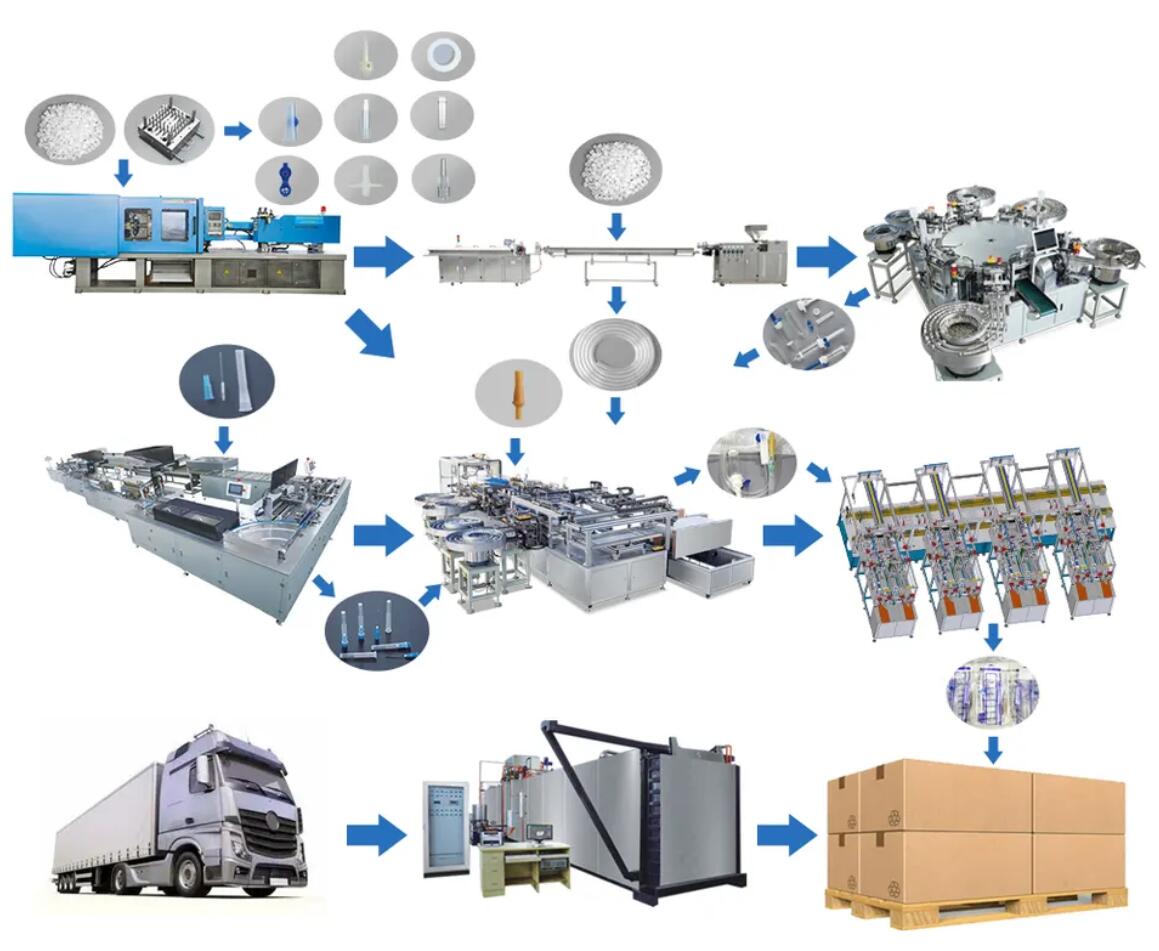
The global intravenous (IV) fluids market is experiencing steady expansion, driven by rising hospital admissions, increasing demand for surgical procedures, and growing prevalence of chronic diseases requiring fluid therapy. According to Mordor Intelligence, the IV fluids market was valued at approximately USD 11.5 billion in 2023 and is projected to grow at a compound annual growth rate (CAGR) of 5.8% from 2024 to 2029. This growth is further fueled by heightened healthcare infrastructure development in emerging economies and increased focus on hydration management in clinical settings. As demand surges, a select group of manufacturers has emerged as leaders in innovation, scale, and regulatory compliance. Below is a data-driven overview of the top 9 IV fluid manufacturers shaping the global landscape.
Top 9 Iv Fluid Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 IV Solutions
Domain Est. 1992
Website: ushospitalproducts.baxter.com
Key Highlights: We offer a broad portfolio of IV solutions and irrigation products, and we’re investing in new technology to help clinicians optimize fluid administration….
#2 About Us
Domain Est. 2022
Website: assureiv.com
Key Highlights: At Assure Infusions, we’re committed to addressing the critical shortage of sterile IV fluids that has plagued healthcare systems for over a decade….
#3 BD IV fluids
Domain Est. 1990
Website: bd.com
Key Highlights: BD ® offers a family of IV fluids to suit customer needs. All IV fluids use the freeflex ® bag, a multilayer polyolefin film that is non-PVC and non-DEHP ……
#4 IV Fluids and Irrigation Solutions
Domain Est. 1997
Website: bbraunusa.com
Key Highlights: Our comprehensive portfolio of IV therapy products, solutions and services can help ensure operational efficiencies, supply reliability and safety….
#5 ICU Medical
Domain Est. 1997
Website: icumed.com
Key Highlights: From IV lines to vital signs, we’re with you at the heart of care. · Products. Infusion Therapy · Products. Pharmacy Preparation · Products. Airway Management….
#6 Pharma
Domain Est. 1998
Website: fresenius-kabi.com
Key Highlights: Enhance patient care with Fresenius Kabi’s comprehensive, high-quality IV therapy products for anesthetics, oncology, anti-infectives, and critical care….
#7 Otsukas IV solutions spreading worldwide
Domain Est. 2001
Website: otsukakj.jp
Key Highlights: The Otsuka Group began selling IV products outside of Japan in the 1970s. Currently, we are involved in the IV business at 17 group companies including OPF….
#8 AbbVie
Domain Est. 2012
Website: abbvie.com
Key Highlights: AbbVie pharmaceuticals combines advanced science with expertise to make strides in drug and treatment discovery, making a remarkable impact on people’s ……
#9 Grifols
Website: grifols.com
Key Highlights: Grifols is a leading global healthcare company. Our trusted and innovative plasma-derived medicines, other biopharmaceuticals and solutions in transfusion ……
Expert Sourcing Insights for Iv Fluid

H2: Projected Market Trends for IV Fluids in 2026
The global intravenous (IV) fluids market is expected to witness steady growth by 2026, driven by increasing demand across healthcare settings, evolving clinical practices, and advancements in fluid therapy solutions. Below are the key market trends anticipated to shape the IV fluids landscape in 2026:
-
Rising Demand in Emerging Markets
Developing regions such as Asia-Pacific, Latin America, and parts of Africa are projected to lead market expansion due to improving healthcare infrastructure, growing geriatric populations, and increased hospital admissions. Countries like India, China, and Brazil will see heightened consumption of IV fluids, particularly in emergency and surgical care. -
Shift Toward Balanced Electrolyte Solutions
There is a growing clinical preference for balanced IV fluids (e.g., Lactated Ringer’s, Plasma-Lyte) over traditional saline (0.9% NaCl), driven by evidence linking high-chloride fluids to adverse outcomes such as acute kidney injury and metabolic acidosis. By 2026, balanced solutions are expected to capture a larger market share, especially in critical care units. -
Increased Focus on Patient Safety and Regulatory Compliance
Regulatory bodies such as the FDA and EMA are placing greater emphasis on the safety, quality, and labeling of IV fluids. Manufacturers will need to comply with stricter standards for sterility, container integrity, and biocompatibility. This trend will accelerate innovation in packaging, such as ready-to-use pre-filled bags with reduced risk of contamination. -
Growth in Home Healthcare and Ambulatory Infusion
The expansion of home-based and outpatient infusion therapies will drive demand for portable, single-dose IV fluid systems. Technological improvements in infusion pumps and wearable delivery devices will support this trend, enabling safe administration of IV fluids outside traditional hospital settings. -
Supply Chain Resilience and Local Manufacturing
Lessons from global supply disruptions during recent health crises (e.g., the pandemic and regional conflicts) will prompt governments and healthcare systems to invest in localized production of essential IV fluids. By 2026, regional manufacturing hubs are expected to reduce dependency on imports and improve supply reliability. -
Sustainability and Eco-Friendly Packaging
Environmental concerns will push manufacturers to adopt greener practices, such as using recyclable materials, reducing plastic waste, and developing biodegradable IV bags. Companies investing in sustainable packaging solutions are likely to gain a competitive advantage in environmentally conscious markets. -
Technological Integration and Smart IV Systems
The integration of digital health technologies—such as IoT-enabled infusion pumps and smart monitoring systems—will enhance precision in fluid delivery and reduce human error. These innovations will be particularly valuable in intensive care and pediatric settings, supporting market growth for advanced IV therapy platforms. -
Impact of Chronic Disease Prevalence
Rising incidence of chronic conditions such as diabetes, cancer, and kidney disease will increase the need for hydration therapy, electrolyte management, and perioperative fluid support. This will sustain long-term demand for both standard and specialized IV formulations.
In conclusion, the IV fluids market in 2026 will be shaped by clinical innovation, regulatory evolution, and shifting healthcare delivery models. Stakeholders who adapt to these trends—by offering safer, more effective, and sustainable products—will be well-positioned to thrive in an increasingly competitive and dynamic market environment.
Common Pitfalls in Sourcing IV Fluids: Quality and Intellectual Property (IP) Concerns
Sourcing intravenous (IV) fluids involves critical considerations, particularly regarding product quality and intellectual property (IP) compliance. Overlooking these aspects can lead to regulatory violations, patient safety risks, and legal liabilities. Below are the most common pitfalls in these two key areas.
Quality-Related Pitfalls
-
Inadequate Supplier Qualification
Failing to thoroughly vet manufacturers—especially overseas suppliers—can result in sourcing from facilities with poor quality management systems. Red flags include lack of certifications (e.g., ISO 13485, GMP), inconsistent audit records, or limited regulatory oversight. -
Non-Compliance with Pharmacopeial Standards
Sourcing IV fluids that do not meet pharmacopeial requirements (e.g., USP, EP, BP) compromises sterility, pyrogen levels, and formulation accuracy. This increases the risk of contamination, adverse reactions, or treatment failure. -
Poor Sterility Assurance
IV fluids must be terminally sterilized or produced under strict aseptic conditions. Suppliers may cut corners in manufacturing or packaging, leading to microbial contamination or endotoxin presence—posing severe risks to immunocompromised patients. -
Inconsistent Batch-to-Batch Quality
Variability in osmolarity, pH, or electrolyte composition between batches can affect clinical outcomes. This often stems from lax process controls or inadequate quality testing protocols. -
Substandard Packaging and Labeling
Poor packaging (e.g., leaks, compromised seals) or incorrect labeling (missing lot numbers, expired dates, or inaccurate contents) increases the risk of product degradation and medication errors. -
Lack of Traceability and Documentation
Incomplete or falsified documentation (e.g., Certificates of Analysis, batch records) can hinder recalls and regulatory investigations, especially during audits or adverse event tracking.
Intellectual Property (IP)-Related Pitfalls
-
Sourcing from Unauthorized Generic Manufacturers
Using suppliers that produce “generic” IV fluids without proper licensing or patent clearance can lead to IP infringement, especially if the formulation, delivery system, or container technology is protected. -
Failure to Verify IP Ownership and Licensing
Procurement teams may overlook whether the supplier has the legal right to manufacture and distribute the product. This is particularly risky with specialty IV formulations or innovative delivery systems (e.g., closed-loop bags, multi-chamber systems). -
Rebranding or Private Labeling Without IP Clearance
Distributing IV fluids under a private label without ensuring that trademarks, packaging designs, or product names do not infringe on existing IP can result in legal disputes and reputational damage. -
Reverse Engineering and Trade Secret Violations
Some suppliers may replicate branded IV fluid formulations or manufacturing processes without authorization, exposing buyers to liability for contributory infringement or trade secret violations. -
Overlooking Regulatory-Linked IP (e.g., Data Exclusivity)
In some markets, regulatory approval data enjoys exclusivity periods. Sourcing generics before such periods expire—even if the patent has lapsed—can breach data protection laws.
Mitigation Strategies
- Conduct rigorous supplier audits and request full compliance documentation.
- Require third-party testing of incoming IV fluid batches.
- Engage legal counsel to review IP rights, licensing agreements, and regulatory status.
- Establish clear contractual terms with suppliers regarding quality standards and IP indemnification.
- Monitor global regulatory updates and patent landscapes for relevant products.
Avoiding these pitfalls ensures the safe, legal, and effective sourcing of IV fluids, protecting both patients and organizational integrity.

Logistics & Compliance Guide for IV Fluids
Ensuring the safe, efficient, and legally compliant handling of intravenous (IV) fluids throughout the supply chain is critical for patient safety and regulatory adherence. This guide outlines key considerations for logistics and compliance related to IV fluid products.
Regulatory Framework and Classification
IV fluids are classified as medical devices or pharmaceuticals depending on jurisdiction and formulation. In the U.S., most IV solutions are regulated by the FDA as drugs under the Federal Food, Drug, and Cosmetic Act. In the EU, they are typically classified as medicinal products under Directive 2001/83/EC or as medical devices if intended solely for irrigation or dilution. Compliance requires adherence to Good Manufacturing Practices (GMP), labeling standards (e.g., FDA 21 CFR Part 201, EU Falsified Medicines Directive), and proper registration with relevant authorities (e.g., FDA, EMA).
Storage Conditions and Environmental Controls
IV fluids must be stored according to manufacturer specifications to maintain sterility and stability. Typically, conditions include:
- Temperature: Store at controlled room temperature (15–25°C or 59–77°F), unless otherwise specified.
- Humidity: Maintain low to moderate humidity to prevent packaging degradation.
- Light: Protect from direct sunlight and UV exposure; store in original packaging.
- Segregation: Keep separate from non-pharmaceutical goods and hazardous materials.
- Handling: Ensure containers are upright and protected from physical damage.
Cold chain requirements apply only if specified (e.g., certain additives or biologics). Regular monitoring with calibrated thermohygrometers and data loggers is required.
Transportation and Cold Chain Management
Transport of IV fluids must ensure product integrity through:
- Use of validated vehicles or containers with temperature monitoring.
- Real-time tracking and alarm systems for temperature excursions.
- Compliance with GDP (Good Distribution Practices) per WHO and regional guidelines.
- Documentation of transport conditions (e.g., temperature logs, chain of custody).
- Minimizing transit time and avoiding extreme environmental exposure.
For international shipments, adherence to IATA regulations (when air freight is used) and customs documentation (e.g., Certificates of Pharmaceutical Product, Free Sale Certificates) is essential.
Packaging and Labeling Requirements
Packaging must preserve sterility and prevent contamination. Primary packaging (e.g., PVC or non-PVC bags, glass bottles) should be tamper-evident. Labeling must include:
- Product name, strength, and dosage form
- Batch/lot number and expiration date
- Storage conditions
- Manufacturer and distributor information
- Barcode/UDI (Unique Device Identification) where applicable
- Regulatory identifiers (e.g., Rx only, CE mark)
Labels must comply with local language requirements and regulatory standards (e.g., FDA labeling rules, EU Annex I of Directive 2001/83/EC).
Inventory Management and Traceability
Robust inventory systems must support full traceability from manufacturer to end-user. Key practices include:
- Use of serialized lot numbers and barcoding.
- Implementation of ERP or specialized pharmaceutical inventory software.
- First-Expired, First-Out (FEFO) stock rotation.
- Regular audits and reconciliation to prevent expired product use.
- Recall readiness: Ability to quickly identify and retrieve affected batches.
Compliance with U.S. DSCSA (Drug Supply Chain Security Act) or EU FMD serialization and verification requirements is mandatory for distributors and dispensers.
Quality Control and Documentation
All logistics operations must be supported by documented quality procedures, including:
- Standard Operating Procedures (SOPs) for handling, storage, and transport.
- Staff training records on GxP compliance.
- Certificate of Analysis (CoA) and Certificate of Conformance (CoC) for each batch.
- Deviation and incident reporting systems.
- Regular internal audits and external inspections.
Retention of records (typically 5–10 years post-expiration) is required by most regulatory bodies.
Import/Export and Customs Compliance
Cross-border movement of IV fluids requires:
- Valid import/export licenses.
- Compliance with CITES (if applicable), though rare for standard IV fluids.
- Accurate HS codes (e.g., 3004 for medicaments in dosage form).
- Adherence to bilateral or multilateral trade agreements.
- Pre-shipment inspections or certifications, as required by destination country.
Collaboration with certified freight forwarders experienced in pharmaceutical logistics is recommended.
Risk Management and Contingency Planning
Organizations must have plans for:
- Temperature excursions during storage or transport.
- Product recalls or withdrawals.
- Natural disasters or supply chain disruptions.
- Cybersecurity for digital tracking systems.
Risk assessments (e.g., FMEA) should be conducted regularly, and business continuity plans maintained and tested.
Training and Personnel Qualifications
Staff involved in IV fluid logistics must receive regular training on:
- GMP/GDP principles
- Hazardous material handling (if applicable)
- Cold chain management
- Regulatory compliance
- Emergency response procedures
Training records must be maintained, and personnel competency assessed periodically.
Conclusion
Effective logistics and compliance for IV fluids demand an integrated approach combining regulatory knowledge, environmental control, traceability, and quality management. Adherence to international standards and local regulations ensures patient safety, avoids legal penalties, and maintains supply chain integrity. Regular review and continuous improvement of processes are essential in this highly regulated environment.
Conclusion: Sourcing IV Fluid Manufacturers
In conclusion, sourcing IV fluid manufacturers requires a strategic and thorough approach to ensure the selection of reliable, compliant, and high-quality suppliers. Given the critical nature of IV fluids in medical and clinical settings, adherence to strict regulatory standards—such as those set by the FDA, WHO, and ISO—is non-negotiable. Factors such as manufacturing certifications, quality control processes, scalability, supply chain reliability, and cost-efficiency must be carefully evaluated.
Establishing partnerships with manufacturers who demonstrate a proven track record in sterile production, compliance with Good Manufacturing Practices (GMP), and a commitment to continuous improvement is essential. Additionally, geographic location, lead times, and logistical considerations play a significant role, especially in global procurement.
Ultimately, effective due diligence, including on-site audits and product testing, coupled with long-term supplier relationship management, will ensure a sustainable and secure supply of IV fluids. By prioritizing safety, quality, and reliability, healthcare providers and procurement organizations can safeguard patient outcomes and maintain operational resilience in an increasingly demanding healthcare landscape.