The global body jewelry market is experiencing robust growth, driven by rising consumer interest in body modifications and increasing demand for high-quality, hypoallergenic materials. According to Grand View Research, the global body jewelry market size was valued at USD 7.8 billion in 2022 and is expected to expand at a compound annual growth rate (CAGR) of 8.4% from 2023 to 2030. Industrial piercings, in particular, have gained popularity due to their bold aesthetic and structural complexity, fueling demand for precision-crafted, durable jewelry. This surge has led to a proliferation of specialized manufacturers focusing on industrial piercing hardware, including barbells, cuffs, and connectors made from surgical-grade titanium, niobium, and bio-compatible alloys. As the market becomes increasingly competitive and design-savvy, identifying manufacturers that combine innovation, quality, and scalability is critical for retailers and piercers alike. Based on production capacity, material standards, distribution reach, and customer feedback, the following nine manufacturers represent leaders in the industrial piercing jewelry space.
Top 9 Industrial Piercings Jewelry Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Wholesale Industrial Barbells
Domain Est. 2001
Website: piercebody.com
Key Highlights: Rating 4.8 (142) · Free deliveryGrab wholesale industrial barbells in G23 Titanium, and 316L steel for your shop. Strong, high-quality designs with bulk options and worldwide s…
#2 Wholesale Body Jewelry Factory
Domain Est. 1999
#3 The World’s No.1 Piercing Shop
Domain Est. 2005
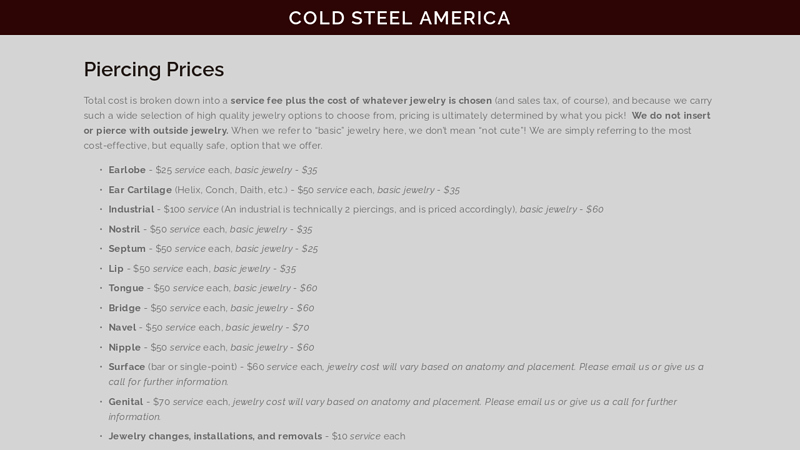
#4 Jewelry & Pricing
Domain Est. 2009
Website: coldsteelpiercing.com
Key Highlights: – $50 service each, basic jewelry – $35. Industrial – $100 service (An industrial is technically 2 piercings, and is priced accordingly), basic jewelry – $60….
#5 Industrial Barbells
Domain Est. 2010
#6 Implant Grade
Domain Est. 2018
Website: implantgrade.com
Key Highlights: Official shop Implant Grade – piercing jewelry manufacturer. High quality titanium ASTM F-136. Free shipping available….
#7 Metal Mafia
Domain Est. 2002
Website: metalmafia.com
Key Highlights: We manufacture the highest quality wholesale piercing jewelry, including 316L Stainless Steel, ASTM F-136 Titanium, Solid Gold and Glass body jewelry….
#8 Hollywood Body Jewelry
Domain Est. 2002
Website: hollywoodbodyjewelry.com
Key Highlights: Free delivery over $300 21-day returnsHollywood Body Jewelry is a website dedicated to wholesale body jewelry. We strive to provide the most complete line of body jewelry and stain…
#9 Bodymod
Domain Est. 2003
Website: bodymod.com
Key Highlights: Free delivery over $20 100-day returnsDiscover the best body piercing jewelry online at Bodymod – A worldwide piercing shop with more than 1500 styles to find your missing pieces o…
Expert Sourcing Insights for Industrial Piercings Jewelry

2026 Market Trends for Industrial Piercing Jewelry
The industrial piercing jewelry market is poised for significant evolution by 2026, driven by shifting consumer preferences, advancements in materials, and the growing influence of social media and body positivity movements. As body modification continues to gain mainstream acceptance, industrial piercings—once considered edgy or rebellious—are increasingly embraced as a form of personal expression. This analysis explores the key trends expected to shape the industrial piercing jewelry market in 2026.
Rising Demand for Sustainable and Ethical Materials
By 2026, sustainability will be a dominant factor in consumer purchasing decisions. Buyers are increasingly prioritizing eco-friendly and ethically sourced materials such as recycled surgical-grade titanium, bioplastics, and reclaimed gold. Jewelry brands that emphasize transparency in their supply chains and offer carbon-neutral production processes will gain a competitive edge. Lab-grown gemstones and conflict-free diamonds are also expected to become standard in high-end industrial barbells and decorative ends.
Growth of Customization and Personalization
Personalization is transforming the industrial piercing experience. In 2026, customers will expect tailored options, including custom bar lengths, unique end designs, engraving, and modular systems that allow interchangeable charms or gemstones. 3D printing technology will enable bespoke designs at scale, allowing consumers to co-create jewelry that reflects their identity. This trend is particularly strong among Gen Z and younger Millennials who view body jewelry as an extension of individuality.
Expansion of Gender-Neutral and Inclusive Designs
The industrial piercing market is moving toward greater inclusivity, with brands developing gender-neutral designs that appeal across the spectrum. Jewelry lines featuring minimalist aesthetics, neutral colors (such as matte black, brushed silver, and rose gold), and versatile styles will dominate. Marketing campaigns will increasingly feature diverse models, promoting body jewelry as a universal form of self-expression rather than tied to gender norms.
Integration of Smart Technology
While still in early stages, smart wearable technology may begin to influence industrial piercing jewelry by 2026. Emerging concepts include barbells embedded with biometric sensors (e.g., temperature or stress monitors) or NFC chips for digital identity or contact sharing. Though niche, these innovations could appeal to tech-savvy consumers and open new avenues for functionality beyond aesthetics.
Influence of Social Media and Influencer Culture
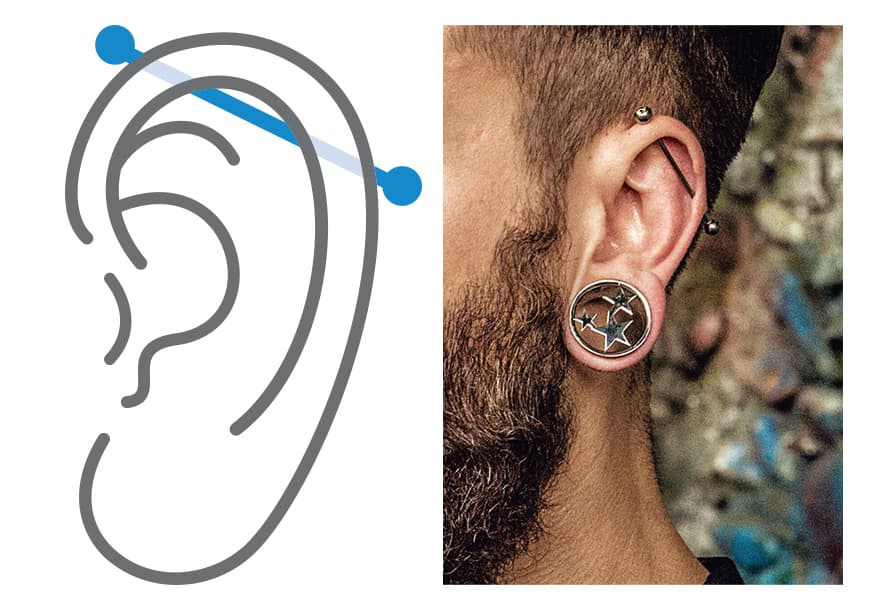
Platforms like Instagram, TikTok, and YouTube will continue to drive trends and normalize industrial piercings. Influencers and piercing artists will play a crucial role in shaping style preferences, with viral “stacked” or “double industrial” looks gaining popularity. Short-form video content will serve as both inspiration and education, helping consumers make informed decisions about jewelry types, aftercare, and healing timelines.
Advancements in Hypoallergenic and Medical-Grade Materials
As awareness of metal sensitivities grows, demand for high-quality, hypoallergenic materials will rise. By 2026, ASTM F136 titanium and ASTM F1295 stainless steel will be industry standards. Innovations such as antimicrobial coatings and bio-compatible polymers will enhance safety and comfort during healing, reducing infection risks and increasing consumer confidence.
E-Commerce and Direct-to-Consumer Dominance
Online retail will remain the primary sales channel, with brands investing in augmented reality (AR) tools that allow customers to visualize how industrial jewelry will look on their anatomy. Subscription boxes, loyalty programs, and virtual consultations with piercing professionals will enhance the digital shopping experience, bridging the gap between online convenience and expert guidance.
Conclusion
The 2026 industrial piercing jewelry market will be defined by innovation, inclusivity, and sustainability. As cultural perceptions evolve and technology advances, consumers will demand more than just aesthetics—they’ll seek meaningful, safe, and personalized experiences. Brands that adapt to these trends by prioritizing ethical practices, customization, and digital engagement will lead the next wave of growth in this dynamic niche.

Common Pitfalls When Sourcing Industrial Piercing Jewelry (Quality & Intellectual Property)
Sourcing industrial piercing jewelry—especially for resale, private label, or brand development—requires careful attention to quality standards and intellectual property rights. Overlooking these areas can lead to customer dissatisfaction, legal issues, and reputational damage. Here are key pitfalls to avoid:
Poor Material Quality and Biocompatibility
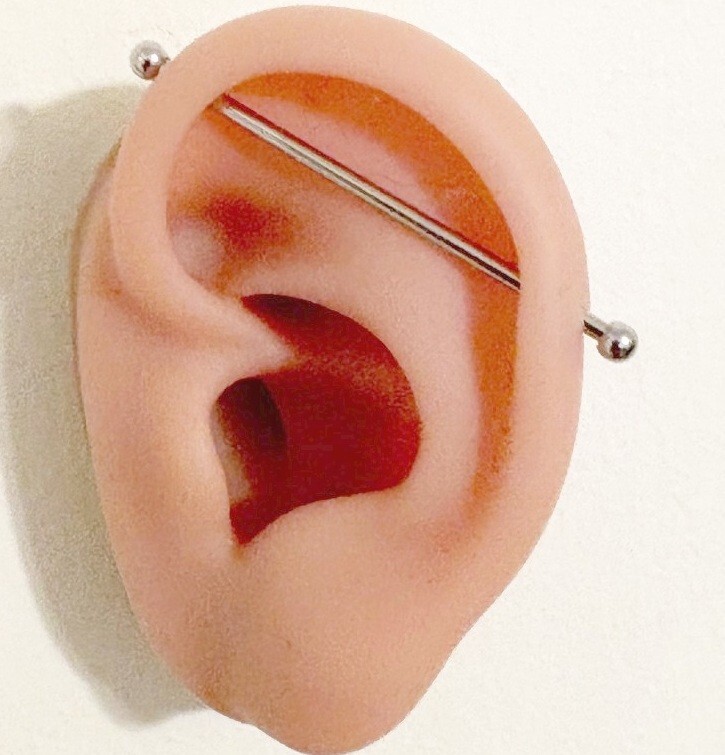
One of the most critical aspects of piercing jewelry is the material used. Industrial piercings are surface piercings prone to migration and rejection, making biocompatible materials essential.
- Using Non-Implant-Grade Materials: Sourcing jewelry made from low-grade surgical steel (e.g., non-ASTM F138 or ISO 5832-1 compliant) can lead to allergic reactions and infections. Always verify that materials meet medical implant standards.
- Mislabeling Titanium: Not all titanium is equal. Ensure that titanium used is ASTM F136 or F67 compliant (implant-grade), as lower grades may contain allergens or impurities.
- Plating Over Base Metals: Jewelry with thin plating (e.g., gold over nickel alloys) can wear off, exposing irritants. This is especially problematic for long-term wear in fresh piercings.
Inadequate Manufacturing Standards
Even with high-quality materials, poor craftsmanship compromises safety and functionality.
- Rough Threads or Edges: Poorly machined threads on barbells can damage tissue or make insertion/removal difficult. Inspect threading precision and surface smoothness.
- Inconsistent Bar Lengths or Gauges: Inaccurate sizing leads to ill-fitting jewelry, increasing the risk of irritation or closure. Ensure consistent manufacturing tolerances.
- Weak or Flimsy Connectors: Industrial bars require strong, secure connectors (e.g., threaded ends). Cheap connectors may loosen or break, risking jewelry loss or injury.
Ignoring Intellectual Property (IP) Rights
When sourcing from manufacturers—especially overseas—copying popular designs can expose you to legal risk.
- Replicating Branded Designs: Copying unique industrial bar configurations, decorative ends, or patented closure mechanisms (e.g., Captive Bead Ring locking systems) may infringe on design patents or trademarks.
- Using Trademarked Names or Logos: Labeling jewelry with brand names like “Anodized Titanium Industrial Bar – Inspired by [Brand X]” can constitute trademark infringement.
- Lack of IP Clearance with Suppliers: Some manufacturers offer “custom” designs that are actually knock-offs. Always request proof of IP ownership or licensing agreements.
Overlooking Certification and Compliance
Selling piercing jewelry often requires adherence to regional health and safety regulations.
- Missing Certifications: In the EU, jewelry must comply with REACH and may require CE marking. In the U.S., while less regulated, using compliant materials is still essential for liability protection.
- No Material Traceability: Reputable suppliers provide material certification (e.g., mill test reports). Without this, you cannot verify claims of implant-grade quality.
Focusing Solely on Price
Low-cost suppliers may cut corners on materials, labor, or IP compliance.
- Hidden Costs of Returns and Complaints: Cheap jewelry may result in higher return rates, negative reviews, and medical complaints—hurting your brand long-term.
- Unethical Manufacturing Practices: Extremely low prices may indicate poor labor conditions or environmental violations, which can damage brand reputation.
Lack of Vendor Due Diligence
Relying on unverified suppliers increases risk across all areas.
- No Factory Audits or Samples: Always request physical samples to assess quality firsthand. Consider third-party audits for high-volume sourcing.
- Unclear Communication on Materials and IP: Ambiguous product descriptions or evasive answers about sourcing should be red flags.
Conclusion
To avoid these pitfalls, prioritize certified biocompatible materials, inspect manufacturing quality rigorously, conduct thorough IP due diligence, and build relationships with transparent, reputable suppliers. Investing in quality and compliance upfront protects your customers—and your business—from avoidable risks.

Logistics & Compliance Guide for Industrial Piercing Jewelry
Product Classification and Regulatory Framework
Industrial piercing jewelry falls under the category of body jewelry and is subject to regulations related to consumer safety, materials used, and import/export compliance. In most jurisdictions, it is classified as a personal accessory rather than a medical device, but it must still adhere to strict standards regarding biocompatibility and material quality. Key regulatory bodies include the U.S. Food and Drug Administration (FDA), the European Union’s REACH and RoHS directives, and similar consumer protection agencies worldwide. Compliance ensures that the jewelry does not contain harmful substances such as nickel above permissible limits, lead, or cadmium.
Material Requirements and Biocompatibility Standards
All industrial piercing jewelry must be manufactured using implant-grade materials to minimize the risk of allergic reactions and infections. Acceptable materials include:
– ASTM F138 or ISO 5832-1 compliant surgical stainless steel
– Titanium (ASTM F67 or F136 grades)
– Solid 14k or 18k nickel-free gold (with proper certification)
– Implant-grade PTFE (for flexible barbells during healing)
Manufacturers and suppliers must provide material certification (e.g., mill test reports) to verify compliance. Jewelry should be free of plating or coatings that may wear off and expose base metals.
Labeling and Packaging Compliance
Proper labeling is essential for regulatory compliance and consumer safety. Each package must include:
– Material composition (e.g., “Implant-Grade Titanium ASTM F136”)
– Size specifications (gauge, length, and curvature)
– Manufacturer or distributor name and contact information
– Country of origin
– Any relevant certifications (e.g., ISO, CE marking where applicable)
Packaging should be tamper-evident and sterile if marketed for new piercings. Multilingual labeling may be required for international distribution.
Import and Export Documentation
Shipping industrial piercing jewelry across borders requires accurate documentation to ensure customs clearance and compliance with destination country laws. Essential documents include:
– Commercial invoice detailing product description, value, and material
– Packing list
– Certificate of Origin
– Material and quality certifications (e.g., mill test report, RoHS/REACH compliance statement)
– Import permits or notifications, if required by the destination country
Ensure Harmonized System (HS) codes are correctly classified—typically under 7113.11 (articles of jewelry and parts, of precious metal) or 7113.20 (of base metal), depending on material.
Storage and Handling Protocols
To maintain product integrity and hygiene, industrial piercing jewelry should be stored in a clean, dry, and temperature-controlled environment. Avoid exposure to moisture, chemicals, or direct sunlight, which may degrade materials or packaging. Handling should follow Good Distribution Practices (GDP), including use of gloves and sanitized tools to prevent contamination, especially for sterile products.
Quality Assurance and Testing Procedures
Suppliers must implement a quality control system that includes:
– Incoming material inspection using spectrometry (e.g., XRF testing) to verify alloy composition
– Dimensional accuracy checks
– Surface finish evaluation (smoothness, absence of burrs)
– Batch sampling and third-party lab testing for restricted substances
Maintain records of all tests and certifications for traceability and audit readiness.
Returns, Recalls, and Incident Reporting
Establish a clear procedure for handling customer complaints, product returns, and potential recalls. If a jewelry item is found to be non-compliant or causes adverse reactions, initiate a recall in accordance with local regulations (e.g., FDA MedWatch in the U.S.). Report serious incidents to relevant authorities and conduct root cause analysis to prevent recurrence.
Sustainability and Ethical Sourcing
Increasingly, compliance includes environmental and social responsibility. Ensure raw materials (especially metals) are sourced from ethical suppliers who adhere to responsible mining practices. Where possible, use recycled metals and minimize packaging waste. Provide transparency through sustainability statements or certifications such as SCS Global or Responsible Jewellery Council (RJC) membership.
Training and Compliance Oversight
Staff involved in logistics, procurement, and customer service should receive regular training on compliance requirements, product specifications, and handling procedures. Appoint a compliance officer or team to monitor regulatory updates, conduct internal audits, and ensure ongoing adherence to all applicable standards.
In conclusion, sourcing industrial piercing jewelry requires careful consideration of material quality, design, safety, and supplier reliability. Opting for implant-grade materials such as titanium (ASTM F136) or high-quality surgical steel ensures biocompatibility and reduces the risk of irritation or infection—especially crucial for a piercing that spans cartilage and involves extended wear. It’s important to choose jewelry with secure, well-fitted ends and appropriate bar length and gauge to suit individual anatomy and promote proper healing.
Working with reputable suppliers who specialize in body jewelry, provide sterilization certifications, and offer transparent sourcing practices helps ensure both safety and longevity of the piece. Whether for personal use or resale, prioritizing quality over cost minimizes complications and enhances customer or wearer satisfaction. Ultimately, informed decisions backed by research and trusted vendors are key to successfully sourcing safe, durable, and aesthetically pleasing industrial piercing jewelry.