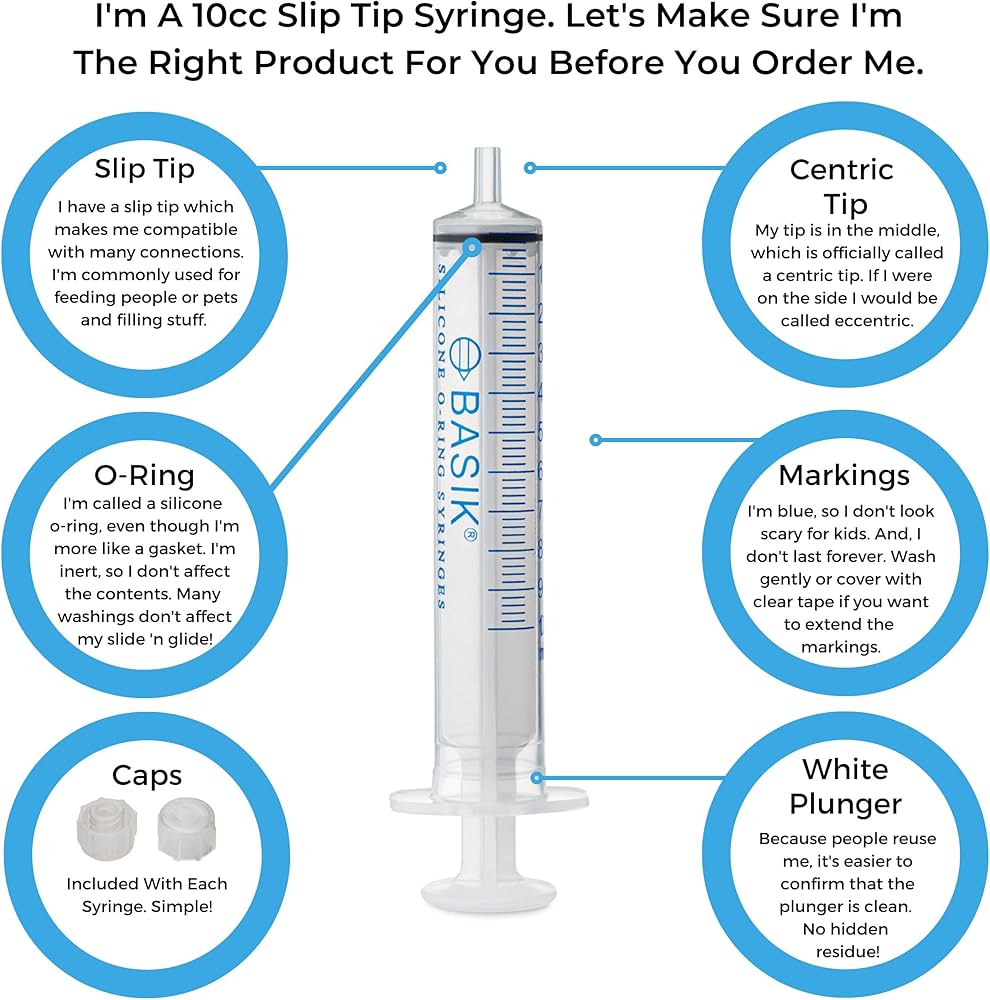
The global o-ring syringes market is poised for steady expansion, driven by increasing demand for precision fluid handling in industries such as pharmaceuticals, biotechnology, and analytical laboratories. According to Grand View Research, the global syringes market size was valued at USD 17.8 billion in 2022 and is expected to grow at a CAGR of 7.1% from 2023 to 2030, with advanced syringe types—including those with o-ring seals—gaining traction due to their superior sealing performance and reliability. Similarly, Mordor Intelligence projects a CAGR of approximately 6.8% for the global syringe market over the forecast period, citing rising adoption of high-precision syringes in research and clinical applications. As quality, accuracy, and chemical compatibility become critical selection criteria, leading manufacturers are innovating in materials, design, and manufacturing processes. In this evolving landscape, here are the top 10 o-ring syringes manufacturers shaping industry standards through technological excellence and global reach.
Top 10 O Ring Syringes Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Silicone O-Rings Supplier
Domain Est. 2004
Website: azseal.com
Key Highlights: We’re a major supplier of standard and FDA-compliant silicone O-rings that meet the needs of both general and specialized industrial applications….
#2 O
Domain Est. 2009
Website: henryspets.com
Key Highlights: Free delivery over $100Henry’s carries medical-grade, easy-to-clean polypropylene syringes by top European manufacturers like Ardes® and CODAN®, perfectly sized for administering…
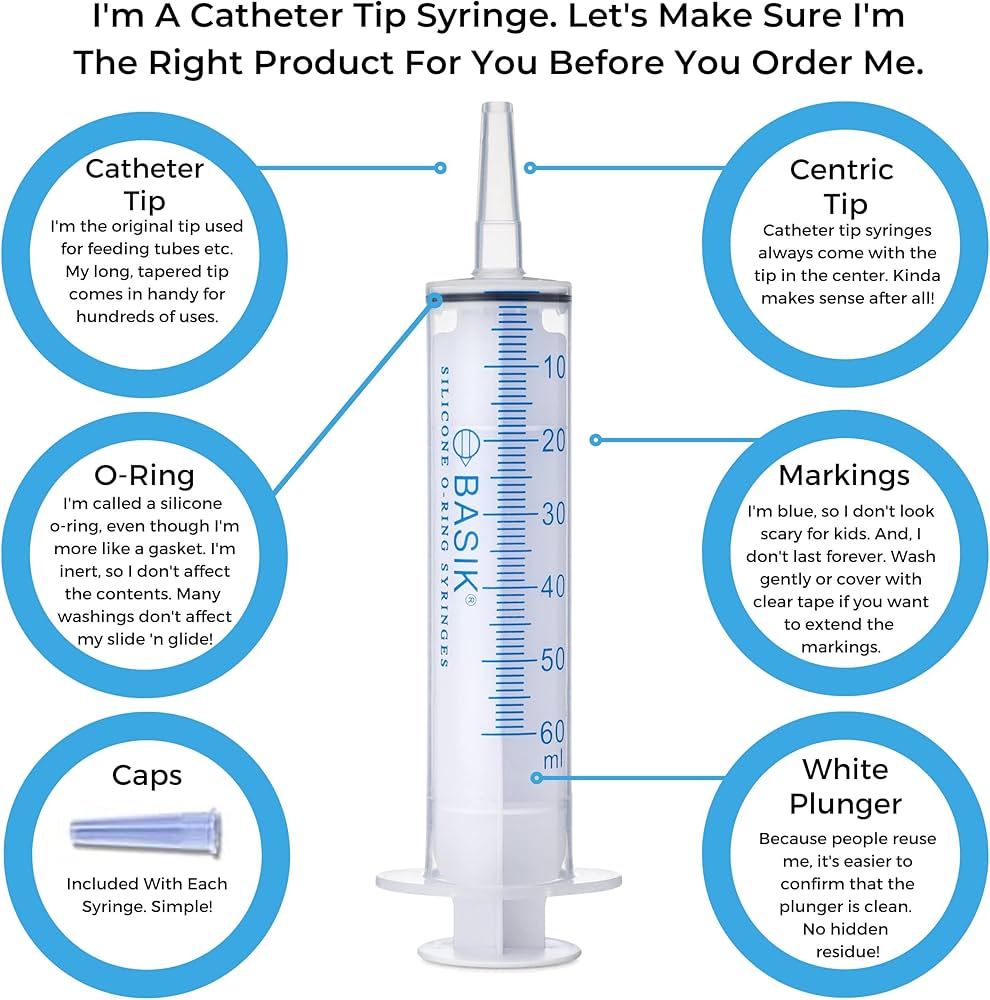
#3 Basik O
Domain Est. 1996
Website: medcareproducts.com
Key Highlights: 45-day returnsBasik silicone O-ring syringes are very popular for enteral feeding, tube feeding, hand feeding animals, artificial insemination and manufacturing….
#4 Irrigation Syringes
Domain Est. 1996
Website: catalogs.bbraun.com
Key Highlights: Irrigation syringes are hypodermic 3-piece syringes with catheter tip. The single-use devices are intended to be used for the following purposes:…
#5 Dosys™ classic 163
Domain Est. 1996
Website: socorex.com
Key Highlights: Self-refilling, two-ring and automatic syringe pipette with integrated, Luer Lock valve system. Volumes ranging 0.025 to 10 mL….
#6 Hamilton Company
Domain Est. 1998
Website: hamiltoncompany.com
Key Highlights: From precision syringes to advanced automation, Hamilton delivers engineered solutions that support every stage of your work. Whether you’re optimizing ……
#7 O
Domain Est. 2000
#8 Buna-N Syringe O-Ring Replacements – NBR Nitrile
Domain Est. 2006
#9 Air
Domain Est. 2007
Website: air-tite-shop.com
Key Highlights: 4-day delivery 540-day returnsOur catalog and website feature high-quality B. Braun Omnifix® Luer Lock Syringes made in Germany. Slow aspiration and injections are made easy by the…
#10 Products
Domain Est. 2013
Website: vescomedical.com
Key Highlights: Enteral Drainage · Enteral Pharmacy Items · Extension Feeding Sets · Enteral Feeding Pump · Enteral Syringes · Enteral Feeding Sets · Enteral Feeding Tubes….
Expert Sourcing Insights for O Ring Syringes

H2: 2026 Market Trends for O-Ring Syringes
The O-ring syringe market in 2026 is poised for significant evolution, driven by advancements in materials science, rising demand in specialized medical and industrial applications, and a growing emphasis on precision and sustainability. Here are the key trends shaping the market:
1. Increased Demand in Precision Medicine and Biopharmaceuticals:
O-ring syringes are gaining traction in high-precision applications such as biologics delivery, cell therapy, and lab automation. Their ability to provide consistent plunger movement and minimal dead space makes them ideal for handling expensive and sensitive formulations. By 2026, expanded use in personalized medicine and cold-chain drug delivery systems will drive volume growth, particularly in pre-filled syringe formats with integrated O-ring seals.
2. Material Innovation and Chemical Resistance:
Advancements in elastomer technology are leading to next-generation O-rings made from fluorocarbon (FKM), perfluoroelastomers (FFKM), and specialty silicones. These materials offer superior resistance to aggressive chemicals, solvents, and high temperatures—critical for industrial and pharmaceutical manufacturing. In 2026, demand will shift toward syringes with chemically inert O-rings capable of withstanding sterilization processes and maintaining integrity over extended storage.
3. Growth in Industrial and Laboratory Automation:
Automated liquid handling systems in R&D, diagnostics, and quality control increasingly rely on O-ring syringes for their durability and leak-free performance. The push for higher throughput and reproducibility in labs will boost demand for motorized syringe pumps and robotic workstations equipped with O-ring sealed syringes. This trend is expected to accelerate through 2026, especially in genomics, proteomics, and analytical chemistry.
4. Focus on Sustainability and Single-Use Systems:
Environmental regulations and sustainability goals are influencing syringe design. While O-ring syringes are often reusable, there is a growing market for single-use, pre-sterilized versions in bioprocessing to reduce cross-contamination risks. By 2026, manufacturers will prioritize recyclable materials and modular designs that extend product life, balancing sterility needs with eco-conscious production.
5. Regional Market Expansion and Regulatory Alignment:
Emerging markets in Asia-Pacific and Latin America are expanding their healthcare infrastructure, increasing demand for reliable and cost-effective syringe solutions. Simultaneously, regulatory bodies like the FDA and EMA are tightening standards for extractables and leachables in medical devices. O-ring syringes that comply with ISO 13485 and USP Class VI standards will have a competitive edge, driving innovation in compliant manufacturing processes.
6. Integration with Smart Technologies:
The rise of connected medical devices is influencing syringe design. By 2026, we may see hybrid O-ring syringes integrated with sensors or RFID tags for dose tracking, usage monitoring, and data logging—especially in clinical trials and home healthcare settings.
In summary, the 2026 O-ring syringe market will be characterized by technological sophistication, material excellence, and application-specific customization. Companies that invest in R&D for high-performance seals, sustainable practices, and smart integration will lead the market in this evolving landscape.

Common Pitfalls Sourcing O-Ring Syringes (Quality, IP)
Sourcing O-ring syringes—commonly used in laboratories, medical devices, and precision fluid handling—can present significant challenges, particularly concerning quality consistency and intellectual property (IP) risks. Overlooking these pitfalls can lead to product failure, regulatory non-compliance, or legal disputes.
Quality Inconsistencies and Material Defects
One of the most frequent issues when sourcing O-ring syringes, especially from low-cost or unverified suppliers, is inconsistent product quality. Variations in syringe barrel smoothness, plunger fit, or O-ring dimensional tolerances can lead to leakage, inconsistent fluid delivery, or premature failure. Poor-quality materials, such as non-chemically resistant plastics or substandard elastomers in the O-ring (e.g., incorrect durometer or material grade), may degrade when exposed to solvents or sterilization processes. Suppliers may also lack proper quality control documentation (e.g., ISO 13485 certification), making traceability and compliance difficult, particularly in regulated industries.
Intellectual Property Infringement Risks
O-ring syringe designs, especially those used in patented medical or analytical devices, are often protected by intellectual property rights. Sourcing generic or “compatible” syringes from third-party manufacturers can inadvertently lead to IP infringement if the design replicates patented features such as locking mechanisms, barrel geometry, or plunger assembly. Some suppliers, particularly in regions with weak IP enforcement, may produce look-alike products that closely mimic branded versions (e.g., equivalents to Hamilton or SGE syringes) without proper licensing. Buyers may face legal liability even if unaware of the infringement, especially when importing or commercializing such products. Conducting due diligence on supplier legitimacy and requesting IP indemnification clauses in contracts is essential to mitigate these risks.

Logistics & Compliance Guide for O-Ring Syringes
O-ring syringes, commonly used in laboratory, pharmaceutical, and industrial applications, require careful handling, storage, and compliance with regulatory standards to ensure product integrity, safety, and legal conformity. This guide outlines key logistics and compliance considerations for the transportation, storage, and use of O-ring syringes.
Regulatory Compliance
O-ring syringes may be subject to various regulatory frameworks depending on their intended use and destination. Compliance is essential to avoid legal issues and ensure user safety.
Medical-Grade Syringes
If the O-ring syringes are intended for medical use (e.g., drug delivery, clinical applications), they must comply with medical device regulations such as:
– FDA 21 CFR Part 820 (Quality System Regulation) – for syringes marketed in the United States.
– EU MDR (Medical Device Regulation 2017/745) – required for CE marking and placement on the European market.
– ISO 13485 – international standard for quality management systems in medical device manufacturing.
Laboratory & Industrial Use
For non-medical applications, compliance with general safety and material standards may apply, such as:
– REACH and RoHS (EU) – for chemical substance restrictions and hazardous material compliance.
– OSHA (USA) – relevant if syringes are used with hazardous materials in workplaces.
Ensure product labeling includes appropriate symbols, lot numbers, expiration dates (if applicable), and usage instructions.
Packaging and Labeling Requirements
Proper packaging and labeling are critical for protecting syringe integrity and meeting regulatory and shipping standards.
- Use sealed, sterile packaging if required (e.g., for medical or cleanroom applications).
- Clearly label packages with:
- Product name and model number
- Quantity and batch/lot number
- Manufacturer and distributor information
- Storage conditions (e.g., “Store in a cool, dry place”)
- Regulatory markings (e.g., CE, FDA registration number)
- Hazard warnings (if containing lubricants or used with hazardous substances)
Storage Conditions
Maintain appropriate environmental conditions to prevent degradation of syringes and O-rings.
- Temperature: Store between 15°C and 25°C (59°F to 77°F) unless otherwise specified by the manufacturer.
- Humidity: Keep in a dry environment (<60% relative humidity) to prevent mold or material deterioration.
- Light Exposure: Protect from direct sunlight and UV radiation, which may degrade certain plastics or elastomers.
- Shelf Life: Monitor expiration dates; O-rings made from elastomeric materials (e.g., silicone, Viton) can degrade over time, affecting seal performance.
Transportation and Handling
Ensure safe and compliant shipping practices to prevent damage and contamination.
- Use protective packaging (e.g., bubble wrap, rigid boxes) to prevent physical damage during transit.
- Avoid extreme temperatures during transport; use climate-controlled logistics for temperature-sensitive batches.
- Follow IATA, IMDG, or other relevant transport regulations if shipping with hazardous substances or across international borders.
- Implement a chain-of-custody process for traceability, especially for regulated or high-risk applications.
Material and Chemical Compatibility
Verify compatibility between O-ring materials and substances used with the syringes.
- Common O-ring materials include silicone, nitrile (Buna-N), EPDM, and fluorocarbon (Viton).
- Refer to chemical resistance charts to ensure the O-ring will not degrade when exposed to solvents, acids, or other fluids.
- Document compatibility assessments for audit or compliance purposes.
Quality Assurance and Documentation
Maintain comprehensive records to support traceability and compliance.
- Retain certificates of conformance (CoC), material declarations (e.g., RoHS, REACH), and sterilization reports (if applicable).
- Conduct regular audits of suppliers and logistics partners.
- Implement a system for handling non-conforming products and recalls, if necessary.
Disposal and Environmental Considerations
Follow proper waste management protocols.
- Dispose of used syringes according to local biohazard, chemical, or industrial waste regulations.
- Recycle packaging materials where possible.
- Avoid incineration of certain plastics or elastomers that may release toxic fumes.
By adhering to this logistics and compliance guide, organizations can ensure the safe, legal, and effective handling of O-ring syringes throughout their lifecycle.
Conclusion for Sourcing O-Ring Syringes
In conclusion, sourcing O-ring syringes requires a careful evaluation of material quality, dimensional accuracy, regulatory compliance, and supplier reliability. These specialized syringes, designed with O-rings to ensure a secure seal and consistent performance, are critical in applications demanding precision and contamination control—such as in pharmaceuticals, diagnostics, and laboratory research.
Key considerations include selecting chemically resistant materials (such as borosilicate glass or specific polymers) that are compatible with the intended fluids, ensuring tight manufacturing tolerances for consistent plunger movement, and verifying compliance with industry standards such as ISO 7886 or USP Class VI. Additionally, choosing suppliers with a proven track record, strong quality management systems (e.g., ISO 13485 certification), and the capacity for scalability ensures long-term supply chain stability.
Ultimately, the right sourcing strategy balances performance requirements with cost-efficiency, while mitigating risks through supplier audits, sample testing, and contractual quality assurances. By prioritizing these factors, organizations can secure reliable, high-quality O-ring syringes that support the integrity and success of their critical applications.