The global heparin syringes market is experiencing steady expansion, driven by rising prevalence of cardiovascular diseases, increasing hospitalizations requiring anticoagulant therapies, and growing demand for prefilled syringes for improved dosage accuracy and infection control. According to Grand View Research, the global heparin market size was valued at USD 7.1 billion in 2022 and is expected to grow at a compound annual growth rate (CAGR) of 5.8% from 2023 to 2030, with prefilled syringes representing a significant and growing segment. Similarly, Mordor Intelligence projects continued growth in anticoagulant delivery devices, citing advancements in injectable formulations and rising geriatric populations as key contributors. In this evolving landscape, manufacturers of heparin syringes are scaling production, enhancing safety features, and expanding geographically to meet clinical demand—setting the stage for the following analysis of the top seven companies shaping the market.
Top 7 Heparin Syringes Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
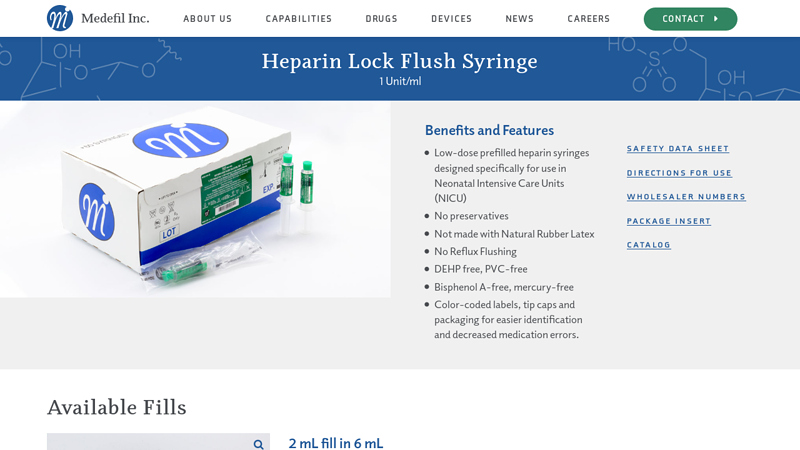
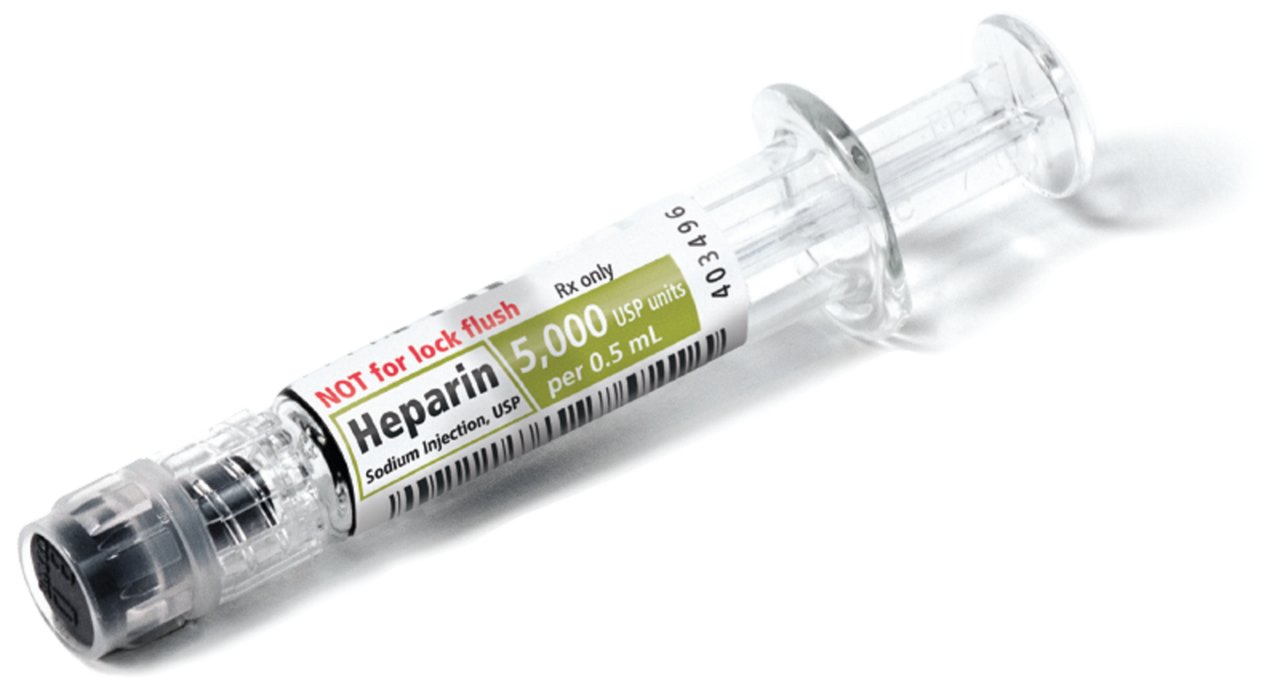
#1 Heparin Lock Flush Syringe, 1 Unit/ml
Domain Est. 2012
Website: medefilinc.com
Key Highlights: Low-dose prefilled heparin syringes designed specifically for use in Neonatal Intensive Care Units (NICU) · No preservatives · Not made with Natural Rubber Latex ……
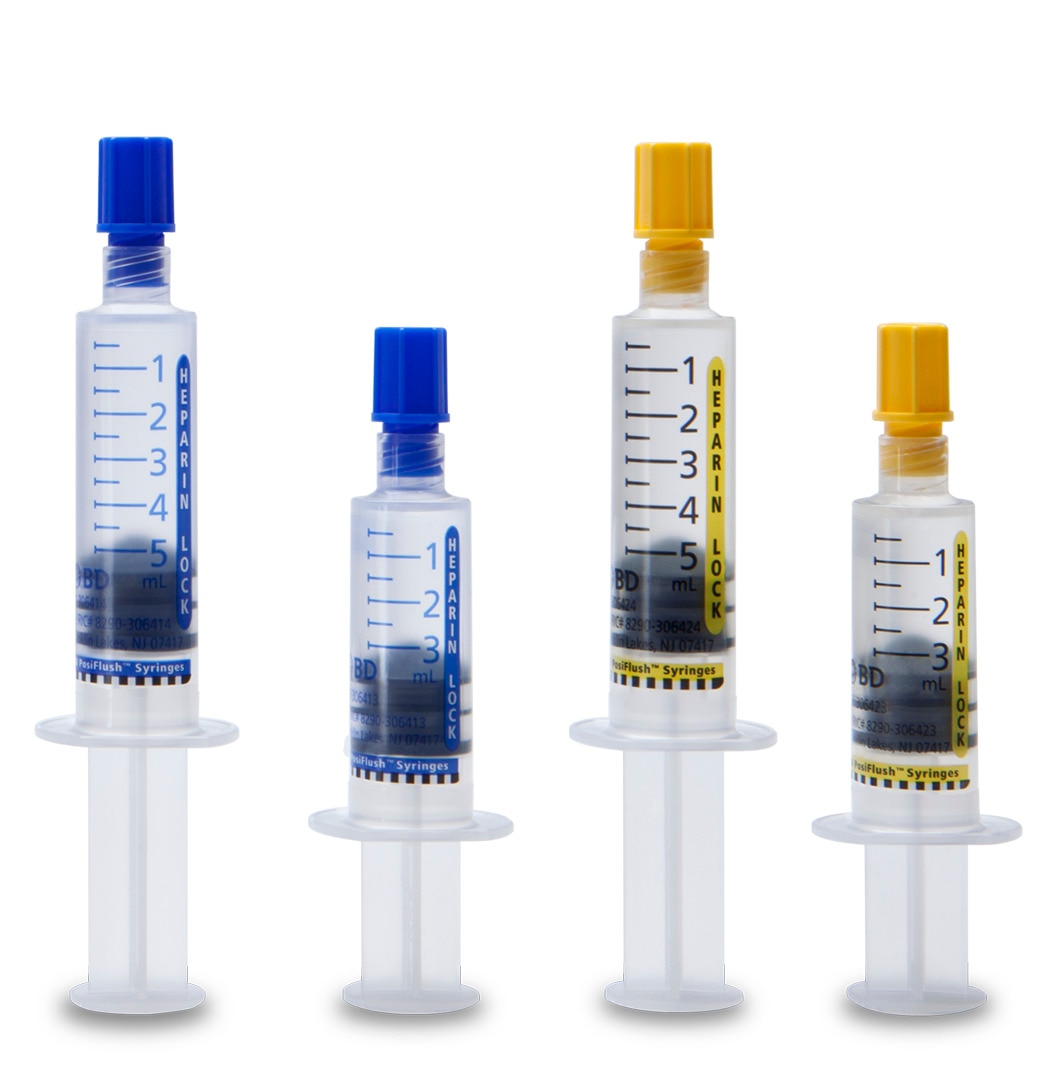
#2 BD PosiFlush™ Prefilled Heparin Lock Flush Syringe
Domain Est. 1990
Website: bd.com
Key Highlights: BD PosiFlush™ Prefilled Heparin Lock Flush Syringe is available in two concentrations—10 USP units per mL and 100 USP units per mL—in 3mL and 5mL syringe ……
#3 Fresenius Kabi Introduces Heparin Sodium Injection, USP Simplist …
Domain Est. 1998
Website: fresenius-kabi.com
Key Highlights: Fresenius Kabi manufactures Simplist Heparin prefilled syringes in the United States. The drug’s active pharmaceutical ingredient is sourced from Europe. ……
#4 Heparin Syringe with an attached needle protection system
Domain Est. 2002
Website: schreiner-group.com
Key Highlights: This is the first prefilled heparin syringe with an integrated needle protection device approved by the US Food and Drug Administration (FDA)….
#5 SagentPharma®
Domain Est. 2006
Website: sagentpharma.com
Key Highlights: We offer a range of therapeutic classes in multiple packaging configurations including premixed bags, prefilled syringes and vials. Exceptional service. Our ……
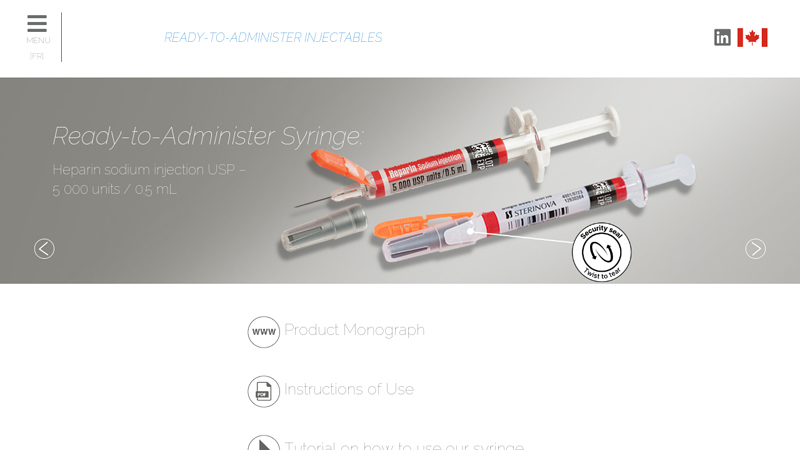
#6 Heparin sodium injection USP : Prefilled syringe
Domain Est. 2010
Website: sterinova.com
Key Highlights: Main characteristics: Single dosage of 5000 units; 27G ½ needle for subcutaneous administration; Needle safety device to help prevent needle stick injuries ……
#7 Heparin Injection, USP
Domain Est. 2016
Website: simplist-us.com
Key Highlights: Heparin Prefilled Syringe 5000 USP unitsper 0.5 mL and 5000 USP units per 1 mLSimplist® ready-to-administer prefilled syringes —available from Fresenius ……
Expert Sourcing Insights for Heparin Syringes

H2: Projected Market Trends for Heparin Syringes in 2026
The global heparin syringes market is poised for continued evolution in 2026, driven by a confluence of healthcare demands, technological advancements, and regulatory shifts. Key trends shaping the landscape include:
- Sustained Demand from Cardiovascular and Renal Care: The escalating global prevalence of cardiovascular diseases (CVDs), deep vein thrombosis (DVT), and chronic kidney disease (CKD) requiring dialysis will remain the primary driver. An aging population inherently more susceptible to these conditions ensures a stable and growing patient base reliant on anticoagulant therapy, including heparin flushes via syringes to maintain vascular access patency.
- Heightened Focus on Safety and Standardization: Regulatory bodies (FDA, EMA, etc.) will continue to emphasize prefilled, single-dose heparin syringes over multi-dose vials or compounded solutions. This trend, driven by the critical need to eliminate medication errors, prevent contamination, and ensure precise dosing (especially the shift towards lower concentrations like 10-100 units/mL for flushes), will solidify prefilled syringes as the standard of care.
- Technological Integration and Smart Features: Expect increased investment in smart syringe technologies. Features like integrated barcode scanning for automated documentation, dose verification systems, and potential integration with Electronic Health Records (EHRs) will gain traction. These innovations aim to enhance patient safety, streamline workflows, and provide valuable data analytics for hospitals.
- Supply Chain Resilience and Diversification: Lingering vulnerabilities in the heparin supply chain (historically reliant on porcine sources, primarily from China) will push manufacturers towards greater transparency, stricter quality control, and diversification of sourcing. Investment in alternative production methods (e.g., biosynthetic heparin, though still emerging) and regional manufacturing hubs may accelerate to mitigate geopolitical and logistical risks.
- Cost-Pressure and Value-Based Procurement: Healthcare systems globally face significant cost pressures. This will intensify competition among syringe manufacturers, driving innovation in cost-effective manufacturing and packaging. Procurement decisions will increasingly focus on total value – encompassing not just unit price, but also safety features, waste reduction, supply reliability, and integration capabilities.
- Regional Market Variations: Growth rates will differ significantly:
- Asia-Pacific: Expected to be the fastest-growing region due to rising healthcare expenditure, expanding dialysis populations, improving hospital infrastructure, and increasing adoption of standardized safety practices.
- North America & Europe: Mature markets focused on safety compliance, technological upgrades, and managing chronic disease burdens. Growth will be steady, driven by aging demographics and replacement demand.
- Emerging Markets: Potential for growth exists, but adoption will be constrained by cost sensitivity and infrastructure limitations, favoring basic prefilled syringes initially.
In summary, the 2026 heparin syringe market will be characterized by consolidation around prefilled, single-dose formats, driven by unwavering safety mandates and chronic disease prevalence, while technological innovation and supply chain security become critical competitive differentiators amidst persistent cost containment pressures.

H2: Common Pitfalls in Sourcing Heparin Syringes – Quality and Intellectual Property (IP) Concerns
Sourcing heparin syringes involves navigating complex regulatory, quality, and intellectual property landscapes. Buyers and healthcare providers must be vigilant to avoid common pitfalls that can compromise patient safety, lead to legal issues, or disrupt supply chains.
1. Quality Assurance Challenges
-
Inconsistent Manufacturing Standards: Heparin syringes sourced from suppliers in regions with less stringent regulatory oversight may not meet international quality standards (e.g., USP, Ph. Eur., or FDA requirements). Variability in heparin purity, syringe material integrity, or sterilization processes can pose serious patient risks.
-
Risk of Contamination: Heparin, being a biologic derived from animal sources (typically porcine intestine), is susceptible to contamination (e.g., oversulfated chondroitin sulfate, as seen in the 2008 global crisis). Inadequate testing or poor supply chain transparency increases contamination risks.
-
Lack of Regulatory Compliance: Suppliers may claim compliance with ISO 13485 or GMP standards without valid certification. Failure to conduct on-site audits or verify documentation can lead to sourcing substandard products.
-
Inadequate Packaging and Storage Conditions: Improper packaging or temperature control during transit can degrade heparin potency or compromise syringe sterility, especially in global supply chains.
2. Intellectual Property (IP) Risks
-
Patent Infringement: Heparin syringes may incorporate proprietary delivery systems, safety mechanisms (e.g., auto-disable features), or specific formulations protected by patents. Sourcing from manufacturers that replicate patented designs without licensing can expose buyers to legal action, product seizures, or liability claims.
-
Counterfeit or Gray Market Products: The presence of counterfeit syringes in the market—especially in high-demand or supply-constrained environments—raises IP and safety concerns. These products may mimic branded designs but lack proper regulatory approval or quality controls.
-
Unclear IP Ownership in Contracts: Failure to address IP rights in procurement agreements can lead to disputes over product design, modifications, or exclusivity. Buyers may inadvertently support suppliers using unlicensed technology.
-
Biosimilar and Innovation Conflicts: As biosimilar heparins enter the market, distinguishing between authorized generics and infringing products becomes critical. Sourcing decisions must account for approved product status and patent expiry timelines.
Mitigation Strategies
- Conduct thorough due diligence on suppliers, including audits and verification of certifications.
- Require full traceability of heparin raw materials and documentation of quality testing.
- Partner with legally compliant manufacturers and verify freedom-to-operate regarding relevant patents.
- Include IP indemnification clauses in supply contracts.
- Collaborate with regulatory authorities and use approved procurement channels to avoid counterfeit goods.
Avoiding these pitfalls ensures the safe, legal, and effective sourcing of heparin syringes in clinical and hospital settings.

Logistics & Compliance Guide for Heparin Syringes
Heparin syringes are high-alert medications requiring strict adherence to logistics and compliance protocols to ensure patient safety, regulatory compliance, and product integrity. This guide outlines essential best practices for handling, storing, transporting, and managing heparin syringes throughout the supply chain.
Storage Conditions
Heparin syringes must be stored under controlled conditions to maintain potency and sterility.
– Temperature: Maintain storage between 20°C to 25°C (68°F to 77°F), with excursions permitted between 15°C to 30°C (59°F to 86°F) as per USP guidelines. Avoid freezing.
– Light Protection: Store in original packaging to protect from light exposure.
– Humidity: Keep in a dry environment; avoid high humidity to prevent packaging degradation.
– Segregation: Store separately from non-pharmaceutical items and incompatible substances. Clearly label storage areas to prevent mix-ups.
Transportation Requirements
Transportation of heparin syringes must ensure temperature control and physical protection.
– Temperature Monitoring: Use validated cold chain solutions (e.g., insulated containers with temperature data loggers) when ambient conditions exceed recommended storage ranges.
– Packaging: Utilize tamper-evident, shock-resistant packaging. Include cushioning materials to prevent breakage.
– Documentation: Ship with a completed Shipper’s Declaration for Dangerous Goods if required (e.g., for air transport of certain formulations). Include temperature monitoring reports when applicable.
– Transit Time: Minimize transit duration and avoid unnecessary handling or transfers between carriers.
Regulatory Compliance
Heparin syringes are subject to stringent regulatory oversight due to their anticoagulant properties and associated risks.
– FDA Regulations: Comply with 21 CFR Parts 210, 211 (cGMP), and 212 (Radiopharmaceuticals, if applicable). Ensure all products are FDA-approved and sourced from licensed manufacturers.
– Labeling Requirements: Labels must include:
– Drug name (e.g., Heparin Sodium Injection)
– Strength (e.g., 1000 units/mL)
– Expiration date
– Lot number
– Storage instructions
– High-alert medication warning
– USP verification (if applicable)
– Pediatric Syringes: Confirm compliance with FDA’s Unapproved Drugs Initiative; only use FDA-approved heparin flush syringes for pediatric use.
– Controlled Substance Status: While heparin is not a DEA-controlled substance, it is classified as a high-alert medication per ISMP guidelines—requiring heightened handling protocols.
Handling and Dispensing Protocols
Proper handling reduces medication errors and contamination risks.
– Staff Training: Ensure all personnel are trained in safe handling, verification procedures, and error prevention (e.g., distinguishing between concentrations).
– Double-Check Procedures: Implement independent double-checks for dose, concentration, and patient identity prior to administration.
– Barcoding and Scanning: Use barcode-assisted dispensing and administration (BCMA) systems to verify product and patient match.
– Expiration Management: Conduct routine inventory checks to identify and quarantine expired or soon-to-expire products.
Documentation and Traceability
Maintain comprehensive records to support traceability and audits.
– Lot Tracking: Record and retain lot numbers for all received and dispensed heparin syringes per FDA Drug Supply Chain Security Act (DSCSA) requirements.
– Temperature Logs: Archive temperature data from storage and transit for minimum 1 year (or per institutional policy).
– Receiving Documentation: Document receipt date, quantity, lot number, expiration date, and condition of packaging.
– Incident Reporting: Report any suspected counterfeit, diverted, or compromised products to the manufacturer and FDA MedWatch program immediately.
Risk Mitigation and Error Prevention
Heparin has a narrow therapeutic index; errors can lead to severe bleeding or thrombosis.
– Concentration Standardization: Use standardized concentrations (e.g., 10 units/mL for flushes, 100 units/mL for therapeutic use) to reduce confusion.
– Tall Man Lettering: Employ Tall Man lettering on labels and systems (e.g., “heparin” vs. “HYDROmorphone”) to differentiate look-alike/sound-alike drugs.
– Storage Separation: Physically separate different concentrations and dosage forms (e.g., syringes vs. vials).
– Emergency Protocols: Establish protocols for heparin overdose (e.g., availability of protamine sulfate).
Disposal and Waste Management
Dispose of unused or expired heparin syringes in accordance with federal and local regulations.
– Sharps Containers: Used syringes must be discarded in FDA-cleared sharps disposal containers.
– Hazardous Waste: While heparin is not typically classified as hazardous waste under RCRA, confirm local regulations.
– Documentation: Maintain logs of waste disposal, including date, quantity, and method.
Audits and Continuous Improvement
Conduct regular internal audits to ensure compliance and identify areas for improvement.
– Audit Frequency: Perform quarterly audits of storage, handling, and documentation practices.
– Regulatory Updates: Monitor FDA, ISMP, and USP updates related to heparin products and revise protocols accordingly.
– Feedback Loop: Encourage staff reporting of near-misses or discrepancies via non-punitive error reporting systems.
Adhering to this logistics and compliance guide ensures the safe and effective use of heparin syringes, protects patient safety, and maintains regulatory readiness.
Conclusion for Sourcing Heparin Syringes
In conclusion, the sourcing of heparin syringes requires a strategic and comprehensive approach that balances quality, regulatory compliance, cost-effectiveness, and supply chain reliability. Given the critical nature of heparin as an anticoagulant used in various clinical settings—including surgery, dialysis, and intensive care—ensuring consistent access to high-quality, sterile, and accurately dosed syringes is paramount for patient safety.
Effective sourcing involves due diligence in selecting suppliers with proven adherence to Good Manufacturing Practices (GMP), regulatory approvals from agencies such as the FDA or EMA, and a transparent supply chain—particularly considering heparin’s origin from animal sources, which presents both biological and geopolitical risks. Diversification of suppliers, long-term contracts, and robust inventory management can mitigate risks of shortages or contamination.
Furthermore, collaboration with healthcare providers, pharmacists, and procurement teams ensures that clinical needs are aligned with procurement strategies. Continued monitoring of market trends, regulatory updates, and emerging alternatives or technologies (such as prefilled syringes or biosimilars) will support agile and informed decision-making.
Ultimately, a proactive, compliant, and patient-centered sourcing strategy for heparin syringes is essential to maintain uninterrupted care, uphold safety standards, and support operational efficiency across healthcare systems.