The global wound drainage systems market is experiencing steady growth, driven by rising surgical volumes, increasing prevalence of chronic diseases, and the growing emphasis on post-operative infection control. According to Mordor Intelligence, the wound care market is projected to grow at a CAGR of approximately 5.8% from 2023 to 2028, with surgical wound management devices like Hemovac drains representing a significant segment. Similarly, Grand View Research valued the global wound care market at USD 20.9 billion in 2022 and forecasts continued expansion, underpinned by advancements in drainage technologies and heightened demand in ambulatory and home healthcare settings. As hospitals and surgical centers prioritize efficient, reliable post-surgical drainage solutions, Hemovac-style closed suction systems remain a staple in patient recovery protocols. This increasing demand has fostered a competitive landscape among medical device manufacturers. Below are the top four Hemovac wound drain manufacturers leading innovation, quality, and market presence in this evolving sector.
Top 4 Hemovac Wound Drain Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Hemovac Drain
Domain Est. 2003
Website: 4amedical.com
Key Highlights: It consists of a catch pit of 200 cc or 400 cc and a blower and it is used with 10, 12, 14 or 16F trocars. It includes a single or double trocar optionally.Missing: wound manufact…
#2 Wound Drainage & Drains
Domain Est. 1996
Website: cardinalhealth.com
Key Highlights: Cardinal Health provides a full assortment of high-quality wound drains and drainage systems used during surgical and clinical procedures….
#3 1018233 CWS 400 CLOSED WOUND SUCTION KIT; HEMOVAC …
Domain Est. 2000
Website: accessdata.fda.gov
Key Highlights: Precautions: ensure that the wound site is dry and free of debris before closure.The surgeon must determine the number of drains needed for an effective ……
#4 Hemovac drain
Domain Est. 2006
Website: boenmedical.com
Key Highlights: Our hemovac drain including 3-spring evacuator, PVC tubing, Y-connector and silicone drain with trocar, is used for closed suction drains after surgery….
Expert Sourcing Insights for Hemovac Wound Drain

H2: Projected 2026 Market Trends for Hemovac Wound Drain
The Hemovac Wound Drain market is anticipated to experience notable shifts by 2026, driven by advancements in post-surgical care, increasing demand for efficient wound management solutions, and rising global surgical volumes. Key trends shaping the market include technological innovation, growing healthcare infrastructure in emerging economies, and a shift toward outpatient and ambulatory surgical settings.
-
Increasing Surgical Procedures and Post-Operative Care Demand
The rising prevalence of orthopedic, plastic, and general surgeries—particularly in aging populations—will drive demand for effective post-operative drainage systems like the Hemovac. By 2026, the global increase in elective and reconstructive surgeries is expected to directly boost Hemovac usage, especially in procedures requiring fluid evacuation to prevent infection and promote healing. -
Technological Advancements and Product Innovation
Manufacturers are focusing on improving Hemovac design for better patient comfort, ease of use, and infection control. Anticipated innovations by 2026 include antimicrobial coatings, integrated volume measurement systems, and compact, portable units designed for home care. These enhancements aim to reduce hospital readmissions and support the growing trend of ambulatory care. -
Expansion in Emerging Markets
Regions such as Asia-Pacific, Latin America, and parts of Africa are expected to witness accelerated market growth due to improving healthcare access, rising surgical rates, and government initiatives to modernize medical infrastructure. Local manufacturing and cost-effective product variants will likely expand Hemovac availability in these regions. -
Shift Toward Reusable and Sustainable Solutions
Environmental and cost concerns are prompting a reevaluation of disposable vs. reusable drainage systems. While disposable Hemovac units remain dominant, reusable silicone-based systems may gain traction by 2026, particularly in cost-sensitive healthcare systems and outpatient centers aiming to reduce medical waste. -
Competitive Landscape and Market Consolidation
The Hemovac market is expected to see increased competition and consolidation among medical device manufacturers. Major players such as Becton Dickinson (BD), Medline Industries, and Teleflex Incorporated are likely to expand their portfolios through strategic acquisitions and R&D investments, focusing on integrated wound care ecosystems. -
Regulatory and Reimbursement Influences
Stringent regulatory standards, particularly in the U.S. (FDA) and EU (MDR), will continue to shape product approvals and quality benchmarks. Simultaneously, favorable reimbursement policies for post-surgical care in key markets will support adoption, while value-based healthcare models may pressure pricing and encourage cost-efficient solutions.
In summary, the Hemovac Wound Drain market in 2026 will be characterized by innovation, geographic expansion, and alignment with evolving care delivery models. As patient-centered outcomes and operational efficiency become paramount, Hemovac and similar devices will play a critical role in modern wound management strategies.
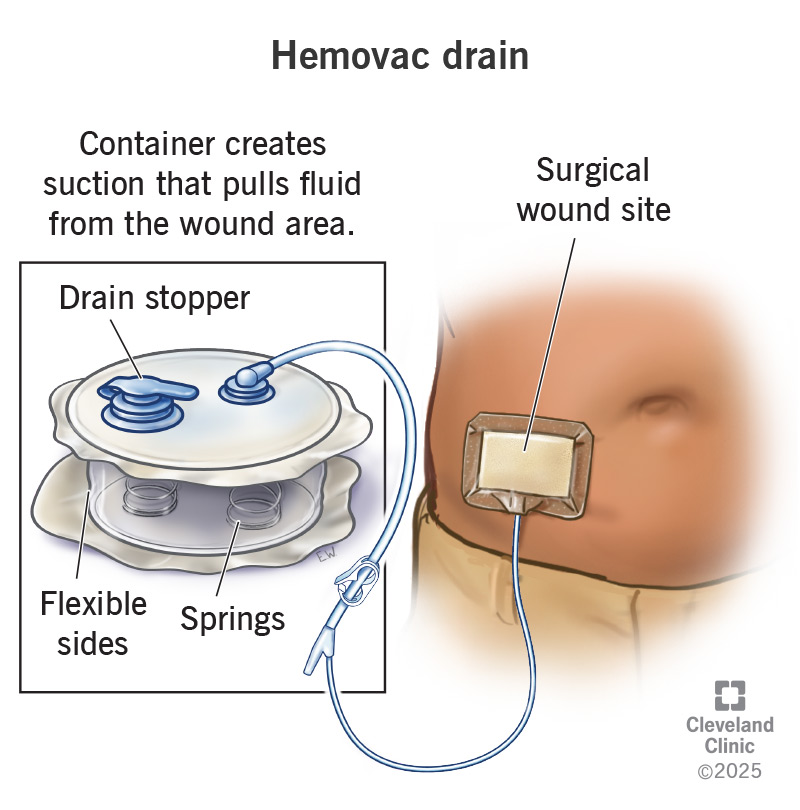
H2: Common Pitfalls in Sourcing Hemovac Wound Drains (Quality and Intellectual Property Risks)
Sourcing Hemovac wound drains—closed-suction drainage systems widely used post-surgery—can present significant challenges, especially concerning product quality and intellectual property (IP) compliance. Organizations must be vigilant to avoid the following common pitfalls:
1. Substandard Product Quality
Many suppliers, particularly from low-cost manufacturing regions, may offer Hemovac drains that do not meet medical-grade standards. Key quality concerns include:
– Use of non-biocompatible or low-grade plastics that may leach harmful substances.
– Inconsistent vacuum seal integrity, leading to reduced drainage efficiency or contamination risk.
– Poor sterility assurance, increasing the risk of postoperative infections.
– Lack of compliance with ISO 13485, FDA 21 CFR Part 820, or other regulatory standards.
Procuring from unverified manufacturers without proper audits or certifications increases the likelihood of receiving defective or unsafe devices.
2. Counterfeit or Imitation Products
The Hemovac® name is a registered trademark (originally by Teleflex Incorporated), and unauthorized production or branding of “Hemovac” products may constitute trademark infringement. Buyers often encounter:
– Products falsely labeled as “Hemovac” despite lacking authorization.
– Look-alike devices that mimic the original design but compromise on performance and safety.
– Suppliers misrepresenting compatibility or equivalence without clinical validation.
These imitations not only pose legal risks but may also expose healthcare providers to liability in case of patient harm.
3. Intellectual Property Infringement
Manufacturers may replicate patented design elements (e.g., bulb reservoir shape, connector systems, or one-way valve mechanisms) without licensing. Sourcing such products—even unknowingly—can result in:
– Customs seizures or import bans due to IP violations.
– Legal action from patent holders against distributors or end-users.
– Reputational damage and supply chain disruption.
Buyers should verify that suppliers hold legitimate rights or design-around patents to avoid contributory infringement.
4. Inadequate Regulatory Documentation
Many suppliers fail to provide essential documentation, such as:
– CE certificates, FDA listings, or local market approvals.
– Technical files, sterilization reports, or biocompatibility test results.
– Traceability data (e.g., lot numbers, expiration dates).
Absence of these documents increases regulatory risk, especially in markets with strict medical device controls.
5. Unreliable Supply Chain Transparency
OEMs or distributors may source components from multiple unverified subcontractors, making it difficult to ensure consistency or trace defects. Lack of transparency complicates recall management and quality investigations.
Mitigation Strategies:
– Source from authorized distributors or manufacturers with proven regulatory compliance.
– Conduct supplier audits and request full quality management system (QMS) certifications.
– Verify trademark and patent status before procurement.
– Include IP indemnification clauses in supply contracts.
– Perform incoming product testing for performance and sterility.
Avoiding these pitfalls ensures patient safety, regulatory compliance, and protection against legal and financial exposure when sourcing Hemovac wound drainage systems.

Logistics & Compliance Guide for Hemovac Wound Drain
Product Overview
The Hemovac Wound Drain is a single-use, closed-suction drainage system designed to remove blood and fluids from surgical sites post-operatively. It consists of a flexible silicone or latex-free rubber drainage tube connected to a compact, portable collection chamber with graduated volume markings. The system operates via negative pressure created by manual compression of the chamber, facilitating continuous drainage while minimizing the risk of infection and promoting wound healing.
Regulatory Classification
The Hemovac Wound Drain is classified as a Class II medical device under the U.S. Food and Drug Administration (FDA) regulations, falling under 21 CFR 878.5025 (Suction Apparatus). It is subject to premarket notification (510(k)) requirements and must comply with general and special controls, including design, performance, and labeling standards. In the European Union, it is categorized as a Class IIa device under Regulation (EU) 2017/745 on Medical Devices (MDR), requiring conformity assessment by a Notified Body and adherence to CE marking requirements.
Labeling and Packaging Requirements
All Hemovac Wound Drains must be labeled in accordance with FDA 21 CFR Part 801 and EU MDR Article 20. Labels must include:
– Device name and intended use
– Manufacturer name and address
– Lot number and expiration date
– Sterile indication and method of sterilization (typically ethylene oxide)
– Single-use statement
– Quantity per package
– UDI (Unique Device Identifier) in compliance with FDA and EU regulations
– Language-specific labeling for local markets (e.g., bilingual labeling in Canada, French in France)
Packaging must maintain sterility throughout distribution and include tamper-evident seals. Inner packaging is typically peelable pouches; outer cartons must be durable and labeled with storage conditions and handling instructions.
Storage and Handling
- Temperature: Store in a dry, cool environment at 15°C to 30°C (59°F to 86°F).
- Humidity: Avoid high humidity; relative humidity should not exceed 60%.
- Light: Protect from direct sunlight and UV exposure.
- Position: Store upright or flat; do not stack excessive weight on packages.
- Shelf Life: Typically 3 to 5 years from manufacture date; confirm expiration on individual packaging.
Transportation Requirements
- Transport in sealed, undamaged cartons on pallets or in containers protected from moisture, extreme temperatures, and physical damage.
- Maintain ambient conditions during transit; use temperature-controlled vehicles if ambient conditions exceed storage limits.
- Avoid freezing and prolonged exposure to temperatures above 40°C (104°F).
- Comply with IATA regulations if shipping internationally via air; no special hazardous material designation applies under normal conditions.
Import/Export Compliance
- U.S. Imports: Submit entry documentation to U.S. Customs and Border Protection (CBP); provide FDA prior notice via the FDA Unified Registration and Listing System (FURLS). The device must be listed, and the facility registered with the FDA.
- EU Exports: Ensure CE certification is valid and technical documentation is available. Notify customs using the EU’s Export Health Certificate (EHC) system where applicable.
- Other Markets: Verify local regulatory requirements (e.g., Health Canada license, ANVISA registration in Brazil, TGA listing in Australia). UDI must be compatible with destination country databases.
Sterility and Quality Assurance
- Each unit is terminally sterilized using ethylene oxide (EtO) and tested for sterility according to ISO 11737-1.
- Manufacturers must comply with ISO 13485:2016 for quality management systems.
- Batch records and certificates of conformance (CoC) must be maintained for traceability and audits.
Distribution Chain Controls
- Distributors and wholesalers must be authorized and maintain compliance with Good Distribution Practice (GDP) for medical devices.
- Cold chain is not required, but temperature excursions must be documented and assessed.
- Implement traceability systems using UDI to support recalls and field actions, if necessary.
Post-Market Surveillance and Reporting
- Adverse events must be reported per FDA’s MedWatch (Form 3500A) and EU’s Eudamed system within required timelines (e.g., 10–15 days for serious incidents).
- Maintain a Post-Market Surveillance (PMS) plan and Periodic Safety Update Reports (PSURs) for EU compliance.
- Monitor customer complaints and initiate field safety corrective actions (FSCA) when warranted.
Disposal and Environmental Compliance
- As a single-use device, Hemovac drains must be disposed of as regulated medical waste following local biohazard regulations (e.g., OSHA Bloodborne Pathogens Standard in the U.S.).
- Do not incinerate unless in approved medical waste facilities; EtO residuals require proper handling.
- Recycling is not recommended due to biological contamination risk.
Training and Documentation
- Ensure clinical and logistics personnel are trained on proper handling, storage, and usage.
- Maintain records of training, device distribution, and quality audits for a minimum of 5 years (or per local requirement).
- Documentation must be available for regulatory inspections by FDA, EU Notified Bodies, or other authorities.
Summary
Proper logistics and compliance for the Hemovac Wound Drain require adherence to medical device regulations, strict control of storage and transportation conditions, accurate labeling, and robust post-market surveillance. Compliance ensures patient safety, regulatory approval, and uninterrupted supply chain operations globally.
Conclusion for Sourcing Hemovac Wound Drains:
Sourcing Hemovac wound drains requires a comprehensive evaluation of product quality, regulatory compliance, supplier reliability, cost-effectiveness, and logistical efficiency. Given the critical role Hemovac drains play in post-surgical care—promoting wound healing by effectively removing blood and fluids—it is essential to partner with reputable manufacturers that adhere to international medical device standards such as ISO 13485 and FDA regulations.
A successful sourcing strategy should prioritize suppliers offering consistent product performance, sterile packaging, and compatibility with standard clinical protocols. Additionally, considerations such as supply chain resilience, lead times, and scalability are crucial to ensuring uninterrupted availability in clinical settings. Engaging in long-term contracts with proven vendors may provide cost advantages and supply stability.
Ultimately, the goal is to secure Hemovac wound drains that meet clinical safety and efficacy requirements while optimizing procurement efficiency and cost management. A well-structured sourcing approach supports improved patient outcomes and operational effectiveness within healthcare facilities.