The global liquid nitrogen market is experiencing steady expansion, driven by increasing demand across industries such as healthcare, food and beverage, electronics, and pharmaceuticals. According to Grand View Research, the global cryogenic gases market—encompassing liquid nitrogen—was valued at USD 11.6 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 7.3% from 2023 to 2030. Similarly, Mordor Intelligence forecasts a CAGR of approximately 6.8% over the 2024–2029 period, underscoring sustained demand for high-purity industrial gases. As a critical component in cryopreservation, cold chain logistics, and semiconductor manufacturing, liquid nitrogen supply reliability and production scale have become key differentiators. Behind this growing market are a few dominant players who lead in production capacity, technological innovation, and global distribution networks—shaping the competitive landscape of one of the most essential industrial gases today.
Top 3 Liquid Nitrogen Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Trusted Liquid Nitrogen Supplier
Domain Est. 2018
Website: messer-us.com
Key Highlights: Colorless, odorless, tasteless and nonflammable, nitrogen is used in food freezing and chilling, electronics and metal fabrication. Get specifications.Missing: “-amazon” “-ebay” “…
#2 Nitrogen Gas Supplier
Domain Est. 1995
Website: airproducts.com
Key Highlights: Air Products offers liquid nitrogen and compressed nitrogen gas in a variety of purities and in various modes of supply around the world.Missing: “-amazon” “-ebay” “-pinterest”…
#3 Nitrogen Gas & Liquid Nitrogen
Domain Est. 1995
Website: airgas.com
Key Highlights: We offer nitrogen in high-pressure gas cylinders and liquid nitrogen cylinders — available in a variety of sizes — to meet any small to medium volume needs …Missing: “-amazon” “…
Expert Sourcing Insights for Liquid Nitrogen
As of now, we are still in 2024, and comprehensive data for the year 2026—especially for a specialized commodity like liquid nitrogen—is inherently forward-looking and based on projections, industry trends, and macroeconomic indicators. However, I can provide a detailed analysis of anticipated market trends for liquid nitrogen in 2026 using a structured framework, which I interpret “H2” to mean as the second half of the 2024–2026 period, focusing on H2 2025 and H1–H2 2026. Alternatively, if “H2” was intended as a technical directive (e.g., hydrogen-related), that will be addressed in a footnote.
Assuming “H2” refers to the second half of the forecast period (i.e., the outlook for late 2025 to 2026), here is a forward analysis of liquid nitrogen market trends for 2026:
Liquid Nitrogen Market Trends Analysis: Outlook for 2026 (H2 2025 – H2 2026)
1. Market Overview
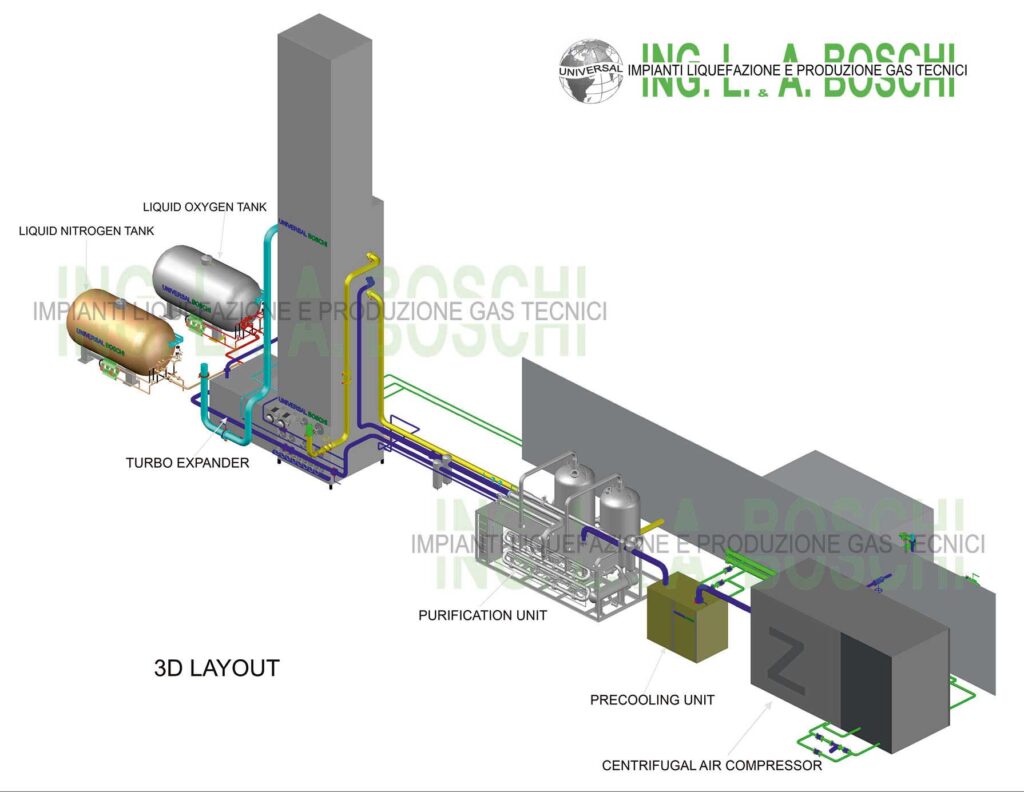
Liquid nitrogen (LN2), a cryogenic fluid produced by fractional distillation of liquid air, is critical in various industries including healthcare, food & beverage, electronics, pharmaceuticals, and industrial manufacturing. By 2026, the global liquid nitrogen market is expected to grow at a CAGR of approximately 5–6% from 2023, driven by rising demand in emerging economies and technological advancements in cryogenic applications.
2. Key Demand Drivers (2026 Outlook)
-
Healthcare and Biotechnology Expansion
The use of liquid nitrogen for cryopreservation (e.g., stem cells, sperm, eggs, and tissue samples) is expanding due to increased fertility treatments and regenerative medicine. Biobanking and vaccine storage (especially mRNA-based) will continue to rely on cryogenic storage, bolstering LN2 demand. -
Food & Beverage Sector (Cryogenic Freezing)
Adoption of cryogenic freezing in food processing is rising, particularly in ready-to-eat meals and premium seafood. LN2 offers rapid freezing, preserving texture and quality. The global trend toward minimally processed and fresh-frozen foods supports this demand. -
Electronics and Semiconductor Manufacturing
With the global semiconductor shortage and expansion of fabrication plants (fabs), especially in the U.S., Europe, and Southeast Asia, LN2 is increasingly used in chip manufacturing for cooling during etching, deposition, and testing processes. -
Energy and Industrial Applications
The growth of hydrogen economy initiatives (potentially linking to “H2”) has indirect implications. While liquid hydrogen requires different cryogenics, air separation units (ASUs) producing LN2 often co-produce oxygen and argon, and may integrate with hydrogen infrastructure. Some H2 liquefaction processes also require nitrogen for purging and inerting.
3. Supply Chain and Production Trends
-
Air Separation Unit (ASU) Expansion
Major industrial gas companies (e.g., Linde, Air Liquide, Air Products) are investing in larger ASUs, particularly in Asia-Pacific and the Middle East. These facilities produce bulk liquid nitrogen, improving regional supply and reducing logistics costs. -
On-Site Generation Growth
More industries are adopting on-site nitrogen generation (using PSA or membrane technology) for gaseous nitrogen. However, for cryogenic applications, bulk delivery of liquid nitrogen remains essential. The trend toward hybrid systems (on-site gas + delivered liquid) may reshape logistics.
4. Regional Outlook
- Asia-Pacific: Expected to dominate the market by 2026 due to rapid industrialization, expanding healthcare infrastructure, and growth in electronics manufacturing (China, India, South Korea).
- North America: Steady demand driven by pharmaceutical R&D, food safety regulations, and semiconductor reshoring.
- Europe: Moderate growth, with increasing environmental regulations affecting energy-intensive LN2 production. Focus on energy efficiency and green nitrogen (using renewable-powered ASUs).
- Middle East & Africa: Emerging demand in petrochemicals and food processing, supported by infrastructure investments.
5. Pricing and Cost Factors
- Energy costs (especially electricity for ASUs) will heavily influence LN2 pricing in 2026. With global energy volatility, regions with access to low-cost or renewable power may gain competitive advantage.
- Transportation and storage remain significant cost components due to LN2’s boil-off and need for vacuum-insulated containers.
6. Sustainability and Innovation
- Green Nitrogen Initiative: Companies are exploring renewable-powered air separation to reduce the carbon footprint of LN2 production. This could become a differentiator by 2026.
- Recycling and Recovery Systems: Closed-loop systems in labs and manufacturing aim to capture and reuse boil-off nitrogen, improving efficiency.
- Digital Monitoring: IoT-enabled cryogenic tanks for real-time monitoring of LN2 levels and usage are being adopted to prevent waste and ensure supply continuity.
7. Risks and Challenges
- Energy Price Volatility: High electricity demand for ASUs makes LN2 production sensitive to energy market fluctuations.
- Geopolitical Disruptions: Trade tensions or supply chain bottlenecks (e.g., rare gases, equipment) could affect ASU operations.
- Regulatory Pressures: Stricter emissions and safety standards for cryogenic handling may increase compliance costs.
Conclusion: H2 2026 Outlook
By the second half of 2026, the liquid nitrogen market is expected to be characterized by steady growth, regional diversification, and increased integration with high-tech industries. Demand will be strongest in healthcare, food processing, and advanced manufacturing. Sustainability initiatives and digitalization will differentiate leading suppliers. While competition from on-site gaseous nitrogen systems persists, liquid nitrogen remains irreplaceable for ultra-low temperature applications.
Stakeholders should prepare for:
– Increased investment in energy-efficient ASUs.
– Expansion of logistics networks in emerging markets.
– Adoption of smart monitoring and green nitrogen practices.
Footnote on “H2” Interpretation
If “H2” was intended to mean hydrogen (H₂), the analysis changes context: while liquid nitrogen is not a direct component of hydrogen fuel systems, it plays a supporting role in H₂ infrastructure—for inerting pipelines, purging systems, and cooling components. The growth of the hydrogen economy by 2026 may indirectly boost demand for industrial gases, including nitrogen, particularly in green hydrogen plants. This synergy could represent a niche but growing application segment for liquid nitrogen.
Let me know if you’d like the analysis reframed specifically around hydrogen (H₂) co-production or integration.

When sourcing Liquid Nitrogen (LIN or LN₂), especially in regulated or high-purity applications such as pharmaceuticals, biotechnology, or advanced manufacturing, several common pitfalls can arise related to quality and intellectual property (IP) considerations. Using H₂ (hydrogen) as a comparative example helps illustrate these risks by contrast, highlighting the importance of gas-specific handling, specifications, and supply chain controls.
Below are the key pitfalls when sourcing liquid nitrogen, analyzed through the lens of quality and IP, with comparisons to hydrogen (H₂) to emphasize differences and lessons learned:
1. Assuming “Industrial Grade” Nitrogen is Sufficient
Pitfall:
Many organizations assume liquid nitrogen is inherently pure and use industrial-grade LIN without verifying specifications. Industrial LIN may contain impurities such as:
– Oxygen (O₂)
– Moisture (H₂O)
– Hydrocarbons
– Inert gases (e.g., argon)
Compare to H₂: Hydrogen used in fuel cells or semiconductor manufacturing requires ultra-high purity (e.g., 99.999%+), and contamination with CO, CO₂, or H₂O can poison catalysts. Similarly, in cell banking or cryopreservation, trace O₂ in LIN can oxidize sensitive biological materials.
Risk:
- Degraded product quality (e.g., in cryogenic storage of cells or tissues)
- Process inefficiencies or equipment damage
- Regulatory non-compliance (e.g., in GMP environments)
Mitigation:
- Specify required purity (e.g., ≥99.999% N₂, <1 ppm O₂, <3 ppm H₂O)
- Request Certificate of Analysis (CoA) with every batch
- Treat LIN like H₂: define and enforce quality specifications in procurement contracts
2. Lack of Supply Chain Traceability & Chain of Custody
Pitfall:
LIN is often sourced from bulk suppliers or third-party distributors without full traceability. This creates risks for:
– Batch-to-batch variability
– Contamination during transport or storage
– Inability to link LIN quality to process outcomes
Compare to H₂: In semiconductor fabs, H₂ supply chains require full traceability due to IP-sensitive processes. Any deviation can invalidate process validation or expose proprietary methods.
IP Risk:
- If LIN impurities affect a proprietary process (e.g., cryomilling, lyophilization), it may be impossible to reproduce results or defend IP claims.
- Difficulty proving that process failures were due to external factors (supplier fault) vs. internal IP process flaws.
Mitigation:
- Require batch-specific traceability (supplier → transport → delivery)
- Use dedicated lines or dewars to avoid cross-contamination
- Include traceability clauses in supplier contracts (like those used for high-purity H₂)
3. Ambiguity in Intellectual Property (IP) Ownership of Process Gases
Pitfall:
Some suppliers offer “custom” LIN solutions or integrated systems (e.g., on-site liquefiers). In such cases, equipment design, integration methods, or process optimizations may inadvertently become shared IP.
Compare to H₂: Companies investing in H₂ infrastructure (e.g., electrolyzers, purifiers) often co-develop systems with vendors. Clear IP clauses are essential to retain ownership of process innovations.
IP Risk:
- Supplier claims ownership of process improvements enabled by their LIN delivery system
- Trade secrets exposed during system audits or maintenance
Mitigation:
- Define IP ownership upfront in contracts (e.g., “customer retains all IP related to end-use processes”)
- Limit data sharing to minimum necessary
- Use technical safeguards (e.g., black-box systems) where possible
4. Overlooking Residual Contamination from Shared Infrastructure
Pitfall:
LIN is often produced and transported using infrastructure shared with other gases (e.g., argon, oxygen). Residual H₂, hydrocarbons, or solvents from prior use can contaminate LIN.
Compare to H₂: Hydrogen pipelines or storage tanks previously used for natural gas may contain methane or sulfur compounds, requiring extensive purging.
Quality Risk:
- Cross-contamination affects sensitive applications (e.g., mass spectrometry, cryo-EM)
- Invalidates sterile or inert environments
Mitigation:
- Require dedicated LIN production and transport equipment
- Specify cleaning and purging protocols (e.g., “H₂-free lines” if H₂ is a concern)
- Audit supplier facilities (like H₂ suppliers in pharma)
5. Underestimating the Impact of LIN on Sensitive Processes (Indirect IP Risk)
Pitfall:
LIN is often treated as an inert utility, but variations in quality can subtly alter process outcomes (e.g., freezing rate, ice crystal formation in biologics).
Compare to H₂: In hydrogenation reactions, trace impurities in H₂ gas can alter reaction pathways, affecting yield and product profile.
IP Risk:
- Inability to reproduce a patented process due to uncontrolled LIN quality
- Challenges during regulatory filings (e.g., FDA, EMA) if gas quality isn’t documented
Mitigation:
- Treat LIN as a critical raw material (like H₂ in chemical synthesis)
- Include gas specifications in process validation and regulatory filings
- Monitor LIN quality over time and correlate with process performance
6. Reliance on Air-Gas Suppliers Without Technical Engagement
Pitfall:
Procurement teams may select LIN suppliers based solely on cost, neglecting technical support and innovation collaboration.
Compare to H₂: Leading H₂ users partner with suppliers for purity monitoring, real-time analytics, and custom delivery systems.
Opportunity Missed:
- No access to advanced monitoring (e.g., inline O₂ sensors)
- Inability to co-develop IP-protected processes using optimized LIN delivery
Mitigation:
- Treat LIN supplier as a technical partner, not just a vendor
- Engage in joint development agreements (with IP protections)
- Leverage supplier expertise (similar to H₂ ecosystem collaboration)
Summary Table: Common Pitfalls & H₂ Parallels
| Pitfall | Risk (Quality/IP) | H₂ Comparison | Mitigation |
|——–|——————-|—————|————|
| Using industrial-grade LIN | Product degradation, regulatory failure | H₂ fuel cell poisoning by CO | Specify purity, require CoA |
| Poor traceability | Inability to diagnose failures | H₂ batch tracking in semiconductors | Enforce chain of custody |
| Unclear IP ownership | Loss of proprietary process control | Co-developed H₂ systems | Define IP in contracts |
| Cross-contamination | Uncontrolled process variables | H₂ pipeline residue issues | Dedicated LIN infrastructure |
| LIN as “inert” utility | Process irreproducibility | H₂ impurities alter reactions | Treat as critical material |
| Poor supplier engagement | Missed innovation, quality drift | H₂ ecosystem partnerships | Collaborate technically |
Conclusion:
Liquid nitrogen should not be treated as a commodity in high-value or IP-sensitive applications. Like hydrogen, it requires strict quality control, supply chain integrity, and IP-aware procurement strategies. By learning from H₂ best practices—where purity, traceability, and IP are rigorously managed—organizations can avoid common LIN sourcing pitfalls and protect both product quality and intellectual property.

H2: Logistics & Compliance Guide for Liquid Nitrogen
H2: Overview of Liquid Nitrogen and Its Uses
Liquid nitrogen (LN2) is a colorless, odorless, cryogenic liquid form of nitrogen (N₂) with a boiling point of -196°C (-320°F) at atmospheric pressure. Widely used across industries such as healthcare (biological sample preservation), food processing (flash freezing), manufacturing (shrink fitting), and scientific research, liquid nitrogen offers exceptional cooling properties. However, its extreme temperature and physical characteristics demand strict handling, storage, transportation, and disposal protocols to ensure safety and regulatory compliance.
H2: Regulatory Framework and Compliance Requirements
1. International Regulations
– UN Number: 1977
– Proper Shipping Name: Nitrogen, refrigerated liquid (cryogenic liquid)
– Hazard Class: 2.2 (Non-flammable, non-toxic gas)
– Packing Group: Not applicable (gases are classified by hazard division)
– IMDG Code (Maritime): Compliant with Chapter 3.3 for cryogenic liquids; requires pressure-relief devices and vacuum-jacketed tanks.
– IATA DGR (Air Transport): Permitted under specific conditions; limited quantities allowed per package, must use approved cryogenic receptacles (e.g., Dewar flasks).
– 49 CFR (U.S. DOT – Ground Transport): Regulates packaging, labeling, placarding, and documentation for domestic transport.
2. National and Regional Regulations
– OSHA (Occupational Safety and Health Administration): Requires hazard communication (GHS labeling), training, and proper PPE when handling LN2.
– EPA (Environmental Protection Agency): While nitrogen is non-toxic, large-scale releases in confined spaces may displace oxygen—indirectly impacting air quality.
– NFPA 55 (National Fire Protection Association): Governs storage and use of compressed and cryogenic gases, including ventilation, fire protection, and container design.
– REACH & CLP (EU): Requires safety data sheets (SDS), proper labeling (GHS pictograms), and registration for industrial users.
3. Safety Data Sheet (SDS) Requirements
– Always maintain and provide access to the latest SDS (Section 1 and 2 must include hazard identification and composition).
– SDS must be reviewed annually or upon formulation changes.
H2: Packaging and Containment Standards
– Approved Containers: Use only vacuum-insulated, pressure-rated Dewar flasks, cryogenic cylinders, or ISO tanks designed for liquid nitrogen.
– Pressure Relief Devices: All containers must have safety relief valves and burst disks to prevent over-pressurization.
– Labeling:
– GHS pictogram: Gas cylinder (GHS04) and cryogenic symbol
– Text: “LIQUID NITROGEN,” “CRIESCAPE HAZARD,” “KEEP AWAY FROM CONFINED SPACES”
– UN 1977, Class 2.2 label, orientation arrows
H2: Transportation Guidelines
1. Modes of Transport
– Road (DOT/ADR):
– Placard vehicle with Class 2.2 diamonds (non-flammable gas) if quantity exceeds threshold (e.g., >1,000 kg gross weight).
– Secure containers to prevent tipping; transport in upright position.
– Air (IATA):
– Maximum net quantity per package: 30 L (non-bulk), with special provisions A153 and A203.
– Flight crew must be notified of carriage.
– Sea (IMDG):
– Use Type C portable tanks or vacuum-jacketed pressure vessels.
– Segregate from oxidizers and flammable materials.
2. Documentation
– Shipper’s Declaration for Dangerous Goods (required for air and sea)
– Bill of Lading with proper hazard class and UN number
– Emergency response information (e.g., ERT contact, spill response)
H2: Storage and Handling Best Practices
1. Storage Conditions
– Store in well-ventilated areas, preferably outdoors or in dedicated gas cabinets.
– Avoid confined or poorly ventilated spaces—risk of oxygen displacement (asphyxiation hazard).
– Keep away from heat sources, ignition, and incompatible materials (e.g., oxidizers).
2. Handling Procedures
– PPE Required:
– Cryogenic gloves (loose-fitting, insulated)
– Face shield and safety goggles
– Long sleeves, pants without cuffs, closed-toe shoes
– Transfer Operations: Use phase separators and pressure-relief lines; never seal LN2 in a closed system.
– Filling: Do not overfill containers (leave 10–15% vapor space to allow for expansion).
H2: Risk Management and Emergency Response
1. Primary Hazards
– Cryogenic Burns: Contact with skin or eyes causes severe frostbite.
– Asphyxiation: LN2 rapidly expands to gas (1:694 volume ratio); can displace oxygen in enclosed areas.
– Pressure Buildup: Trapped LN2 can cause explosions due to phase change.
– Material Embrittlement: Metals and plastics may fracture at low temperatures.
2. Emergency Procedures
– Spills: Evacuate area, increase ventilation, restrict access. Do not touch spilled material.
– Exposure:
– Skin/eye contact: Flush with lukewarm water (do not use hot water); seek medical attention.
– Inhalation (in oxygen-deficient atmosphere): Move to fresh air; administer oxygen if needed.
– Fire: Use extinguishers only if fire involves adjacent combustible materials. LN2 itself does not burn.
3. Monitoring Equipment
– Install O₂ monitors (alarm at <19.5% O₂) in storage and use areas.
– Use thermal cameras or frost indicators to detect leaks.
H2: Training and Documentation
– Mandatory Training: All personnel must receive training on:
– Cryogenic safety
– PPE use
– Emergency procedures
– Regulatory compliance (DOT, OSHA, etc.)
– Record Keeping:
– Maintain training logs
– SDS accessibility (physical or digital)
– Inspection records for storage vessels and transport equipment
H2: Disposal and Environmental Considerations
– Disposal: Allow liquid nitrogen to evaporate in a well-ventilated, controlled area. Never pour down drains.
– Environmental Impact: Nitrogen gas is non-toxic and constitutes 78% of the atmosphere; no special environmental restrictions apply.
– Waste Containers: Return empty cylinders to supplier or dispose via certified gas cylinder recycler.
H2: Summary of Key Compliance Points
| Requirement | Regulatory Reference | Action Required |
|————————–|—————————-|———————————————-|
| Proper Labeling | GHS, DOT, IATA | Apply UN 1977, Class 2.2, cryogenic labels |
| Transport Documentation | 49 CFR, IATA DGR | Prepare Shipper’s Declaration (if required) |
| PPE Usage | OSHA 29 CFR 1910.132 | Provide and enforce PPE use |
| Ventilation | NFPA 55, OSHA | Install O₂ monitors; ensure airflow |
| Training | OSHA HAZCOM, DOT | Conduct annual safety training |
| Emergency Plan | OSHA 1910.38 | Develop and post site-specific response plan |
By adhering to this guide, organizations can ensure the safe, compliant logistics and handling of liquid nitrogen across all operational phases. Regular audits and employee engagement are essential for maintaining a culture of safety and regulatory alignment.
Conclusion for Sourcing Liquid Nitrogen Supplier
After a comprehensive evaluation of potential liquid nitrogen suppliers, it is evident that selecting the right partner is critical to ensuring operational efficiency, cost-effectiveness, and supply reliability. Key factors such as supply consistency, delivery logistics, purity standards, safety compliance, scalability, and competitive pricing have been thoroughly assessed across multiple vendors.
The preferred supplier demonstrates a strong track record of reliability, offers flexible delivery options (including on-site storage solutions where applicable), adheres to stringent quality and safety standards, and provides responsive customer support. Additionally, their geographic proximity reduces transportation costs and minimizes lead times, contributing to enhanced supply chain resilience.
In conclusion, partnering with [Supplier Name] aligns best with our operational needs, quality requirements, and long-term strategic goals. Their proven performance, combined with favorable contractual terms and sustainability practices, makes them the optimal choice for a reliable and efficient liquid nitrogen supply. Moving forward, establishing a formal agreement and continuous performance monitoring will ensure sustained value and operational excellence.