The global eyeglasses market is witnessing robust expansion, driven by rising vision impairment, increasing fashion consciousness, and growing demand for premium and customized eyewear. According to a report by Grand View Research, the global eyewear market size was valued at USD 161.9 billion in 2022 and is expected to grow at a compound annual growth rate (CAGR) of 4.7% from 2023 to 2030. Similarly, Mordor Intelligence projects a CAGR of over 5% during the forecast period of 2023–2028, citing technological advancements in lens design, the proliferation of online eyewear retail, and increasing disposable incomes in emerging economies as key growth drivers. With the Asia Pacific region emerging as a manufacturing and consumption hub and Europe maintaining dominance through luxury branding and innovation, the competitive landscape is shaped by a mix of long-established manufacturers and agile newcomers. In this evolving market, identifying the top eyeglass manufacturers requires evaluating production scale, R&D investment, global distribution, and brand influence—factors that define industry leadership in both optical performance and style innovation.
Top 10 Eyeglass Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Marchon Eyewear
Domain Est. 1995
Website: marchon.com
Key Highlights: As one of the world’s largest designers, manufacturers and distributors of quality eyewear and sun wear, Marchon Eyewear specializes in premium fashion….
#2 Altair Eyewear
Domain Est. 1997
Website: altaireyewear.com
Key Highlights: As the first not-for-profit eye health company, care and compassion have led our growth for nearly 70 years. Today, our focus remains on helping those we serve….
#3 OTIS Eyewear US
Domain Est. 2000
Website: us.otiseyewear.com
Key Highlights: Welcome to the OTIS Eyewear US site. Shipping only within the US. Shop designer eyewear from naturally sourced, scratch resistant lenses….
#4 DETAILS DEFINE US
Domain Est. 2003
Website: prodesigndenmark.com
Key Highlights: Explore Prodesign’s September collection—innovative frames in titanium, acetate, and stainless steel, designed with precision and bold style….
#5 Eyeglasses
Domain Est. 2004
#6 LamyAmerica.com
Domain Est. 2005
Website: lamyamerica.com
Key Highlights: Welcome to the new L’AmyAmerica.com. All existing and new accounts will require a one-time registration for our new website. Register Now….
#7 FGXI
Domain Est. 2005
Website: fgxi.com
Key Highlights: A leading global designer and marketer of non-prescription reading glasses and sunglasses, we comprise over 50 established, highly recognized eyewear brands….
#8 Designer Eyewear Frames and Luxury Sunglasses
Domain Est. 2007
Website: bartonperreira.com
Key Highlights: Free delivery Free 10-day returnsBarton Perreira is an independent eyewear brand that takes time to invest in craftsmanship that produces nothing less than the finest eyewear in th…
#9 Coco and Breezy Eyewear
Domain Est. 2009
Website: cocoandbreezy.com
Key Highlights: Coco and Breezy designer optical glasses and sunglasses. Free US ground shipping and easy returns. Protecting eyes with modern style….
#10 EssilorLuxottica
Domain Est. 2017
Website: essilorluxottica.com
Key Highlights: EssilorLuxottica: welcome to EssilorLuxottica’s official website. Discover our products, services, and commitment to vision care….
Expert Sourcing Insights for Eyeglass

H2: Emerging Market Trends in the Eyeglasses Industry for 2026
As we approach 2026, the global eyeglasses market is undergoing a transformative shift driven by technological innovation, evolving consumer preferences, and increased health awareness. Key trends shaping the industry include the integration of smart technology, a growing emphasis on sustainability, expansion of direct-to-consumer (DTC) models, and rising demand for personalized eyewear. These dynamics are redefining how consumers interact with vision correction products and influencing both product development and market strategy.
One of the most significant developments is the rise of smart eyewear. With advancements in augmented reality (AR), embedded sensors, and health monitoring capabilities, eyeglasses are evolving into multifunctional devices. By 2026, major tech and optical brands are expected to launch next-generation smart glasses that offer features such as real-time translation, navigation overlays, and even intraocular pressure monitoring for glaucoma patients. This convergence of fashion, function, and health tech is attracting a younger, tech-savvy demographic and expanding the market beyond traditional vision correction.
Sustainability is also becoming a core focus. Consumers are increasingly demanding eco-friendly materials and ethical production practices. Leading eyewear brands are responding by using bio-based acetates, recycled metals, and biodegradable packaging. In 2026, sustainability is not just a marketing advantage but a competitive necessity, with certifications and transparent supply chains becoming key differentiators.
The direct-to-consumer business model continues to disrupt traditional retail. Online platforms offering virtual try-ons, AI-powered face mapping, and home try-on kits have made purchasing eyeglasses more convenient and personalized. These models reduce costs and allow for greater customization, enabling consumers to design frames that reflect individual style and facial ergonomics. By 2026, DTC brands are projected to capture a larger share of the market, especially in North America and Europe.
Additionally, the aging global population and increased screen time are fueling demand for specialized lenses, such as blue light filters, progressive lenses, and photochromic options. Eye health awareness campaigns and tele-optometry services are improving access to vision care, particularly in underserved regions, further expanding the customer base.
In conclusion, the 2026 eyeglasses market is characterized by innovation, personalization, and sustainability. Companies that embrace digital transformation, prioritize environmental responsibility, and meet the growing demand for intelligent, stylish, and health-integrated eyewear will be best positioned for success in this evolving landscape.

Common Pitfalls When Sourcing Eyeglasses: Quality and Intellectual Property Risks
Sourcing eyeglasses, particularly from overseas manufacturers, presents several challenges that can impact brand reputation, customer satisfaction, and legal compliance. Two major areas of concern are product quality and intellectual property (IP) protection. Ignoring these aspects can lead to costly recalls, customer dissatisfaction, and legal disputes.
Quality-Related Pitfalls
Inconsistent Material Standards
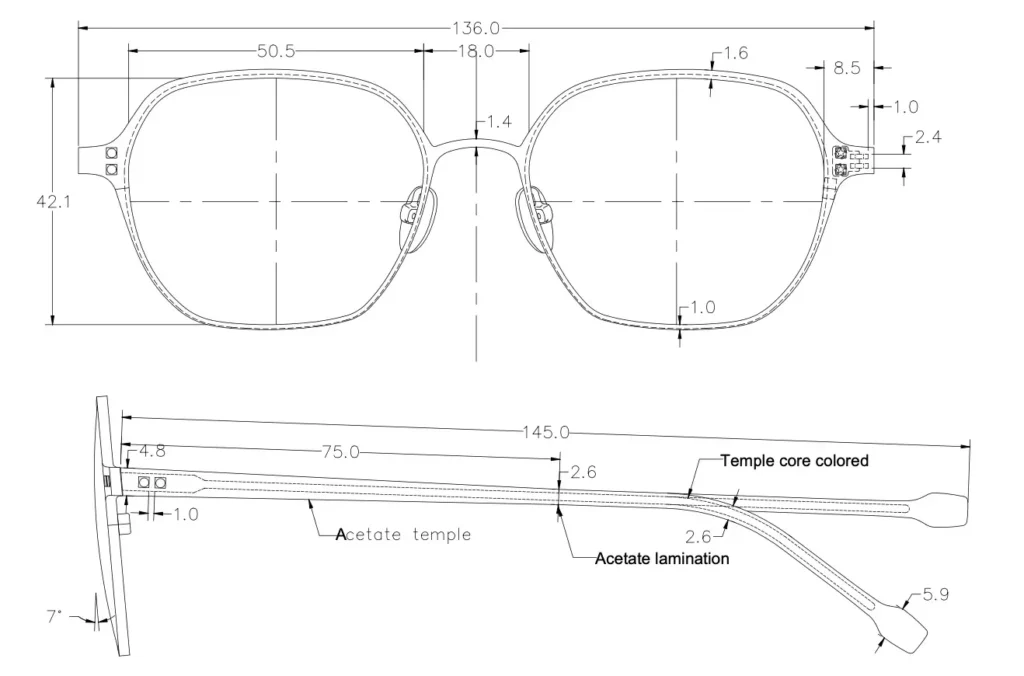
Eyeglass frames and lenses must meet specific optical and durability standards. Sourcing from suppliers who cut corners on materials—such as using low-grade acetate, substandard metals prone to corrosion, or non-impact-resistant lenses—can result in poor product performance and safety risks. Always verify material specifications and ensure suppliers adhere to international standards like ISO or ANSI.
Poor Craftsmanship and Fit
Inadequate quality control during manufacturing can lead to misaligned hinges, uneven frame symmetry, or lenses that don’t center properly. These defects not only affect comfort but also visual clarity. Implementing rigorous pre-shipment inspections and working with factories that have proven track records in eyewear production is essential.
Lack of Compliance with Optical Regulations
Eyeglasses are regulated medical devices in many countries. Sourcing non-compliant products—such as lenses that don’t meet UV protection or refractive accuracy requirements—can lead to regulatory penalties. Ensure suppliers provide necessary certifications (e.g., FDA, CE marking) and conduct third-party testing when required.
Inadequate Packaging and Lens Coating Quality
Low-cost suppliers may use flimsy packaging or apply inconsistent anti-reflective, scratch-resistant, or blue-light coatings. Poor coatings degrade quickly, leading to customer complaints. Specify coating standards and test samples before mass production.
Intellectual Property-Related Pitfalls
Unauthorized Use of Branded Designs
Many suppliers offer “inspired-by” versions of high-end designer frames. Sourcing such products exposes your business to trademark and design patent infringement claims. Even if the supplier claims the design is generic, copying distinctive elements of brands like Ray-Ban or Gucci can result in legal action and seizure of goods at customs.
Lack of Design Ownership and IP Clarity
When developing custom frames, failing to secure formal IP assignment agreements with suppliers can result in ownership disputes. A manufacturer might claim rights to the design or resell it to competitors. Always use contracts that explicitly transfer design rights to your company and include confidentiality clauses.
Counterfeit Goods and Grey Market Risks
Some suppliers may offer authentic-looking packaging and branding for well-known brands at suspiciously low prices. These are often counterfeit goods. Purchasing or distributing them—even unknowingly—can lead to legal liability, customs seizures, and reputational damage.
Weak Contractual Safeguards
Without strong contracts that address IP ownership, non-disclosure, and non-compete clauses, suppliers may replicate your designs for other clients. Ensure all agreements are legally enforceable in the supplier’s jurisdiction and consider involving legal counsel with international trade experience.
Mitigation Strategies
To avoid these pitfalls, conduct thorough due diligence on suppliers, request product samples for testing, audit manufacturing facilities when possible, and work with legal experts to protect your IP. Building long-term relationships with reputable, compliant manufacturers is key to sustainable and safe eyewear sourcing.

Logistics & Compliance Guide for Eyeglasses
Proper logistics and compliance are essential for the safe, legal, and efficient distribution of eyeglasses across international and domestic markets. This guide outlines key considerations for manufacturers, distributors, and retailers involved in the eyeglass supply chain.
Product Classification and Regulatory Requirements
Eyeglasses are generally classified as medical devices or consumer optical products, depending on their intended use and jurisdiction. Correct classification determines compliance obligations.
- United States (FDA): Prescription eyeglasses are considered Class I medical devices and are exempt from premarket notification (510(k)) but must comply with registration, labeling, and quality system regulations (QSR).
- European Union (EU MDR): Eyeglasses intended to correct vision are classified as Class I under the Medical Device Regulation (EU) 2017/745. They require CE marking, technical documentation, and adherence to essential safety and performance requirements.
- Other Regions: Countries such as Canada (Health Canada), Australia (TGA), and Japan (PMDA) have specific frameworks for optical devices. Always verify local classification and registration requirements.
Labeling and Packaging Compliance
Accurate labeling is critical for regulatory compliance and consumer safety.
- Mandatory Information:
- Manufacturer name and address
- Product identification (model, lens power, frame size)
- CE mark (EU), FDA establishment registration number (US), or equivalent
- Intended use statement
- Warnings and instructions for use (if applicable)
- Language Requirements: Labels and instructions must be in the official language(s) of the destination country.
- Packaging: Must protect against damage during transit and comply with environmental regulations (e.g., recyclable materials in the EU under Packaging Waste Directives).
Import/Export Documentation
Ensure all required documentation is prepared to avoid customs delays.
- Commercial Invoice: Details product description, value, quantity, and Harmonized System (HS) code.
- Packing List: Includes weight, dimensions, and item breakdown per shipment.
- Certificate of Origin: Required for preferential tariffs under trade agreements.
- Regulatory Certifications: Include copies of CE certificates, FDA registration, or other applicable approvals.
- HS Code Example: 9004.10 – Eyeglasses, goggles, and similar appliances correcting or protecting vision.
Shipping and Handling Considerations
Protect eyeglasses from damage during transit with appropriate logistics practices.
- Packaging Standards: Use rigid boxes, protective inserts, and cushioning to prevent lens scratches or frame deformation.
- Temperature and Humidity: Avoid extreme conditions that may affect lens coatings or frame materials (e.g., acetate warping).
- Shipping Modes: Choose reliable carriers with tracking; consider air freight for high-value or time-sensitive shipments.
- Insurance: Insure shipments against loss, theft, or damage, especially for international deliveries.
Customs Clearance and Duties
Understand tariff structures and import duties applicable to eyeglasses.
- Duty Rates: Vary by country and material composition (e.g., plastic vs. metal frames). Some regions offer reduced or zero tariffs for optical goods.
- Import Licenses: Generally not required for eyeglasses, but verify with local customs authorities.
- Value-Added Tax (VAT): Applicable in many countries (e.g., 20% standard rate in the UK). Collect and remit as required.
Quality and Safety Standards
Adhere to international standards to ensure product safety and interoperability.
- ISO 16012: Specifies requirements for spectacle frames.
- ISO 8980: Covers uncut finished spectacle lenses (transmittance, refractive power, etc.).
- EN 14139 (EU): Personal eye protection – prescription lenses.
- FDA 21 CFR Part 801: Labeling requirements for medical devices in the US.
Post-Market Surveillance and Reporting
Monitor product performance and comply with adverse event reporting.
- Incident Reporting: Report serious incidents or malfunctions to relevant authorities (e.g., FDA MedWatch, EU Vigilance System).
- Field Safety Notices: Issue corrective actions if defects are identified post-distribution.
- Product Recall Plan: Establish procedures for rapid and effective recalls if necessary.
Sustainability and Environmental Compliance
Address environmental responsibilities throughout the product lifecycle.
- WEEE (EU): Eyeglasses are generally exempt, but electronic components (e.g., smart glasses) may fall under WEEE.
- REACH and RoHS: Comply with restrictions on hazardous substances in materials (e.g., lead in solder, phthalates in plastics).
- Extended Producer Responsibility (EPR): Register and report under packaging EPR schemes in applicable countries.
Conclusion
Successfully managing the logistics and compliance of eyeglasses requires attention to regulatory details, accurate documentation, and protective shipping practices. By following this guide, stakeholders can ensure smooth global distribution while maintaining legal and safety standards. Always consult local regulatory authorities or legal counsel for jurisdiction-specific requirements.
In conclusion, sourcing eyeglass manufacturers requires a strategic approach that balances quality, cost, scalability, and reliability. Whether sourcing domestically or internationally, it is essential to conduct thorough due diligence on potential manufacturers, evaluating factors such as production capabilities, material standards, compliance with industry regulations (such as FDA or CE marking), and ethical manufacturing practices. Building strong partnerships with manufacturers who demonstrate consistent quality control, timely delivery, and the ability to accommodate customization is key to long-term success.
Additionally, leveraging trade shows, online B2B platforms, and industry referrals can help identify reputable suppliers. Communication, clear specifications, and sample testing are critical steps before committing to large-scale production. Ultimately, the right manufacturing partner not only meets current business needs but also supports future growth and innovation in the competitive eyewear market.