The global pacemaker market is witnessing steady expansion, driven by rising prevalence of cardiovascular diseases, an aging global population, and advances in cardiac rhythm management technologies. According to Mordor Intelligence, the pacemaker market was valued at approximately USD 5.2 billion in 2023 and is projected to grow at a compound annual growth rate (CAGR) of over 6.8% from 2024 to 2029. This growth is further underpinned by increasing adoption of minimally invasive procedures and the introduction of innovative devices such as leadless pacemakers. Grand View Research also highlights a similar trajectory, citing growing demand for remote monitoring capabilities and device longevity as key market drivers. In this evolving landscape, a handful of key manufacturers dominate the industry, combining technological innovation, global distribution networks, and strong regulatory footholds to maintain leadership. Below are the top 7 pacemaker manufacturers shaping the future of cardiac care.
Top 7 Pacemaker Companies Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Pacemakers
Domain Est. 1996
Website: bostonscientific.com
Key Highlights: Boston Scientific advanced pacemaker technology. Offering respiration-based pacing for improved rate response….
#2 Cardiovascular Products and Solutions
Domain Est. 1988
Website: abbott.com
Key Highlights: Abbott’s cardiovascular products include pacemakers, heart valve therapies, LVADs, insertable cardiac monitors and ablation systems to support heart health….
#3 Medtronic
Domain Est. 1990
Website: medtronic.com
Key Highlights: From the world’s smallest pacemaker to robotic-assisted surgery, Medtronic transforms the lives of millions worldwide. Learn more. Your browser does not ……
#4 BIOTRONIK
Domain Est. 1996
Website: biotronik.com
Key Highlights: At BIOTRONIK, we engineer trusted, life-changing therapies through our advanced active implants in Cardiac Rhythm Management, Monitoring, ……
#5 Vitatron
Domain Est. 1997
Website: vitatron.com
Key Highlights: Vitatron is a European based international pacemaker company with a rich history. We started in 1956, implanting our first pacemaker in 1962….
#6 EBR Systems, Inc
Domain Est. 2005
Website: ebrsystemsinc.com
Key Highlights: EBR aims for outstanding treatment of cardiac rhythm diseases with an innovative leadless therapy….
#7 Cardiac Pacemakers
Domain Est. 2018
Website: cardiovascular.abbott
Key Highlights: Abbott offers multiple pacemaker options with unique pacemaker functionality, so you can determine the best pacemaker option for your patients’ conditions….
Expert Sourcing Insights for Pacemaker Companies
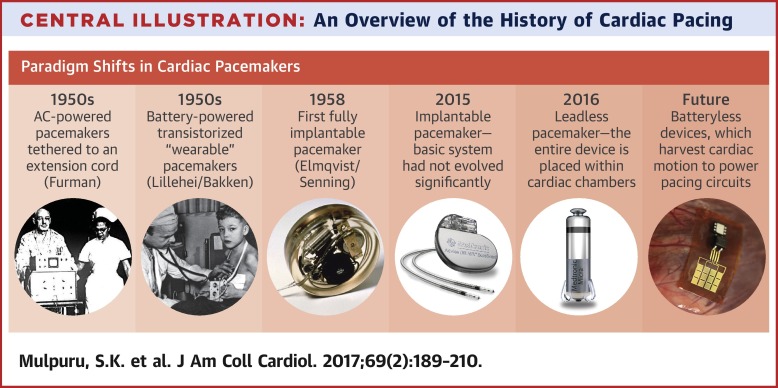
2026 Market Trends for Pacemaker Companies
The global pacemaker market is poised for significant transformation by 2026, driven by demographic shifts, technological innovation, and evolving healthcare dynamics. Pacemaker companies must navigate these trends strategically to maintain competitiveness and address growing patient needs.
1. Rising Demand Driven by Aging Populations and Cardiovascular Disease Prevalence
By 2026, the global burden of cardiovascular diseases (CVDs), particularly arrhythmias like atrial fibrillation and bradycardia, will continue to escalate. The aging population—especially in North America, Europe, and parts of Asia—will be a primary growth driver. As life expectancy increases, so does the incidence of age-related cardiac conduction disorders, directly boosting demand for both traditional and advanced pacemakers. The World Health Organization estimates CVDs remain the leading cause of death worldwide, reinforcing long-term market expansion.
2. Technological Advancements: Leadless and MRI-Conditional Devices
Innovation will define market leadership in 2026. Leadless pacemakers—small, self-contained units implanted directly into the heart—are expected to gain substantial market share. Devices like the Micra AV/VR by Medtronic and the Aveir by Abbott offer minimally invasive implantation, reduced complication risks (e.g., lead fractures or infections), and faster recovery. Additionally, MRI-conditional pacemakers will become the standard, allowing patients greater access to critical diagnostic imaging without device interference. Integration with remote monitoring and AI-powered diagnostics will further enhance treatment personalization and proactive care.
3. Expansion of Remote Patient Monitoring and Digital Health Integration
Remote monitoring will be a cornerstone of pacemaker management by 2026. Systems such as Boston Scientific’s Latitude™ and Medtronic’s CareLink® will leverage cloud-based platforms and mobile connectivity to transmit real-time device data. This reduces hospital visits, enables early detection of arrhythmias or device malfunctions, and improves patient outcomes. Integration with electronic health records (EHRs) and telehealth platforms will be essential, supported by favorable reimbursement policies in key markets like the U.S. and parts of Europe.
4. Competitive Landscape and Strategic Consolidation
The pacemaker market will remain highly consolidated, dominated by major players such as Medtronic, Abbott, Boston Scientific, and Biotronik. However, increasing competition from emerging-market entrants and startups focusing on cost-effective or AI-enhanced solutions may disrupt traditional models. In 2026, strategic partnerships, mergers, and acquisitions will likely accelerate as companies seek to broaden portfolios, access new geographies, or integrate digital health capabilities.
5. Regulatory and Reimbursement Challenges
Navigating complex regulatory pathways—especially FDA approvals in the U.S. and CE marking in Europe—will remain critical. Companies investing in robust clinical trials demonstrating safety, efficacy, and cost-effectiveness will gain faster market access. Reimbursement pressures, particularly in public healthcare systems, will push manufacturers to justify premium pricing through outcomes-based data. Value-based healthcare models may incentivize devices that reduce long-term hospitalization and improve quality of life.
6. Emerging Markets as Growth Frontiers
While developed markets remain dominant, regions such as Asia-Pacific (especially China and India), Latin America, and the Middle East will see accelerated adoption. Rising healthcare spending, improved medical infrastructure, and growing awareness of cardiac care will open new opportunities. Local manufacturing partnerships and tiered pricing strategies will be key for pacemaker companies to penetrate these cost-sensitive yet high-potential markets.
In conclusion, by 2026, pacemaker companies will need to prioritize innovation, digital integration, and global accessibility to thrive. Success will depend on balancing cutting-edge technology with cost-effectiveness and seamless patient care ecosystems.

Common Pitfalls Sourcing Pacemaker Companies (Quality, IP)
Sourcing pacemaker components or partnering with pacemaker manufacturers involves navigating significant risks, particularly concerning quality and intellectual property (IP). Failing to address these pitfalls can result in regulatory non-compliance, product recalls, legal disputes, and reputational damage.
Quality-Related Pitfalls
Inadequate Supplier Qualification and Auditing
One of the most critical mistakes is failing to conduct thorough due diligence on potential suppliers. Without rigorous audits—especially unannounced on-site inspections—companies may unknowingly partner with vendors lacking robust quality management systems (QMS) compliant with ISO 13485 and FDA 21 CFR Part 820. Substandard manufacturing practices, inconsistent component tolerances, or poor documentation can compromise device reliability and patient safety.
Lack of Traceability and Documentation
Pacemakers require full traceability from raw materials to finished devices. Sourcing from suppliers who do not maintain comprehensive device history records (DHR) or device master records (DMR) increases the risk of undetectable defects and complicates root cause analysis during investigations or recalls. Missing or falsified documentation is a red flag for poor quality control.
Insufficient Validation of Processes and Materials
Using components without proper process validation (e.g., sterilization, sealing, or battery longevity testing) can lead to premature device failure. Suppliers may cut corners on material sourcing or fail to validate long-term performance under real-world conditions, which is especially critical for implantable devices with lifespans of 10+ years.
Intellectual Property-Related Pitfalls
Unclear IP Ownership and Licensing
Ambiguity in contracts regarding IP ownership—especially for custom-designed components or firmware—can result in legal disputes. Sourcing from suppliers who claim rights to improvements or jointly developed technology may lead to infringement claims or loss of exclusivity. Ensure agreements explicitly assign IP to the buyer or define clear licensing terms.
Risk of IP Infringement
Partnering with suppliers who use third-party patented technologies (e.g., in microelectronics or telemetry) without proper licensing exposes the sourcing company to litigation. Due diligence must include IP landscape analysis and freedom-to-operate (FTO) assessments to avoid unintentional infringement.
Inadequate Protection of Trade Secrets
When sharing sensitive design specifications or algorithms with suppliers, insufficient non-disclosure agreements (NDAs) or weak cybersecurity measures can lead to trade secret exposure. This is especially dangerous in high-competition fields like cardiac rhythm management, where algorithm efficiency and miniaturization are key differentiators.
Geopolitical and Regulatory Compliance Risks
Sourcing from regions with lax IP enforcement increases the risk of reverse engineering and unauthorized replication. Additionally, export controls (e.g., ITAR, EAR) may restrict the transfer of certain medical device technologies, and non-compliance can lead to severe penalties.
Mitigation Strategies
To avoid these pitfalls, companies should implement robust supplier qualification programs, conduct regular audits, enforce clear contractual IP terms, perform FTO analyses, and invest in secure collaboration frameworks. Engaging legal and regulatory experts early in the sourcing process is essential to ensure both quality and IP integrity in pacemaker development and manufacturing.
Logistics & Compliance Guide for Pacemaker Companies
Pacemaker manufacturing and distribution involve complex logistics and stringent regulatory compliance due to the life-sustaining nature of the devices. This guide outlines key considerations for companies involved in the supply chain of pacemakers.
Regulatory Framework and Compliance Requirements
Pacemaker companies must adhere to a global network of medical device regulations. Key regulatory bodies include the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and Health Canada. Compliance includes:
- FDA 21 CFR Part 820 (Quality System Regulation): Mandates design controls, production and process controls, and corrective and preventive actions (CAPA).
- EU Medical Device Regulation (MDR) 2017/745: Requires conformity assessment, technical documentation, and post-market surveillance.
- ISO 13485 Certification: International standard for quality management systems specific to medical devices. Essential for global market access.
All pacemakers must undergo rigorous clinical evaluation and obtain regulatory approval (e.g., 510(k) or PMA in the U.S.) before commercial distribution.
Supply Chain Management and Vendor Qualification
Due to the critical nature of pacemakers, supply chain integrity is paramount. Companies must:
- Qualify and audit all suppliers of critical components (e.g., batteries, microprocessors, leads).
- Maintain traceability from raw materials to finished devices using Unique Device Identification (UDI) systems.
- Implement dual sourcing strategies for essential components to mitigate supply disruptions.
Regular audits and supplier performance reviews are required to ensure continuous compliance with quality and delivery standards.
Inventory and Distribution Logistics
Pacemaker distribution requires strict temperature control, secure handling, and rapid delivery:
- Cold Chain Management: While pacemakers themselves are typically not temperature-sensitive, certain components or testing materials may require controlled environments. Monitor storage conditions per manufacturer specifications.
- Secure Transportation: Use tamper-evident packaging and GPS-tracked shipments to prevent theft or tampering.
- Just-in-Time (JIT) Delivery: Many healthcare facilities require immediate delivery; logistics partners must support 24/7 availability and emergency dispatch capabilities.
Inventory systems must support real-time tracking and reconciliation to ensure device availability and support recalls if necessary.
Import/Export and Customs Compliance
International shipments of pacemakers are subject to customs regulations and export controls:
- Obtain proper export licenses (e.g., U.S. Commerce Department’s Export Administration Regulations).
- Comply with import requirements in destination countries, including local registration, labeling in local language, and adherence to import permits.
- Classify devices correctly under the Harmonized System (HS) codes to avoid delays.
Collaborate with experienced freight forwarders familiar with medical device logistics and regulatory documentation.
Post-Market Surveillance and Recall Management
Post-launch monitoring is a regulatory mandate:
- Implement robust post-market surveillance (PMS) systems to collect and analyze adverse events.
- Establish a medical device reporting (MDR) process to notify regulatory authorities of malfunctions or patient injuries.
- Develop a comprehensive recall plan that includes rapid communication to distributors, hospitals, and implanted patients.
UDI integration enables precise identification of affected devices, minimizing public health risks during recalls.
Data Security and Patient Privacy
Pacemakers may transmit patient data (e.g., remote monitoring systems), requiring compliance with data protection laws:
- Ensure HIPAA compliance in the U.S. and GDPR adherence in the EU.
- Encrypt data transmissions and implement access controls to protect patient health information (PHI).
- Conduct regular cybersecurity risk assessments, especially for connected devices.
Training and Documentation
Maintain comprehensive records and train personnel on:
- Regulatory updates and changes in compliance requirements.
- Standard operating procedures (SOPs) for logistics, handling, and distribution.
- Emergency response protocols, including recall execution and incident reporting.
All activities must be documented and available for regulatory inspections.
Adhering to these logistics and compliance guidelines ensures patient safety, regulatory approval, and operational excellence in the pacemaker industry.
Conclusion: Sourcing Pacemaker Companies
In conclusion, sourcing pacemaker suppliers requires a strategic approach that balances quality, regulatory compliance, technological innovation, cost-efficiency, and reliable supply chain management. The global market for pacemakers is dominated by key players such as Medtronic, Abbott, Boston Scientific, and Biotronik, all of which offer advanced, FDA- and CE-approved devices with proven clinical performance. When selecting a supplier, healthcare providers, distributors, or procurement organizations must prioritize manufacturers with strong regulatory credentials, robust post-market support, and a commitment to innovation in areas such as leadless technology, remote monitoring, and battery longevity.
Additionally, considerations such as total cost of ownership, training availability, and integration with existing healthcare systems play a crucial role in decision-making. Engaging with suppliers that demonstrate transparency, ethical manufacturing practices, and responsiveness to customer needs ensures long-term partnership success. Ultimately, the right sourcing decision enhances patient outcomes, supports clinical efficacy, and strengthens the reliability of cardiac care delivery. A thorough evaluation of pacemaker companies—aligned with organizational goals and clinical requirements—will lead to optimal procurement outcomes in this highly specialized and critical medical device sector.