The global needle decompression kit market is experiencing steady growth, driven by rising demand for emergency medical interventions in pre-hospital and battlefield settings. According to a report by Mordor Intelligence, the global emergency medical devices market—which includes critical care tools like needle decompression kits—is projected to grow at a CAGR of approximately 7.2% from 2023 to 2028. This expansion is fueled by increasing incidences of traumatic injuries, heightened focus on emergency preparedness, and the integration of advanced medical technologies in first-response care. With tension pneumothorax remaining a life-threatening condition requiring immediate intervention, the need for reliable, portable, and easy-to-use needle decompression kits has become paramount across military, EMS, and hospital environments. In line with this demand, several manufacturers have emerged as key innovators, combining clinical efficacy with ergonomic design to improve survival outcomes. The following analysis identifies the top seven manufacturers shaping this essential segment of emergency care.
Top 7 Needle Decompression Kit Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
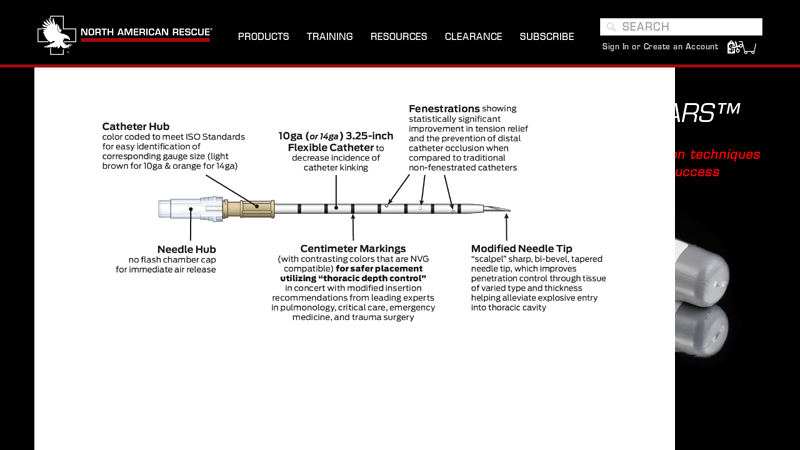
#1 Enhanced ARS Decompression Needle
Domain Est. 1996
Website: narescue.com
Key Highlights: The Enhanced ARS™ features an improved needle tip design with “scalpel” sharp, bi-bevel, tapered needle tip, which improves penetration control through tissue ……
#2 Chest Drain & Needle Decompression Trainer with Augmented Reality
Domain Est. 1996
Website: limbsandthings.com
Key Highlights: The Chest Drain and Needle Decompression Trainer is the ideal simulator for training in both surgical and guide-wire assisted thoracostomy and ……
#3 Decompression Needle
Domain Est. 1999
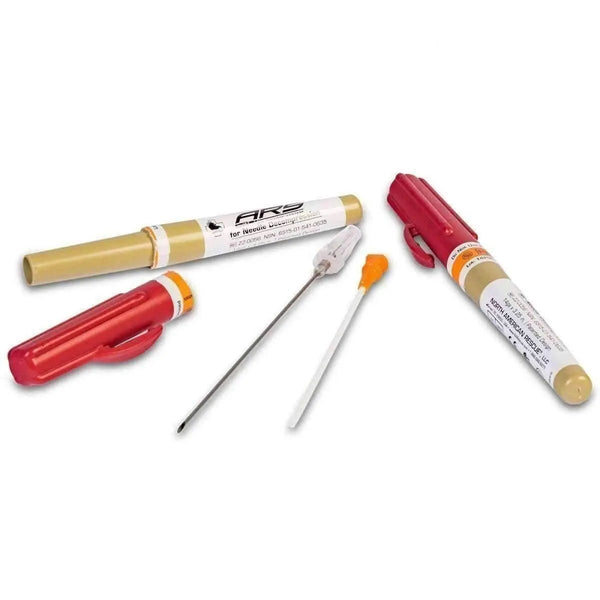
#4 Needle Decompression Kit (ARS) by North American Rescue
Domain Est. 2002
#5 NAR ARS for Needle Decompression
Domain Est. 2006
Website: rescue-essentials.com
Key Highlights: In stock Rating 5.0 (10) This needle is a great tool for advanced management of sucking chest wounds and is a small rigid packages….
#6 NIK Kit
Domain Est. 2014
Website: medsourcelabs.com
Key Highlights: Introducing the Needle Decompression Kit. MedSource Labs, in partnership with Pneumeric, is proud to announce the launch of the Needle Decompression ……
#7 ARS Needle decompression
Domain Est. 2017
Expert Sourcing Insights for Needle Decompression Kit
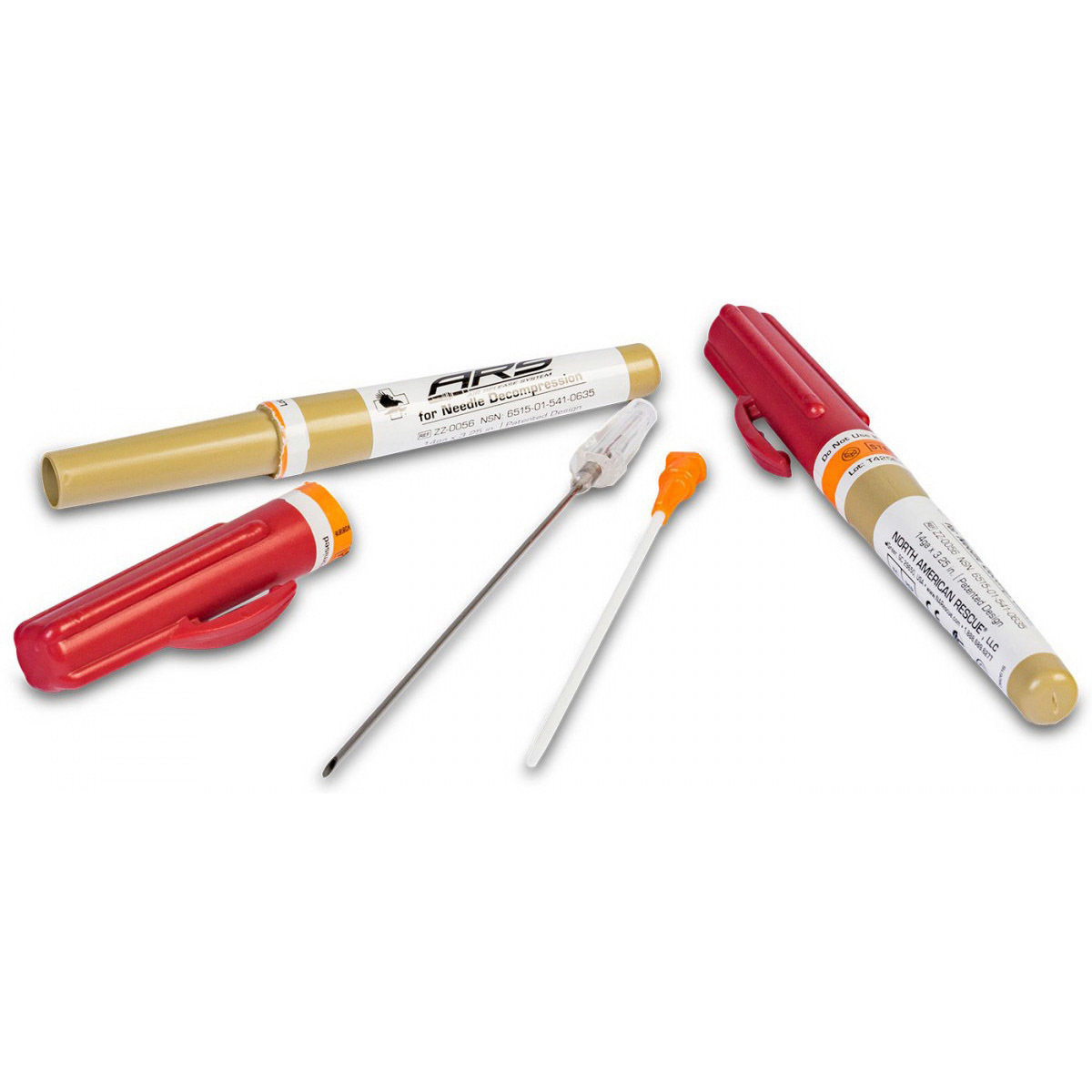
H2: 2026 Market Trends for Needle Decompression Kits
The global market for needle decompression kits is projected to experience steady growth by 2026, driven by increasing demand for emergency medical interventions, rising incidence of traumatic injuries, and advancements in pre-hospital care systems. As a critical tool in the management of tension pneumothorax—a life-threatening condition often resulting from trauma—needle decompression kits are becoming more integrated into both civilian and military emergency response protocols.
One of the key trends shaping the 2026 market is the expansion of emergency medical services (EMS) infrastructure, particularly in emerging economies. Countries in Asia-Pacific, Latin America, and Africa are investing heavily in ambulance services and first responder training, which is directly increasing the adoption of essential emergency tools like needle decompression kits. Governments and healthcare organizations are also implementing standardized trauma care guidelines, further solidifying the role of these kits in emergency treatment algorithms.
Technological innovation is another significant driver. Manufacturers are focusing on improving device safety, ease of use, and reliability. By 2026, the market is expected to see a rise in kits featuring integrated safety mechanisms, color-coded components, and pre-packaged, single-use designs that reduce error during high-stress situations. Some next-generation kits are incorporating ultrasound guidance compatibility or smart sensors, although these remain in early adoption phases.
The military and defense sector continues to be a major end-user, with armed forces modernizing their individual first aid kits (IFAKs). Needle decompression is a standard procedure in combat casualty care, and ongoing geopolitical tensions are accelerating procurement. Additionally, civilian applications in law enforcement, wilderness medicine, and industrial emergency response are expanding the user base.
Regulatory developments are also influencing market dynamics. Increased scrutiny from agencies like the FDA and EMA is pushing manufacturers to ensure higher compliance with sterilization, labeling, and post-market surveillance requirements. This is likely to consolidate the market, favoring established players with robust quality systems.
Furthermore, the impact of the COVID-19 pandemic has indirectly boosted awareness of respiratory emergencies, leading to broader training in airway management. While needle decompression is not used in typical respiratory infections, the heightened focus on emergency preparedness has led to increased stockpiling and training—including for trauma-related procedures—supporting market growth.
In summary, the 2026 needle decompression kit market is characterized by geographic expansion, technological enhancement, regulatory maturation, and growing integration into emergency care ecosystems. These trends suggest sustained demand and innovation, positioning the market for moderate but meaningful growth in the coming years.
Common Pitfalls Sourcing Needle Decompression Kits (Quality, IP)
Sourcing Needle Decompression Kits (NDKs) requires careful attention to both quality standards and intellectual property (IP) considerations. Overlooking these aspects can lead to serious risks, including compromised patient safety, legal liabilities, and supply chain disruptions.
Poor Manufacturing Quality and Non-Compliance
One of the most critical pitfalls involves procuring kits that fail to meet medical device quality standards. Low-cost suppliers may cut corners on materials, sterilization processes, or manufacturing controls. This can result in devices with inconsistent needle sharpness, compromised catheter integrity, or inadequate packaging—posing direct risks during emergency use. Additionally, non-compliant kits may lack necessary regulatory approvals (e.g., FDA 510(k), CE Marking), making their use legally and ethically problematic.
Inadequate Verification of Regulatory Certification
Buyers often assume that product listings or supplier claims are accurate without independently verifying certifications. Some suppliers may present forged or outdated documentation, or sell kits under misleading regulatory classifications. Relying solely on supplier-provided certificates without cross-checking with regulatory databases increases the risk of acquiring non-compliant devices, which can lead to product recalls, usage bans, or liability in case of adverse events.
Intellectual Property Infringement Risks
Many established NDK designs are protected by patents, trademarks, or design rights. Sourcing unbranded or “compatible” kits from manufacturers that replicate patented features—such as specific catheter over-the-needle mechanisms or packaging configurations—can expose buyers to IP infringement claims. Even if the supplier is overseas, international trade laws and enforcement mechanisms (e.g., customs seizures, litigation in the buyer’s jurisdiction) may still apply, leading to financial penalties and reputational damage.
Lack of Traceability and Supply Chain Transparency
Sourcing from opaque supply chains increases vulnerability to counterfeit products and inconsistent quality. Without clear traceability—from raw materials to final assembly—it is difficult to verify manufacturing conditions, ensure batch consistency, or conduct effective recalls if needed. This lack of transparency complicates compliance with quality management systems like ISO 13485 and undermines confidence in the reliability of the medical devices being deployed.
Insufficient Evaluation of Supplier Reliability
Procurement decisions based solely on price or speed, without assessing the supplier’s track record, quality management systems, or after-sales support, can result in long-term operational issues. Unreliable suppliers may deliver delayed, incomplete, or substandard batches, jeopardizing emergency preparedness and eroding trust in the supply chain. Establishing long-term partnerships with vetted, reputable manufacturers is essential for consistent quality and regulatory adherence.

Logistics & Compliance Guide for Needle Decompression Kit
Overview and Intended Use
The Needle Decompression Kit is a medical device designed for emergency treatment of tension pneumothorax, a life-threatening condition caused by trapped air in the pleural space. The kit typically includes a large-bore catheter-over-needle (commonly 10–14 gauge, 8–10 cm in length), sterile packaging, and may include antiseptic wipes, gloves, and instructions for use. It is intended for use by trained healthcare providers or first responders in pre-hospital or clinical emergencies.
Regulatory Classification
The Needle Decompression Kit is classified as a medical device under regulatory authorities such as the U.S. Food and Drug Administration (FDA), European Medicines Agency (EMA), and other national health regulatory bodies. In the United States, it is generally classified as a Class II medical device under 21 CFR 878.4490 (Thoracic drainage systems) and requires 510(k) premarket notification. In the European Union, it must meet requirements under the Medical Device Regulation (MDR) (EU) 2017/745 and carry the CE mark.
Manufacturing and Quality Standards
Manufacturers must comply with Quality Management Systems (QMS) such as ISO 13485:2016, which outlines requirements for design, production, packaging, labeling, storage, and distribution of medical devices. Sterility assurance must follow ISO 11135 (ethylene oxide sterilization) or ISO 11137 (radiation sterilization), and packaging must comply with ISO 11607 for sterile barrier systems.
Labeling and Packaging Requirements
All kits must include clear labeling in compliance with FDA 21 CFR Part 801 and EU MDR Annex I. Labels must feature:
– Device name and intended use
– Manufacturer name and address
– Unique Device Identifier (UDI)
– Sterility status and expiration date
– Single-use designation
– Latex or other allergen warnings, if applicable
– Instructions for Use (IFU) in the local language(s)
Packaging must maintain sterility during transport and storage, with tamper-evident seals and environmental protection against moisture, heat, and physical damage.
Storage and Transportation
- Temperature: Store at controlled room temperature (15°C to 30°C / 59°F to 86°F), unless otherwise specified by the manufacturer.
- Humidity: Avoid high humidity environments to prevent packaging degradation.
- Transportation: Ship in secure, temperature-monitored containers when necessary. Ensure compliance with IATA/ADR regulations if transported by air or road.
- Shelf Life: Typically 3–5 years; monitor expiration dates and rotate stock using First-Expired, First-Out (FEFO) principles.
Import and Export Compliance
When shipping internationally, ensure compliance with:
– FDA Export Certificate (for U.S.-based exporters)
– Certificates of Free Sale (CFS) from the country of origin
– Customs Documentation: Proper HS Code classification (e.g., 9018.90 for other medical instruments)
– Import Permits: Required in certain countries (e.g., Health Canada, TGA in Australia, MOH in Saudi Arabia)
– Dual-Use Concerns: Verify no military application restrictions apply
Distribution and Supply Chain Controls
Distributors must be authorized and registered with relevant regulatory bodies (e.g., FDA registration for U.S. distributors). Maintain traceability through UDI and implement a robust distribution record system to support recalls if necessary. Temperature-controlled logistics (cold chain, if specified) and real-time tracking are recommended for high-risk environments.
Training and Clinical Use
Only trained personnel should use the Needle Decompression Kit. Training programs must cover:
– Indications and contraindications
– Proper insertion technique (typically second intercostal space, midclavicular line)
– Complication recognition (e.g., lung laceration, infection)
– Post-procedure monitoring
Documentation of training and competency is required in clinical and pre-hospital settings.
Post-Market Surveillance and Adverse Event Reporting
Manufacturers and distributors must establish post-market surveillance systems per FDA 21 CFR Part 803 (MDR reporting) and EU MDR Article 83–86. Report all adverse events, device malfunctions, or suspected counterfeit products to relevant authorities. Conduct periodic safety update reports (PSURs) and field safety corrective actions (FSCAs), if needed.
Disposal and Environmental Compliance
After single use, kits must be disposed of as biohazardous medical waste in accordance with local regulations (e.g., OSHA Bloodborne Pathogens Standard in the U.S., EU Directive 2000/59/EC). Needles must be placed in puncture-resistant sharps containers. Do not reprocess or reuse.
Summary of Key Compliance Requirements
| Region | Regulatory Body | Key Regulation | Certification Needed |
|——–|——————|—————-|————————|
| United States | FDA | 21 CFR 801, 803, 878 | 510(k), UDI, Registration |
| European Union | EMA / Notified Body | MDR (EU) 2017/745 | CE Mark, UDI, Technical File |
| Canada | Health Canada | Medical Devices Regulations (SOR/98-282) | Medical Device License (MDL) |
| Australia | TGA | Therapeutic Goods Act 1989 | ARTG Inclusion |
Adherence to this guide ensures safe, legal, and effective distribution and use of Needle Decompression Kits worldwide.
Conclusion: Sourcing a Needle Decompression Kit
In conclusion, sourcing a needle decompression kit requires careful consideration of several key factors, including regulatory compliance (such as FDA or CE marking), needle specifications (typically 14-gauge, 5 cm length), sterility, packaging, and ease of access in emergency situations. The kit must be reliable, portable, and suitable for rapid deployment in pre-hospital or tactical environments where tension pneumothorax is a risk. Evaluating suppliers based on product quality, consistency, certifications, and reputation is essential to ensure patient safety and clinical effectiveness. Ultimately, selecting the right needle decompression kit involves balancing cost-efficiency with clinical performance to support timely, life-saving interventions in critical care and emergency medical services.