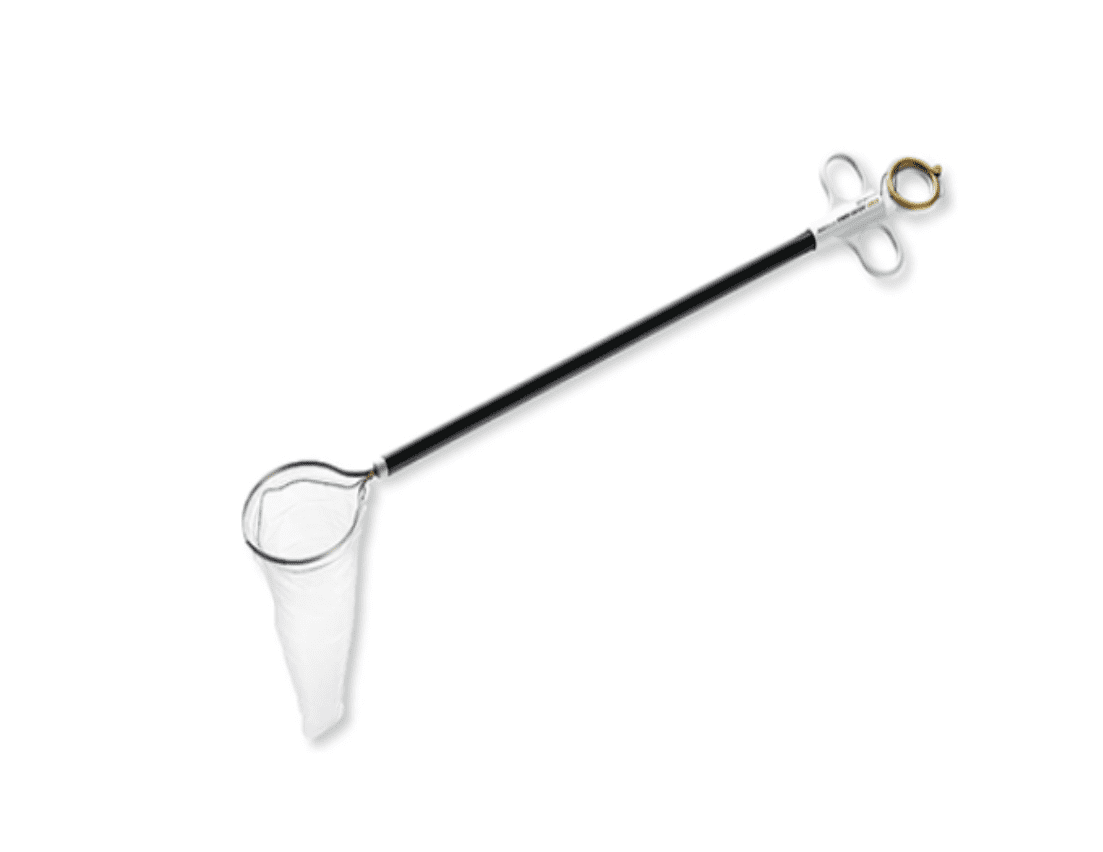
The global endocatch bag market is experiencing robust growth, driven by the rising adoption of minimally invasive surgical procedures and increasing demand for efficient specimen retrieval systems. According to Mordor Intelligence, the market is projected to grow at a CAGR of over 7.5% from 2023 to 2028, with expanding applications in oncology, bariatric, and colorectal surgeries. This growth is further supported by advancements in biocompatible materials and the growing emphasis on reducing intraoperative contamination risks. As healthcare providers prioritize patient safety and procedural efficiency, the demand for high-quality endocatch bags has intensified, fostering a competitive landscape among manufacturers. In this evolving market, six key players have distinguished themselves through innovation, regulatory compliance, and global distribution networks, making them leaders in the endocatch bag industry.
Top 6 Endocatch Bag Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Endo Bag Series
Domain Est. 2002
Website: unimaxmeds.com
Key Highlights: Detachable (retrieval) bag, size available for 5mm, 10mm, 15mm trocar. Unique patented technique for quick introduction and easy detachment of the bag….
#2 Premium Endocatch Bag Manufacturer
Domain Est. 2019
Website: segimedical.com
Key Highlights: The Endocatch bag is a sterile, single-use medical device made from a durable, transparent material that allows surgeons to capture and remove tissue samples ……
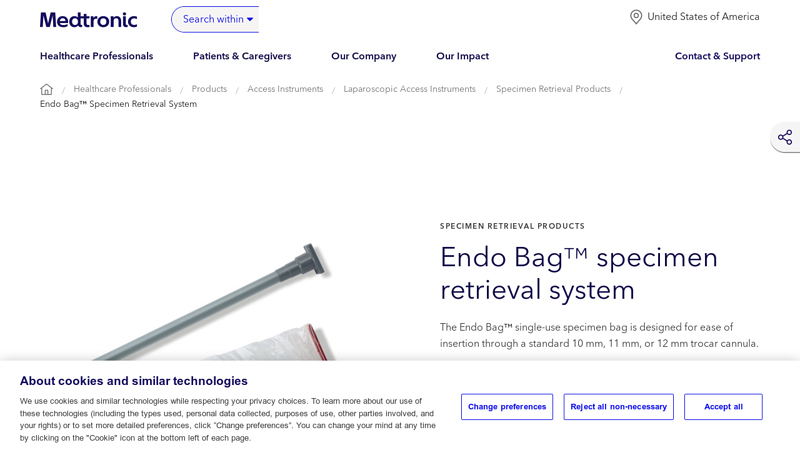
#3 Endo Bag™ Specimen Retrieval System
Domain Est. 1990
Website: medtronic.com
Key Highlights: The Endo Bag™ single-use specimen bag is designed for ease of insertion through a standard 10 mm, 11 mm, or 12 mm trocar cannula….
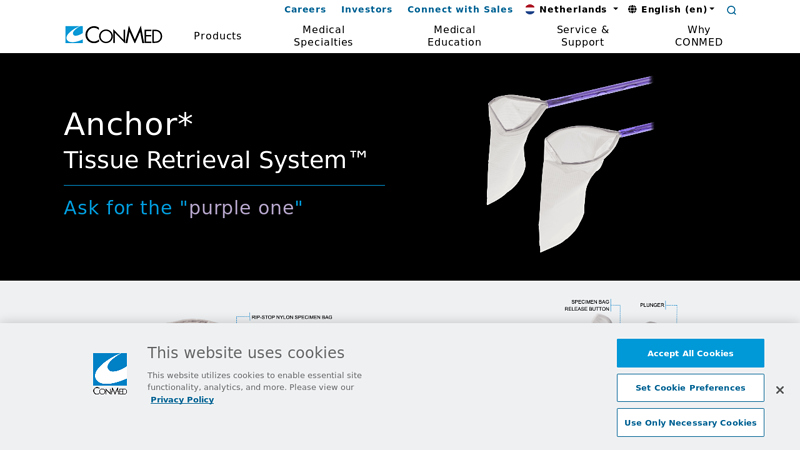
#4 Anchor Tissue Retrieval System
Domain Est. 1995
Website: conmed.com
Key Highlights: The Anchor Tissue Retrieval System is a comprehensive, premium medical specimen bag offering that provides safe and effective surgical specimen retrieval ……
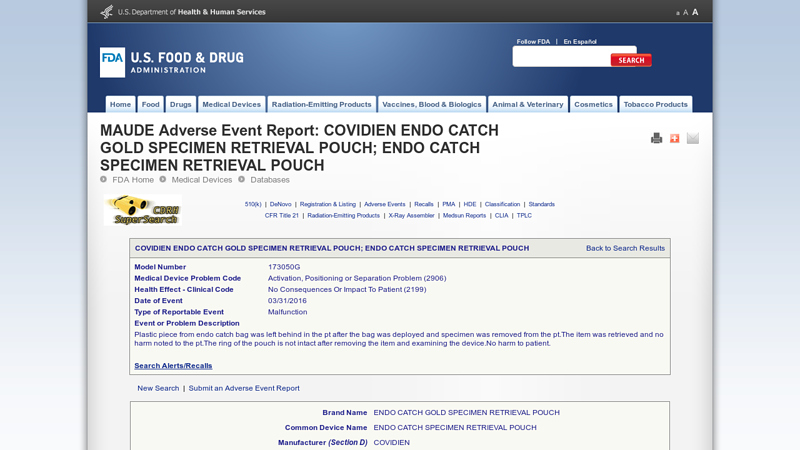
#5 COVIDIEN ENDO CATCH GOLD SPECIMEN RETRIEVAL POUCH
Domain Est. 2000
Website: accessdata.fda.gov
Key Highlights: Plastic piece from endo catch bag was left behind in the pt after the bag was deployed and specimen was removed from the pt.The item was retrieved and no ……
#6 EzCatch ® – Tissue Retrieval Bag
Domain Est. 2012
Website: veolmedtech.com
Key Highlights: The EzCatch Tissue Retrieval Bag is a secure, cost-effective solution for tissue extraction. These Endocatch bags are available to suit a variety of surgical…
Expert Sourcing Insights for Endocatch Bag

H2: Market Trends for Endocatch Bags in 2026
The global endocatch bag market is poised for substantial growth and transformation by 2026, driven by advancements in minimally invasive surgery, rising surgical volumes, and increasing emphasis on patient safety and procedural efficiency. As healthcare systems continue to adopt laparoscopic and robotic-assisted techniques across oncology, urology, and gynecology, demand for high-performance specimen retrieval devices—such as endocatch bags—is expected to rise significantly.
1. Rising Adoption of Minimally Invasive Surgeries (MIS)
One of the primary drivers of the endocatch bag market is the global shift toward minimally invasive procedures. By 2026, the number of laparoscopic and robotic surgeries is projected to increase due to shorter recovery times, reduced hospital stays, and lower risk of complications. Endocatch bags play a critical role in these procedures by safely containing and removing excised tissue, particularly in cancer resections where tumor spillage must be avoided. This growing surgical preference is expected to boost demand for advanced endocatch solutions.
2. Technological Innovations and Product Differentiation
Manufacturers are focusing on innovation to differentiate their endocatch bags in a competitive market. Anticipated trends for 2026 include the development of bags with enhanced tear resistance, antimicrobial coatings, larger capacity, and improved sealing mechanisms. Some next-generation models may incorporate smart features such as RFID tagging for specimen tracking or integration with robotic surgical platforms. These innovations aim to improve surgical outcomes and compliance with regulatory standards.
3. Expansion in Emerging Markets
Emerging economies in Asia-Pacific, Latin America, and parts of Africa are expected to witness accelerated growth in the endocatch bag market by 2026. Increased healthcare spending, improved access to surgical technologies, and rising awareness of oncology care are contributing factors. Local manufacturing and strategic partnerships with multinational medical device companies are likely to expand distribution networks and reduce costs, making endocatch bags more accessible in these regions.
4. Regulatory and Safety Standards
Regulatory scrutiny around medical devices, particularly those used in oncologic procedures, is intensifying. By 2026, compliance with standards such as ISO 13485 and FDA guidelines will be critical for market entry and sustainability. Endocatch bags may face stricter requirements regarding sterility, biocompatibility, and performance validation. Manufacturers investing in robust quality management systems and clinical evidence will gain a competitive edge.
5. Competitive Landscape and Market Consolidation
The endocatch bag market is moderately consolidated, with key players including Medtronic, Boston Scientific, CONMED Corporation, and Stryker. By 2026, further consolidation through mergers and acquisitions is anticipated as companies seek to broaden their portfolios in surgical devices. Additionally, niche players may emerge focusing on specialty applications, such as bariatric or pediatric surgeries, fostering innovation and segment-specific growth.
6. Impact of Robotic Surgery Growth
With the rapid adoption of robotic platforms like the da Vinci Surgical System, there is a growing need for compatible endocatch bags designed for precise manipulation through narrow trocars. By 2026, demand for robotic-specific retrieval systems is expected to rise, encouraging product redesigns that enhance dexterity, visibility, and ease of use within robotic workflows.
Conclusion
By 2026, the endocatch bag market will be shaped by the convergence of technological advancement, regulatory rigor, and expanding surgical applications. With increased focus on patient safety, efficiency, and global accessibility, the market is projected to grow steadily, offering significant opportunities for innovation and strategic expansion. Stakeholders who align with evolving clinical needs and invest in high-quality, differentiated products are likely to lead the market in this dynamic landscape.

Common Pitfalls Sourcing Endocatch Bags (Quality, IP)
Sourcing Endocatch bags—commonly used in laparoscopic and robotic surgical procedures for specimen retrieval—can present significant challenges, particularly concerning product quality and intellectual property (IP) rights. Failure to address these issues can lead to regulatory non-compliance, patient safety risks, and legal exposure. Below are key pitfalls to avoid:
1. Compromised Product Quality
Sourcing low-cost Endocatch bags from unverified suppliers often results in substandard materials and manufacturing processes. Poor-quality bags may tear during specimen retrieval, risking tissue contamination or spillage, which increases the chance of tumor seeding in oncologic cases. Buyers must verify compliance with international standards such as ISO 13485 and ensure biocompatibility testing (e.g., ISO 10993) has been performed.
2. Lack of Regulatory Approval
Many suppliers, especially in unregulated markets, offer Endocatch bags without proper regulatory clearance (e.g., FDA 510(k), CE marking). Using non-compliant devices can expose healthcare providers and procurement departments to liability and may result in surgical complications or facility audits.
3. Intellectual Property Infringement
Endocatch is a trademarked product originally developed by Medtronic. Sourcing “generic” or “compatible” versions that closely mimic the design and branding of the original may infringe on patents, trademarks, or design rights. Purchasing such products—even unknowingly—can lead to legal action against both the supplier and end-user institutions.
4. Inadequate Supplier Vetting
Procurement teams may focus solely on cost and availability, overlooking critical due diligence. Suppliers should provide evidence of legitimate manufacturing authorization, quality management systems, and IP clearance. Failure to conduct audits or request documentation increases exposure to counterfeit or unauthorized products.
5. Inconsistent Performance and Sizing
Low-quality or non-certified Endocatch bags may exhibit inconsistent performance, such as unreliable closure mechanisms or inadequate bag strength. Additionally, improper sizing can limit clinical utility, requiring multiple bag types and increasing inventory complexity.
6. Supply Chain Transparency Issues
Opaqueness in the supply chain—such as undisclosed manufacturing locations or third-party rebranding—can mask quality and IP risks. Transparent sourcing with traceability from manufacturer to end-user is essential for risk mitigation.
To avoid these pitfalls, organizations should prioritize suppliers with proven regulatory compliance, conduct thorough IP assessments, and implement stringent quality control protocols during procurement.

Logistics & Compliance Guide for Endocatch Bag
This guide provides essential information for the proper handling, transportation, storage, and regulatory compliance of the Endocatch Bag, a medical device used in urological and laparoscopic procedures for specimen retrieval.
Product Overview
The Endocatch Bag is a sterile, single-use medical device designed to contain and remove tissue specimens during minimally invasive surgeries. It is typically introduced through a trocar port, deployed under endoscopic visualization, used to capture resected tissue, and then withdrawn while maintaining specimen containment to prevent spillage and contamination.
Regulatory Classification
The Endocatch Bag is classified as a Class II medical device under the U.S. Food and Drug Administration (FDA) regulations (product code FKA). It is also CE marked in accordance with the Medical Device Regulation (MDR) EU 2017/745 in the European Union, indicating conformity with health, safety, and environmental protection standards.
Intended Use
The device is intended for the safe retrieval of tissue specimens during endoscopic or laparoscopic procedures, including but not limited to nephrectomy, prostatectomy, and cystectomy. It is not intended for long-term implantation or use with morcellation devices unless specifically indicated by the manufacturer.
Storage Conditions
Store the Endocatch Bag in its original packaging in a dry, cool environment with temperatures between 15°C and 30°C (59°F to 86°F). Protect from direct sunlight, excessive heat, moisture, and physical damage. Do not freeze. Ensure storage areas are clean and free from contaminants.
Handling and Transportation
During transportation, ensure cartons are stacked securely and protected from extreme temperatures, humidity, and physical impact. Use validated shipping containers when transporting over long distances or in variable climates. Maintain the cold chain if required by the manufacturer. Do not transport with hazardous materials.
Shelf Life
The Endocatch Bag has a labeled shelf life, typically ranging from 2 to 3 years from the date of manufacture. Always verify the expiration date printed on the packaging before use. Do not use expired devices.
Sterility and Packaging
The device is supplied sterile via ethylene oxide (EtO) or gamma irradiation, as indicated on the packaging. Inspect the packaging prior to use for breaches, tears, or signs of tampering. Do not use if the sterile barrier is compromised. Use aseptic technique during handling in the clinical environment.
Labeling Requirements
Primary packaging must include:
– Product name and model number
– Lot number
– Expiration date
– Sterile indicator
– Single-use symbol
– Manufacturer name and address
– Regulatory markings (e.g., CE, FDA)
– UDI (Unique Device Identification) barcode
Ensure all labels remain legible and intact throughout storage and distribution.
Import/Export Compliance
When importing or exporting the Endocatch Bag, comply with the regulatory requirements of the destination country. This may include:
– Valid Certificate of Free Sale (CFS)
– Import licenses or permits
– Customs documentation with accurate HS codes (e.g., 9018.90 for surgical instruments)
– Language-specific labeling and instructions for use (IFU)
Verify compliance with local health authorities (e.g., ANVISA in Brazil, PMDA in Japan, TGA in Australia).
Distribution Chain Management
Distributors must be authorized and comply with applicable medical device regulations (e.g., FDA 21 CFR Part 809, EU MDR Article 15). Maintain traceability through proper recordkeeping of distribution, including lot numbers and customer information, to support potential recalls or field actions.
Post-Market Surveillance
Report any adverse events, device malfunctions, or complaints to the manufacturer and relevant regulatory bodies (e.g., FDA MedWatch, EUDAMED) per local requirements. Maintain records of all incidents and corrective actions.
Disposal
Dispose of used Endocatch Bags in accordance with local biohazard and medical waste disposal regulations. As a single-use device, it must not be reprocessed or reused.
Training and Documentation
Ensure all personnel involved in handling, storing, or using the Endocatch Bag are trained on proper procedures. Maintain training records and up-to-date copies of the Instructions for Use (IFU), regulatory certificates, and quality documentation.
Conclusion for Sourcing Endocatch Bags:
After a thorough evaluation of potential suppliers, product quality, regulatory compliance, cost-effectiveness, and supply chain reliability, it is concluded that sourcing Endocatch bags requires a strategic approach focused on balancing clinical performance with operational efficiency. Prioritizing vendors that adhere to international quality standards (such as ISO 13485 and FDA clearance), offer proven biocompatibility and burst strength, and provide consistent delivery timelines is essential to ensure patient safety and procedural success.
Additionally, establishing long-term partnerships with suppliers who demonstrate innovation, scalability, and responsive customer support will enhance supply continuity and enable adaptation to evolving surgical demands. Competitive pricing should be pursued without compromising on sterility, material integrity, or design functionality.
In summary, the optimal sourcing strategy involves selecting a reputable supplier that meets stringent quality and regulatory requirements while offering reliable logistics and value-added services, ultimately supporting high standards of patient care and operational excellence in urological and laparoscopic procedures.