The global ophthalmic equipment market is experiencing robust growth, driven by an increasing prevalence of cataracts, technological advancements in surgical devices, and rising demand for minimally invasive procedures. According to Mordor Intelligence, the global phacoemulsification devices market was valued at approximately USD 1.32 billion in 2022 and is projected to grow at a CAGR of 7.4% from 2023 to 2028. Similarly, Grand View Research estimates that the market size surpassed USD 1.4 billion in 2023 and is anticipated to expand further due to continuous innovation in cataract surgery technology and growing geriatric populations worldwide. In this evolving landscape, leading manufacturers are enhancing device precision, integration with intraoperative imaging, and fluidics control to improve surgical outcomes. As demand rises, identifying the top players shaping the phacoemulsification space becomes critical for healthcare providers and procurement decision-makers alike.
Top 8 Phacoemulsification Machine Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
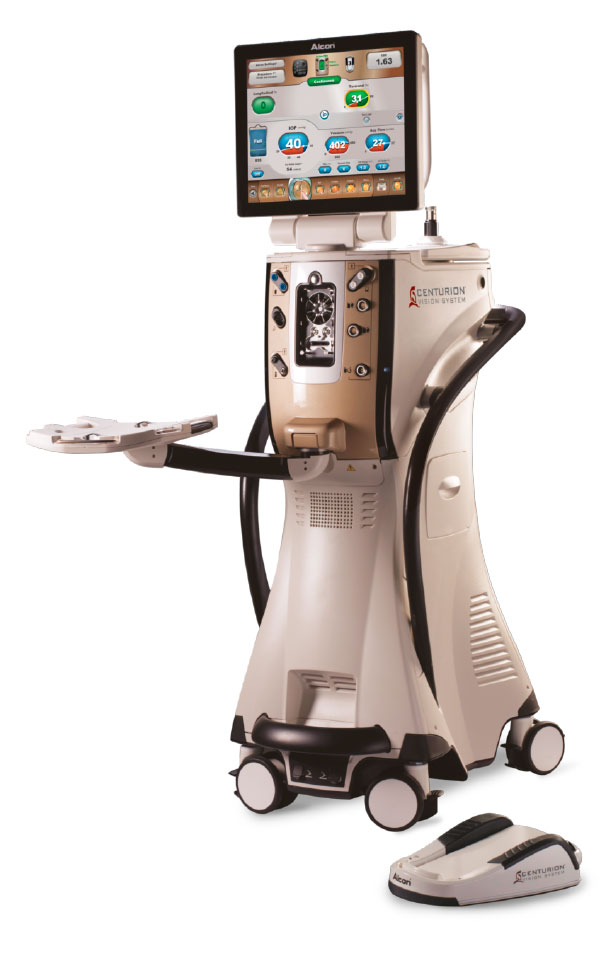
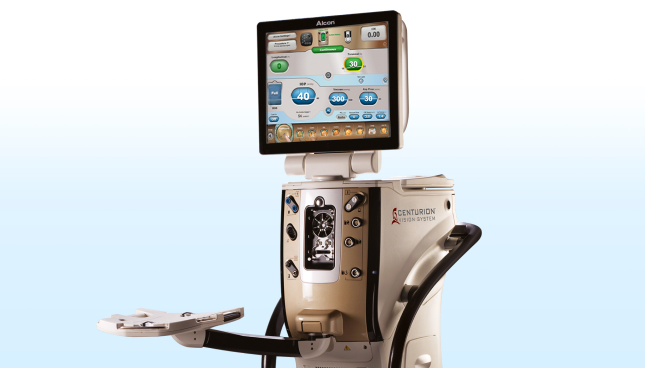
#1 CENTURION-based Phaco Performance
Domain Est. 2001
Website: myalcon.com
Key Highlights: Alcon’s CENTURION-based phaco systems provide you and your practice with the leading technology in phacoemulsification….
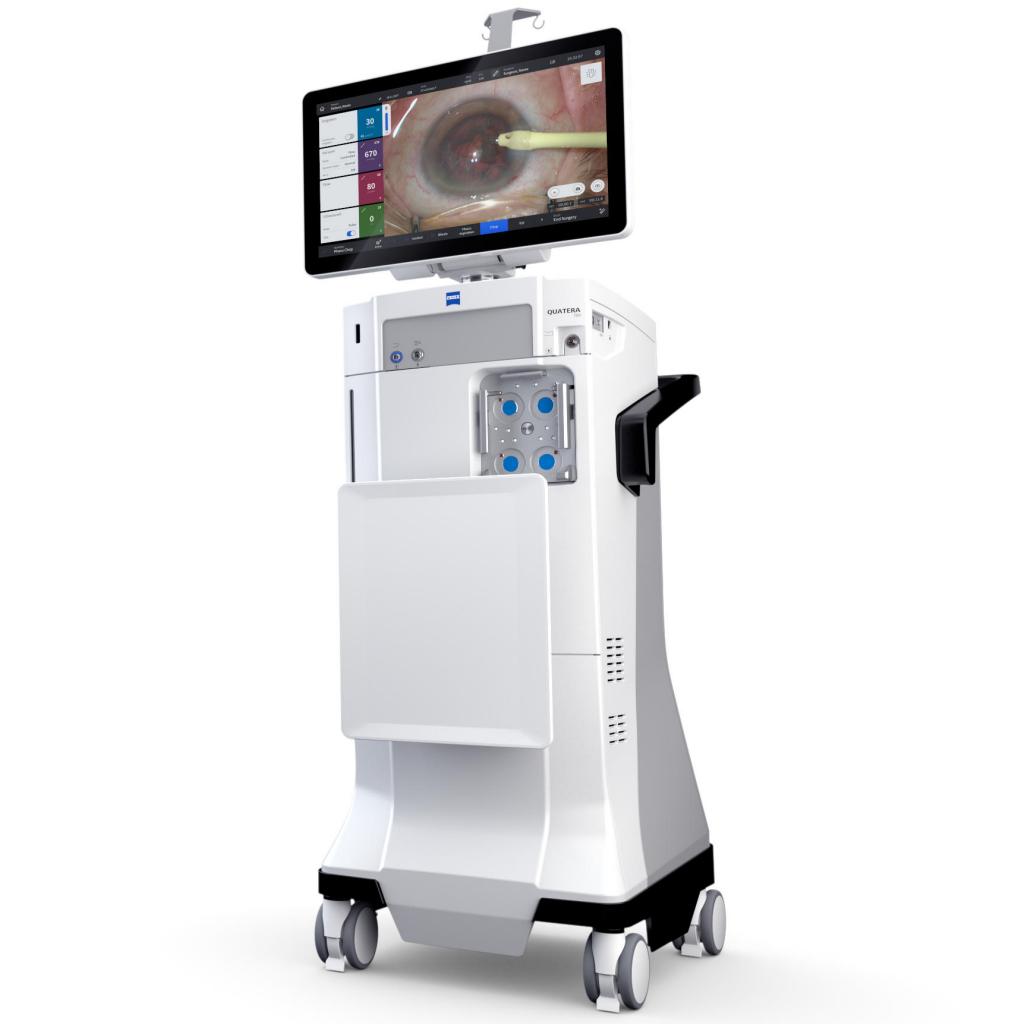
#2 ZEISS Phacoemulsification, Lens Fragmentation and Surgical Packs
Domain Est. 1995
Website: zeiss.com
Key Highlights: A microinterventional device designed to deliver zero-energy 1 endocapsular lens fragmentation. All to optimize your surgical workflow….
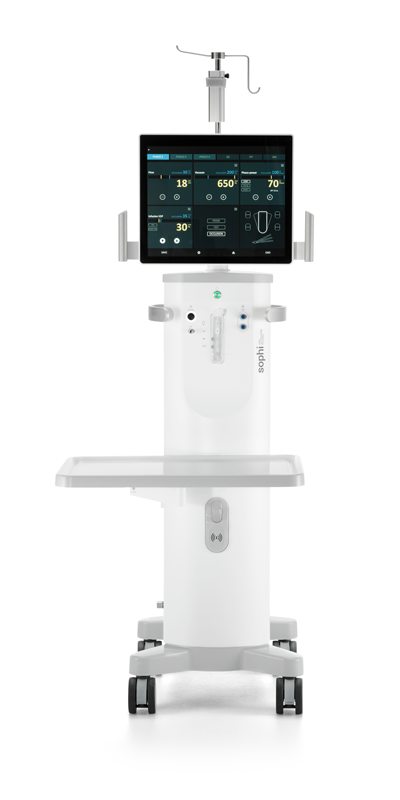
#3 Sophi Phacoemulsification System
Domain Est. 1998
Website: rayner.com
Key Highlights: Sophi is the first wireless Phaco system to offer high flexibility and mobility, providing ease of handling in the operating theatre and increased usibility….
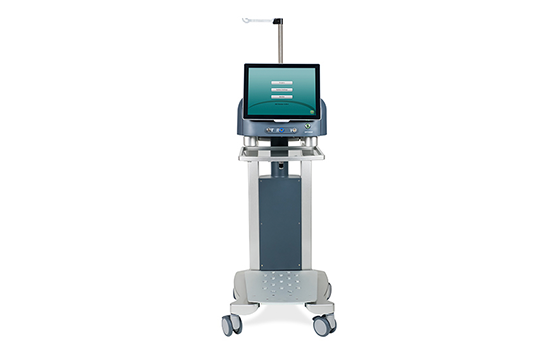
#4 BVI Medical
Domain Est. 2010
Website: bvimedical.com
Key Highlights: BVI® is a global medical company manufacturing and distributing surgical devices dedicated to ophthalmology in more than 90 countries….
#5 Phacoemulsification surgery equipment: Handpieces
Domain Est. 2017
Website: jnjvisionpro.com
Key Highlights: Our phaco handpieces deliver true ultrasound efficiency to optimize lens extraction with lower corneal edema and endothelial cell loss….
#6 CataRhex 3
Domain Est. 1998
Website: oertli-instruments.com
Key Highlights: The CataRhex 3 phaco machine is for cataract and glaucoma surgery. Discover the compactness of the phaco device now….
#7 Alcon
Domain Est. 1998
Website: alcon.com
Key Highlights: Our mission is to provide innovative vision products that enhance quality of life by helping people see better. From vision research to eye health, ……
#8 LENSAR Robotic Cataract Laser System for Refractive Cataract Eye …
Domain Est. 2008
Website: lensar.com
Key Highlights: ALLY delivers a fragmentation pattern based on cataract density, designed to reduce phaco energy and inflammation for faster visual recovery….
Expert Sourcing Insights for Phacoemulsification Machine
H2: Emerging Market Trends in Phacoemulsification Machines for 2026
As the global ophthalmic devices market evolves, the phacoemulsification machine segment is poised for significant transformation by 2026. Driven by technological innovation, demographic shifts, and increasing demand for minimally invasive cataract surgery, several key trends are shaping the market landscape.
-
Advancements in Technology and Automation
By 2026, next-generation phacoemulsification systems are expected to feature enhanced automation, real-time intraoperative feedback, and artificial intelligence (AI)-assisted surgical guidance. These innovations will improve surgical precision, reduce complications, and shorten learning curves for surgeons. Integration with digital platforms and surgical data analytics is also expected to support personalized treatment planning and outcomes tracking. -
Shift Toward Portable and Cost-Effective Systems
Growing demand in emerging markets and ambulatory surgical centers (ASCs) is driving the development of compact, portable, and affordable phacoemulsification machines. These systems cater to outpatient clinics and regions with limited access to high-end surgical infrastructure, expanding market reach and improving accessibility to cataract treatment in low- and middle-income countries. -
Rising Global Cataract Prevalence
With an aging global population—especially in Asia-Pacific and Latin America—the incidence of age-related cataracts is increasing rapidly. This demographic trend is a primary driver of demand for advanced phacoemulsification equipment. The World Health Organization estimates that cataracts remain the leading cause of blindness worldwide, fueling investments in surgical solutions. -
Expansion of Refractive Cataract Surgery
Patients are increasingly seeking premium outcomes, including reduced dependency on glasses post-surgery. This trend is accelerating the adoption of phacoemulsification platforms compatible with advanced intraocular lenses (IOLs), such as toric, multifocal, and extended-depth-of-focus (EDOF) lenses. Integrated biometry and surgical planning tools are becoming standard in high-end machines. -
Consolidation and Strategic Partnerships Among Key Players
The competitive landscape is witnessing mergers, acquisitions, and strategic collaborations among major manufacturers such as Alcon, Johnson & Johnson Vision, Bausch + Lomb, and Zeiss. These partnerships aim to expand product portfolios, enhance R&D capabilities, and strengthen distribution networks in high-growth regions. -
Regulatory and Reimbursement Dynamics
Regulatory approvals and favorable reimbursement policies—particularly in North America and Europe—will continue to influence market adoption. However, in developing economies, inconsistent reimbursement and regulatory hurdles may slow penetration, prompting manufacturers to adopt flexible pricing and service models. -
Sustainability and Service-Centric Business Models
By 2026, sustainability considerations are influencing product design, with manufacturers focusing on energy-efficient systems and reusable components. Additionally, service-based models—including equipment leasing, maintenance contracts, and remote diagnostics—are gaining traction, particularly among smaller clinics.
In conclusion, the phacoemulsification machine market in 2026 will be characterized by technological sophistication, geographic expansion, and patient-centered innovation. Stakeholders who adapt to these evolving dynamics—through R&D investment, strategic market entry, and digital integration—are likely to capture significant growth opportunities.

Common Pitfalls When Sourcing Phacoemulsification Machines: Quality and Intellectual Property Concerns
Sourcing phacoemulsification machines—critical devices in cataract surgery—requires careful due diligence to ensure both high performance and legal compliance. Two major areas of risk are product quality and intellectual property (IP) infringement. Below are common pitfalls in these areas:
1. Compromised Quality Due to Substandard Manufacturing
-
Inadequate Regulatory Compliance: Some suppliers, especially from regions with lax oversight, may offer machines that do not meet FDA, CE, or other international regulatory standards. This increases patient risk and exposes healthcare providers to liability.
-
Use of Inferior Components: Low-cost machines may use subpar transducers, handpieces, or fluidics systems, leading to inconsistent emulsification, increased surgical time, or intraoperative complications.
-
Lack of Clinical Validation: Some manufacturers do not provide peer-reviewed clinical data or real-world performance metrics, making it difficult to assess efficacy and safety.
-
Poor After-Sales Support and Maintenance: Machines sourced from unreliable vendors often come with limited technical support, unavailable spare parts, or untrained service personnel, affecting long-term usability.
2. Intellectual Property (IP) Infringement Risks
-
Counterfeit or Clone Devices: Some suppliers produce near-identical copies of established brands (e.g., Alcon, Bausch + Lomb, Johnson & Johnson), violating patents and trademarks. These clones may mimic design and functionality but lack the innovation and safety testing of the original.
-
Unauthorized Use of Proprietary Technology: Certain machines may incorporate patented ultrasonic control algorithms, torsional phaco technology, or software features without licensing, exposing buyers to legal risks if used in regulated markets.
-
Difficulty Tracing IP Ownership: In complex supply chains, especially with OEM manufacturers in Asia, it can be challenging to verify whether the product was developed legally or if it infringes on existing IP rights.
-
Exposure to Legal Action: Hospitals or clinics using IP-infringing equipment may face cease-and-desist orders, fines, or reputational damage, especially in jurisdictions with strong IP enforcement.
Mitigation Strategies
-
Verify Regulatory Approvals: Ensure the device has valid certifications (e.g., FDA 510(k), CE Mark, ISO 13485) and is listed in official medical device databases.
-
Conduct Supplier Audits: Evaluate manufacturing facilities, quality management systems, and service capabilities before procurement.
-
Perform IP Due Diligence: Consult legal experts to review patents and verify that the technology does not infringe on existing IP, particularly if sourcing from lesser-known manufacturers.
-
Purchase from Authorized Distributors: Whenever possible, source directly from original manufacturers or certified distributors to avoid counterfeit or unauthorized devices.
Avoiding these pitfalls ensures not only the safety and effectiveness of cataract procedures but also protects healthcare institutions from legal and operational risks.

Logistics & Compliance Guide for Phacoemulsification Machine
This guide outlines the critical logistics and compliance considerations for the safe, legal, and efficient import, distribution, installation, and use of phacoemulsification machines.
Regulatory Classification & Approval
Phacoemulsification machines are classified as medical devices, typically falling into Class II (moderate to high risk) or Class III (high risk) in most regulatory jurisdictions. Key regulatory bodies include:
* USA: Food and Drug Administration (FDA) – Requires 510(k) premarket notification or Premarket Approval (PMA).
* European Union: Conformité Européenne (CE) marking under the Medical Device Regulation (MDR) 2017/745.
* Canada: Health Canada – Medical Device License (MDL) required.
* Other Countries: Local regulatory authorities (e.g., TGA in Australia, PMDA in Japan, NMPA in China) with specific registration and approval processes.
Ensure the device has valid regulatory clearance or approval for each target market before shipment or sale.
Import & Export Documentation
Accurate and complete documentation is essential for international trade:
* Commercial Invoice: Detailed description of goods, value, currency, parties involved.
* Packing List: Itemized list of contents per package, weights, dimensions.
* Bill of Lading (B/L) or Air Waybill (AWB): Contract of carriage and receipt of goods.
* Certificate of Origin: Indicates country of manufacture.
* Regulatory Certificates: Copy of FDA 510(k)/PMA, CE Certificate of Conformity, or other local market approvals.
* Manufacturer’s Declaration: Statement confirming compliance with relevant standards.
* Export License (if required): For certain countries or dual-use technologies.
Verify specific import requirements (e.g., import permits, pre-shipment inspections) with the destination country’s customs and health authority.
Packaging & Transportation
Phacoemulsification machines require specialized handling:
* Original Manufacturer Packaging: Use only the manufacturer-approved packaging with shock-absorbing materials.
* Climate Control: Protect from extreme temperatures, humidity, and direct sunlight. Ship via temperature-controlled (if specified) or ambient stable conditions.
* Fragile Handling: Clearly label packages as “Fragile,” “This Side Up,” and “Medical Equipment.”
* Secure Transport: Use reputable carriers experienced in handling high-value medical devices. Ensure the machine is secured to prevent movement during transit.
* Avoid Stacking: Prevent placing heavy items on top of the package.
Customs Clearance
Facilitate smooth customs clearance:
* Accurate HS Code: Use the correct Harmonized System (HS) code (e.g., 9018.19 for ophthalmic instruments) for the device.
* Duties & Taxes: Be aware of applicable import duties, VAT, or GST. Utilize duty exemptions where available (e.g., under the Information Technology Agreement – ITA).
* Regulatory Inspection: Be prepared for customs or health authority inspection to verify documentation and compliance.
* Authorized Representative: In regions like the EU, ensure an EU Authorized Representative (EU AR) is designated to liaise with authorities.
Installation & Commissioning
Proper setup is critical for safety and performance:
* Trained Technician: Installation must be performed by a manufacturer-certified or qualified biomedical technician.
* Site Preparation: Ensure adequate power supply (voltage, grounding), space, ventilation, and integration with other OR equipment.
* Verification & Calibration: Perform initial safety checks, functional testing, and calibration as per manufacturer’s protocol.
* Documentation: Record installation details, calibration results, and sign-off by the technician and facility representative.
Quality Management System (QMS) Compliance
Distributors and importers must comply with QMS standards:
* ISO 13485: Implement a Quality Management System for medical device distribution, covering documentation, storage, handling, and traceability.
* Unique Device Identification (UDI): Ensure devices bear UDI labels (DI and PI) and maintain UDI records in compliance with FDA, EU MDR, and other regulations.
* Traceability: Maintain records to trace devices from manufacturer to end-user (and vice versa) for recalls or field safety actions.
Post-Market Surveillance & Vigilance
Ongoing monitoring is mandatory:
* Adverse Event Reporting: Establish processes to report serious incidents and field safety corrective actions (FSCAs) to relevant authorities (e.g., FDA MAUDE, EUDAMED).
* Customer Feedback: Monitor and document customer complaints and technical issues.
* Recall Management: Have a plan to execute recalls or safety alerts promptly if required.
Training & User Documentation
Ensure safe and effective use:
* User Manuals: Provide up-to-date, language-appropriate instructions for use (IFU), including warnings, precautions, and maintenance.
* Operator Training: Mandatory training for surgeons and OR staff on device operation, safety features, and emergency procedures.
* Service Manuals: Provide technical documentation to authorized service personnel.
Environmental & Disposal Compliance
Dispose of devices and packaging responsibly:
* WEEE Directive (EU): Comply with Waste Electrical and Electronic Equipment regulations.
* Local E-Waste Laws: Follow national and regional regulations for medical device disposal.
* Hazardous Materials: Identify and handle any hazardous components (e.g., batteries) according to local environmental laws.
Adherence to this guide ensures regulatory compliance, patient safety, and uninterrupted clinical use of phacoemulsification equipment.
Conclusion for Sourcing a Phacoemulsification Machine
In conclusion, sourcing a phacoemulsification machine is a critical investment that significantly impacts the quality, efficiency, and safety of cataract surgeries within an ophthalmic practice or healthcare facility. The selection process should be guided by a comprehensive evaluation of clinical requirements, technological features, budget constraints, service and support availability, and long-term value. Key considerations include the machine’s surgical versatility (e.g., torsional ultrasound, fluidics control, customizable settings), compatibility with current surgical instruments, ease of use, and integration with digital operating room systems.
While premium brands may offer advanced features and superior reliability, cost-effective alternatives from emerging manufacturers can provide valuable functionality for facilities with limited budgets—provided they meet regulatory standards and clinical performance expectations. Additionally, post-purchase support, warranty terms, training programs, and spare parts availability are essential for ensuring uninterrupted service and staff proficiency.
Ultimately, the ideal phacoemulsification machine balances innovation, reliability, and cost-efficiency to enhance surgical outcomes, improve patient safety, and support the growth of ophthalmic services. A well-informed sourcing decision, based on thorough research, stakeholder input, and clinical trials if possible, ensures long-term success and sustainability in delivering high-quality eye care.