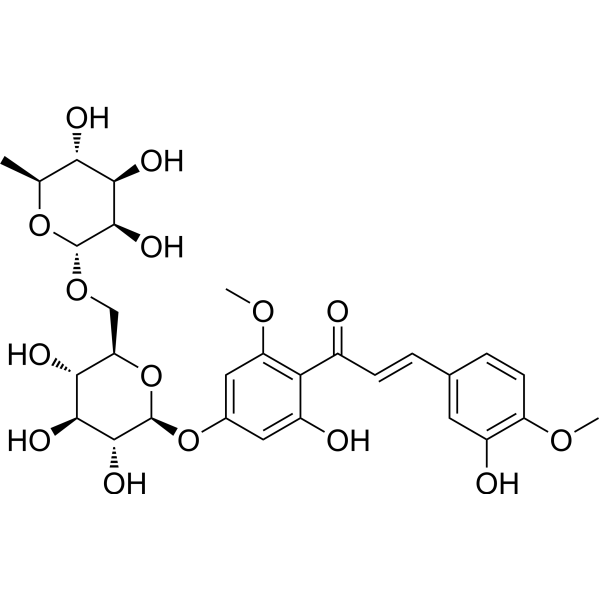
The global hesperidin methyl chalcone market is gaining momentum due to rising consumer demand for natural, plant-derived compounds in skincare and nutraceuticals. According to Grand View Research, the global flavonoids market—of which hesperidin derivatives are a key segment—was valued at USD 2.8 billion in 2022 and is expected to grow at a compound annual growth rate (CAGR) of 7.6% from 2023 to 2030, driven by increased R&D investments and expanding applications in anti-aging, vascular health, and antioxidant formulations. As a methylated derivative of hesperidin, hesperidin methyl chalcone offers enhanced bioavailability and efficacy, making it a sought-after ingredient in premium cosmetic and pharmaceutical products. With Asia-Pacific emerging as a dominant region in flavonoid production and innovation, key manufacturers are ramping up capacity and investing in green extraction technologies to meet growing demand. This increasing market traction underscores the importance of identifying leading producers who combine quality, scalability, and regulatory compliance—factors critical for formulators and brand developers in the health and beauty sectors.
Top 7 Hesperidin Methyl Chalcone Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Hesperidin Methyl Chalcone
Domain Est. 2004
Website: foodchem.cn
Key Highlights: High Quality Hesperidin Methyl Chalcone Supplier, FOODCHEM is Top Hesperidin Methyl Chalcone Supplier and Manufacturer in China for more than 10 years, ……
#2 Hesperidin methylchalcone
Domain Est. 2011
Website: chemfaces.com
Key Highlights: ChemFaces is Reference Standard and high-purity Natural Products Manufacturer of Hesperidin methylchalcone(CFN80405) CAS no.:24292-52-2….
#3 High
Domain Est. 2019
Website: nansproducts.co
Key Highlights: Nans Products is the leading manufacturer and exporter of high-quality Hesperidin Methyl Chalcone (HMC) based products in India. Our HMC is an effective source ……
#4 Hesperidin Methyl Chalcone
Domain Est. 1996
#5 Hesperidin methylchalcone
Domain Est. 1997
Website: pubchem.ncbi.nlm.nih.gov
Key Highlights: Description: Hesperidin methylchalcone has been reported in Myrtus communis with data available. LOTUS – the natural products occurrence database…
#6 Hesperidin Methyl Chalcone For Health
Domain Est. 2017
Website: sentonpharm.com
Key Highlights: Hesperidin methyl chalcone is a compound derived from hesperidin. Hesperidin is an antioxidant bioflavonoid found in large quantities in citrus fruits….
#7 Hesperidin Methyl Chalcone Supplier
Domain Est. 2024
Website: diaopharma.com
Key Highlights: It can even produce cashmere with a perfect diameter of 14.5μm, with a pure cashmere content of over 55%, which is unique worldwide. The preciousness of ……
Expert Sourcing Insights for Hesperidin Methyl Chalcone

H2: 2026 Market Trends Analysis for Hesperidin Methyl Chalcone
Hesperidin Methyl Chalcone (HMC), a semi-synthetic derivative of the natural flavonoid hesperidin, is poised for significant growth and evolution in the 2026 market landscape. Driven by converging trends in consumer health, cosmetic science, and medical research, HMC is transitioning from a niche ingredient to a key player in multiple high-value sectors. Here’s a breakdown of the key trends shaping its 2026 market:
1. Skincare & Cosmetics: Dominant Growth Engine
* Anti-Aging & Brightening Focus: HMC’s potent antioxidant properties and ability to inhibit tyrosinase (the enzyme responsible for melanin production) make it a cornerstone ingredient in premium anti-aging and skin-brightening formulations. Demand will surge for products targeting hyperpigmentation, dark spots, and uneven skin tone.
* Vascular Health & Redness Reduction: Its well-documented benefits in improving microcirculation and strengthening capillary walls position HMC as a critical active for products addressing rosacea, couperose, dark circles, and general skin redness. Consumers seek “calming” and “soothing” solutions, driving formulation demand.
* Clean Beauty & Natural-Derived Actives: While synthetic, HMC’s origin from citrus bioflavonoids (hesperidin) aligns with the “natural-derived” trend. Manufacturers emphasize its plant-based roots to appeal to clean beauty consumers, differentiating it from purely synthetic alternatives.
* Multi-Functional Formulations: HMC will be increasingly combined with other actives (Vitamin C, Niacinamide, Peptides, Hyaluronic Acid) in sophisticated serums and creams, offering synergistic benefits for comprehensive skin health.
2. Dietary Supplements & Nutraceuticals: Expanding Horizons
* Venous Health & Circulation Support: This remains the core application. HMC’s efficacy in reducing capillary permeability and improving venous tone will solidify its position in supplements targeting chronic venous insufficiency (CVI), varicose veins, hemorrhoids, and leg swelling. Aging populations in developed markets are a key driver.
* Antioxidant & Anti-Inflammatory Synergy: HMC will be bundled with other flavonoids (like diosmin, rutin) and antioxidants (Vitamin C, E) in complex formulations promoting overall vascular health, reducing inflammation, and supporting immune function.
* Cognitive & Metabolic Health Exploration: Emerging preclinical research on HMC’s potential neuroprotective and metabolic benefits (e.g., insulin sensitivity) may start influencing supplement formulations by 2026, particularly in the “brain health” and “metabolic support” categories, though clinical evidence will still be developing.
* Focus on Bioavailability: Supplement manufacturers will increasingly highlight HMC’s superior bioavailability compared to standard hesperidin, using this as a key marketing differentiator.
3. Pharmaceutical & Clinical Research: Building the Foundation
* Clinical Trial Validation: Ongoing and planned clinical trials (particularly in Europe and Asia) focusing on HMC’s efficacy in CVI, hemorrhoids, and post-surgical edema will be crucial. Positive results in 2024-2025 will directly fuel market confidence and physician recommendations by 2026.
* Combination Therapies: Research into HMC combined with other phlebotonics (like diosmin) or anti-inflammatory agents for enhanced therapeutic effects will gain traction, potentially leading to new prescription or high-dose OTC products.
* Mechanistic Understanding: Deeper research into HMC’s molecular mechanisms (beyond capillary stabilization, e.g., effects on nitric oxide, inflammatory cytokines) will strengthen its scientific credibility and open doors to new applications.
4. Market Dynamics & Competitive Landscape:
* Supply Chain & Sourcing: Demand will push for stable, high-quality HMC supply. Manufacturers with reliable, scalable synthesis processes (often starting from citrus peel waste) will have a competitive advantage. Sustainability in sourcing may become a minor differentiator.
* Price Pressure & Competition: As more suppliers enter the market (especially from Asia), moderate price competition is likely. However, high-purity grades for pharmaceuticals and premium cosmetics will maintain higher price points.
* Regulatory Landscape: Clear regulatory status (GRAS, approved as a cosmetic ingredient, recognized in pharmacopoeias like Ph. Eur.) provides stability. However, evolving regulations around health claims in supplements and cosmetics will require careful navigation by marketers.
* Geographic Growth: Strong growth is expected in North America (driven by supplements and premium skincare), Europe (established phlebotonic market and advanced skincare), and Asia-Pacific (rapidly expanding skincare market and growing health awareness, particularly in China, Japan, South Korea).
5. Key Challenges for 2026:
* Claim Substantiation: While benefits are established for venous health and skin brightening, robust human clinical data for newer potential applications (cognitive, metabolic) will still be limited, restricting definitive health claims.
* Consumer Education: Explaining the difference between HMC and regular hesperidin, and its specific benefits, remains crucial for both supplement and cosmetic adoption.
* Formulation Stability: Ensuring stability and efficacy of HMC in complex cosmetic formulations (especially at low pH or with certain other actives) requires expertise.
Conclusion for 2026:
The Hesperidin Methyl Chalcone market in 2026 will be characterized by robust growth, particularly in premium skincare and established nutraceuticals, driven by strong scientific rationale for vascular and skin health benefits. Its position as a highly bioavailable, multi-functional ingredient with a “natural-derived” appeal will solidify its value. Success will depend on continued investment in clinical research, effective consumer education, strategic formulation, and navigating competitive pressures. While primarily anchored in venous and skin health, emerging research may begin to open secondary application avenues, setting the stage for further expansion beyond 2026.

H2: Common Pitfalls in Sourcing Hesperidin Methyl Chalcone (Quality & IP)
Sourcing Hesperidin Methyl Chalcone (HMC), a semi-synthetic derivative of citrus bioflavonoids valued for its stability and bioavailability in nutraceuticals and cosmetics, presents specific challenges. Overlooking quality and intellectual property (IP) aspects can lead to significant risks, including product failure, regulatory non-compliance, and legal disputes.
H3: Quality-Related Pitfalls
-
Inadequate Purity & Identity Verification:
- Pitfall: Accepting supplier-provided Certificates of Analysis (CoA) without independent verification. CoAs may be inaccurate, incomplete, or based on non-specific tests (like simple UV-Vis), failing to detect impurities or isomers.
- Risk: Receiving material with low active HMC content, high levels of residual solvents, unreacted starting materials (hesperidin), degradation products, or incorrect isomers (e.g., trans vs. cis chalcone), compromising efficacy and safety.
- Mitigation: Require rigorous analytical methods in specifications: HPLC-UV or HPLC-MS for precise quantification and identification of HMC and key impurities. Demand method validation data from the supplier. Conduct independent batch testing upon receipt.
-
Insufficient Impurity Profiling:
- Pitfall: Focusing only on HMC content while ignoring the impurity profile. Residual catalysts (e.g., palladium from hydrogenation steps), solvents (DMF, DMSO), or toxic by-products from the methylation/chalcone formation process can be present.
- Risk: Contamination exceeding safety thresholds (e.g., ICH Q3D for elemental impurities, ICH Q3C for residual solvents), leading to regulatory rejection or health concerns.
- Mitigation: Specify strict limits for residual solvents, heavy metals (especially Pd, Ni), chlorides, sulfates, and known process-related impurities in the purchase agreement. Require CoAs reporting these tests using validated methods (GC for solvents, ICP-MS/AAS for metals).
-
Inconsistent Batch-to-Batch Quality:
- Pitfall: Sourcing from suppliers without robust Quality Management Systems (QMS) or process validation. Small changes in synthesis or purification can drastically alter HMC quality.
- Risk: Variable product performance, inconsistent clinical results (if applicable), and formulation instability.
- Mitigation: Audit supplier facilities (or require third-party audit reports like ISO 9001/13485 certification). Demand process validation data and evidence of process control. Implement stringent incoming inspection protocols.
-
Stability & Degradation Concerns:
- Pitfall: Assuming HMC is inherently stable without assessing supplier storage and handling practices. HMC can degrade under heat, light, or moisture.
- Risk: Receiving degraded material, leading to reduced potency and potential formation of unknown degradation products.
- Mitigation: Specify storage conditions (temperature, humidity, light protection) in the contract. Require stability data (accelerated and long-term) on the supplied batches. Ensure appropriate packaging (e.g., nitrogen-flushed, opaque containers).
H3: Intellectual Property (IP)-Related Pitfalls
-
Infringing on Patented Processes:
- Pitfall: Sourcing HMC produced using a patented synthetic route without a license. The methylation and chalcone formation steps are complex, and specific catalysts, conditions, or purification methods may be covered by patents.
- Risk: Direct infringement liability for the buyer (you) and/or the supplier. Potential lawsuits, injunctions halting product sales, and significant financial damages. Supply chain disruption.
- Mitigation: Conduct a freedom-to-operate (FTO) analysis before finalizing sourcing. Investigate key patents related to HMC synthesis (e.g., patents held by major producers like Chisso or its successors). Require the supplier to warrant they have the right to use the manufacturing process and indemnify you against IP infringement claims. Consider sourcing from patent holders or licensees.
-
Unclear IP Ownership of Specifications/Modifications:
- Pitfall: Collaborating with a supplier to develop a custom HMC grade (e.g., specific particle size, co-crystal) without a clear agreement on who owns the resulting IP.
- Risk: Disputes over ownership of the improved product, inability to use the modified material freely, or supplier demanding higher prices or exclusivity.
- Mitigation: Establish clear IP ownership clauses in development agreements. Define upfront who owns background IP, foreground IP (the new modification), and know-how. Specify licensing terms if necessary.
-
Trade Secret Misappropriation Risk:
- Pitfall: A supplier might use a competitor’s proprietary (but unpatented) process learned through questionable means to produce cheaper HMC.
- Risk: While you might not directly misappropriate, sourcing from such a supplier could still expose you to legal entanglement (e.g., claims of contributory infringement or unfair competition) and reputational damage.
- Mitigation: Perform due diligence on the supplier’s reputation and sourcing practices. Include contractual clauses requiring the supplier to use only lawful and non-infringing processes and materials.
In summary, successfully sourcing Hesperidin Methyl Chalcone requires a proactive approach. Demand rigorous, independently verifiable quality specifications covering identity, purity, and impurities. Conduct thorough IP due diligence, particularly on the manufacturing process, to avoid costly infringement. Always secure clear contractual protections regarding quality, IP, and liability.

H2: Logistics & Compliance Guide for Hesperidin Methyl Chalcone
Hesperidin Methyl Chalcone (HMC) is a semi-synthetic flavonoid used primarily in cosmetics and dietary supplements for its purported anti-inflammatory and vascular-strengthening properties. Ensuring compliant and efficient logistics for HMC requires strict adherence to regulatory frameworks and careful handling protocols. This guide outlines key considerations.
Regulatory Classification & Documentation
- Primary Use Dictates Regulation:
- Cosmetic Ingredient: Regulated by local cosmetic authorities (e.g., EC No 1223/2009 in EU, FDA guidelines in US). Requires a Cosmetic Product Safety Report (CPSR) in the EU. The supplier should provide a full INCI name (Hesperidin Methyl Chalcone) and Certificate of Analysis (CoA).
- Dietary Supplement Ingredient: Regulated as a food ingredient/supplement. In the US, requires compliance with DSHEA (Dietary Supplement Health and Education Act) and FDA regulations (21 CFR Part 111 for GMPs). In the EU, may require Novel Food authorization unless established before May 1997. A detailed CoA and potentially a GRAS (Generally Recognized As Safe) dossier are critical.
- Safety Data Sheet (SDS): Mandatory. Must be obtained from the supplier and comply with the target market’s GHS (Globally Harmonized System) version (e.g., OSHA HazCom 2012 in US, CLP Regulation in EU). Classify based on available data (typically handled as non-hazardous or low hazard, but confirm specifics like dust explosion potential, eye/skin irritation).
- Certificate of Analysis (CoA): Essential for quality control. Must include specifications (e.g., assay/purity ≥XX%, identification by HPLC/UV, heavy metals <X ppm, residual solvents <X ppm, microbial limits) and actual test results for the batch.
- Certificate of Origin: Often required for import customs clearance.
- Import/Export Licenses: Verify if specific licenses are needed based on the country of origin, destination, and intended use (e.g., some countries regulate botanical derivatives more strictly).
Packaging & Labeling
- Primary Packaging: Use sealed, moisture-resistant, light-protective containers (e.g., double-layered PE bags inside fiber drums, HDPE bottles with liners). Ensure compatibility to prevent leaching or degradation.
- Secondary Packaging: Use sturdy cartons or drums. Clearly label with:
- Product Name: Hesperidin Methyl Chalcone
- Batch/Lot Number
- Net Weight
- Manufacturer/Supplier Name & Address
- “Keep container tightly closed. Store in a cool, dry place.”
- Relevant GHS Pictograms (if classified as hazardous) and Hazard/Precautionary Statements from the SDS.
- INCI Name (for cosmetics).
- Expiry/Use-By Date.
- Transport Labels: Affix appropriate UN transport hazard labels only if the material meets classification criteria (e.g., if dust explosion risk is significant, UN 1350 for Combustible Dust might apply, but HMC is often non-classified). “Fragile” and “This Way Up” labels are recommended.
Storage & Handling
- Conditions: Store in a cool (typically 15-25°C), dry, well-ventilated area away from direct sunlight, heat sources, and incompatible materials. Protect from moisture.
- Containers: Keep containers tightly sealed when not in use.
- Handling: Use in well-ventilated areas. Avoid generating dust. Use appropriate PPE: dust mask (e.g., N95), safety glasses, and gloves. Implement local exhaust ventilation if handling powder frequently.
- Segregation: Store away from strong oxidizing agents and acids.
Transportation
- Mode: Typically shipped as general cargo via air (IATA) or sea (IMDG) freight, or ground transport (ADR/RID for road/rail in Europe).
- Classification: Crucial Step. Classify according to:
- IATA DGR (Air): Check if meets criteria for Dangerous Goods (e.g., Class 4.1 Flammable Solid, Class 9 Miscellaneous). Most HMC shipments are not classified as dangerous goods if purity is high and dust explosion risk is low/controlled, but professional assessment is mandatory.
- IMDG Code (Sea): Same classification principles as IATA.
- ADR/RID (Road/Rail Europe): Same principles.
- Packaging: Use packaging certified for the mode of transport if classified as dangerous goods. For non-hazardous shipments, use robust packaging suitable for the journey (e.g., UN-certified fiberboard boxes or drums if required by carrier). Ensure adequate cushioning.
- Documentation: Commercial Invoice, Packing List, Bill of Lading/Air Waybill, SDS, CoA, Certificate of Origin. Dangerous Goods Declaration is mandatory only if classified as hazardous.
Key Compliance Considerations by Region
- European Union:
- Cosmetics: Full compliance with EC 1223/2009. CPSR, notification via CPNP, presence on Annex list or safety substantiation.
- Food/Supplements: Check Novel Food Catalogue. May require authorization. Compliance with food contact material regulations for packaging.
- REACH: Confirm if registration is needed (likely as an intermediate or with tonnage thresholds).
- United States:
- Cosmetics: FDA oversight (no pre-market approval, but must be safe and properly labeled). Voluntary Cosmetic Registration Program (VCRP).
- Dietary Supplements: DSHEA compliance. GMPs (21 CFR 111). Notification of New Dietary Ingredient (NDI) if applicable.
- TSCA: Confirm chemical substance is listed.
- Other Regions: Research specific regulations in target markets (e.g., Health Canada, China NMPA, Japan MHLW). Regulations for cosmetics and supplements vary significantly.
Critical Recommendations
- Confirm Classification: Never assume HMC is non-hazardous. Obtain a current, jurisdiction-specific SDS and have it reviewed by a qualified professional for transport classification (IATA/IMDG/ADR).
- Verify Intended Use: Ensure the regulatory strategy (Cosmetic vs. Food/Supplement) is clear and all documentation (CoA specs, SDS, regulatory dossiers) aligns perfectly.
- Supplier Qualification: Only source from reputable suppliers providing full, accurate documentation (SDS, CoA, CoO) and adhering to GMP.
- Stay Updated: Regulations (especially Novel Foods, cosmetic bans) change frequently. Monitor updates from relevant authorities.
- Consult Experts: Engage regulatory consultants or legal experts specializing in chemicals, cosmetics, or food supplements in your target markets.
Disclaimer: This guide provides general information. Regulations are complex and subject to change. Always consult with qualified regulatory, safety, and logistics professionals for your specific product, batch, and shipment details.
Conclusion for Sourcing Hesperidin Methyl Chalcone
Sourcing hesperidin methyl chalcone requires a strategic approach due to its specialized applications in the cosmetic, nutraceutical, and pharmaceutical industries. As a derivative of hesperidin known for its enhanced bioavailability and potent antioxidant, anti-inflammatory, and vasoprotective properties, it is essential to identify reliable suppliers who adhere to high standards of purity, quality control, and regulatory compliance.
Key considerations in the sourcing process include evaluating supplier certifications (such as GMP, ISO, or organic compliance), verifying analytical testing (e.g., HPLC for purity), and ensuring sustainable and ethical raw material sourcing. Additionally, transparency in manufacturing processes and documentation, including COAs (Certificates of Analysis), is critical for product safety and regulatory approval.
Given that hesperidin methyl chalcone is not as widely available as other flavonoids, building long-term relationships with reputable suppliers—particularly those with experience in active pharmaceutical or cosmetic ingredients—can ensure consistent supply and quality. Competitive pricing, scalability, and technical support should also factor into the final supplier selection.
In conclusion, successful sourcing of hesperidin methyl chalcone hinges on balancing quality, compliance, and supply chain reliability. Investing time in due diligence and supplier vetting will ultimately support product efficacy, regulatory adherence, and market competitiveness.