionally, evaluating cost-effectiveness and long-term service agreements ensures sustainability and readiness. Strategic placement of AEDs in workplaces, schools, public venues, and community spaces, combined with proper staff training, maximizes their effectiveness. Ultimately, investing in high-quality, accessible AEDs reflects a proactive commitment to health and safety, potentially saving lives and fostering a culture of preparedness.