The global electrolyte powder market is experiencing robust growth, driven by increasing consumer awareness of hydration and the rising demand for performance-enhancing and functional beverages. According to Grand View Research, the global sports nutrition market—of which electrolyte powders are a key segment—was valued at USD 18.5 billion in 2022 and is expected to expand at a compound annual growth rate (CAGR) of 8.2% from 2023 to 2030. Similarly, Mordor Intelligence projects that the global electrolyte drinks market will register a CAGR of over 6.5% during the forecast period 2023–2028, fueled by health-conscious lifestyles, growing fitness trends, and expanded product availability in emerging economies. With demand surging, manufacturers are scaling up bulk production capabilities to meet commercial needs across sports, wellness, and clinical hydration sectors. As sourcing consultants and brands seek reliable supply partners, identifying high-capacity, quality-focused electrolyte powder manufacturers has become critical. Based on production scale, certifications, global reach, and compliance with regulatory standards, here are the top nine bulk electrolyte powder manufacturers shaping the industry’s supply chain.
Top 9 Electrolyte Powder Bulk Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Liquid I.V.
Domain Est. 2013
Website: liquid-iv.com
Key Highlights: Free delivery over $40 · 60-day returnsLiquid I.V. is a great-tasting electrolyte drink mix powered by Cellular Transport Technology (CTT)®️ ️to hydrate faster and more efficientl…
#2 SOS Hydration
Domain Est. 2014
Website: soshydration.com
Key Highlights: Rescue yourself from dehydration with SOS Daily Hydration! SOS is rapid hydration with vitamins, minerals, electrolytes, and thirst-quenching flavors…with …Missing: bulk manuf…
#3 Electrolit®
Domain Est. 2000
Website: electrolit.com
Key Highlights: Our products are formulated with 5 vital ions to support your body, hydrating you faster than water alone. Made with hard-working ingredients….
#4 Cure
Domain Est. 2017
Website: curehydration.com
Key Highlights: Cure is a premium, hydrating electrolyte drink mix made with organic coconut water, pink Himalayan salt and no added sugar. Based on the WHO’s ORS (Oral ……
#5 Supplement Formulation & Powdered Supplements
Domain Est. 2021
Website: halohydration.com
Key Highlights: Experts in powdered supplements and supplement formulation, we specialize in single serve white label electrolyte powder, with low MOQs….
#6 Redmond Re-Lyte
#7 DASH Hydrate
Domain Est. 2023
Website: dashhydration.com
Key Highlights: Free delivery over $40 30-day returnsDASH Hydrate, created by First Responders in the USA, is an electrolyte powder hydration drink mix made for rapid hydration & recovery with no …
#8 Best Electrolyte Powder & Drinks for Ultimate Hydration
Domain Est. 2003
#9 Superieur Electrolytes
Domain Est. 2015
Website: superieurelectrolytes.com
Key Highlights: A light, refreshing electrolyte drink with zero sugar. It’s the most complete electrolyte formula on the market and includes sea salt, sea minerals and real ……
Expert Sourcing Insights for Electrolyte Powder Bulk

Market Trends Analysis for Electrolyte Powder Bulk (2026) – Hydrogen Economy (H₂) Context
As we approach 2026, the global market for electrolyte powder in bulk is undergoing significant transformation, driven by shifting consumer health behaviors, advancements in sports nutrition, and emerging industrial applications—particularly those linked to the growing hydrogen (H₂) economy. While electrolyte powders are traditionally associated with hydration and wellness, their role is expanding into high-tech and energy sectors, especially in conjunction with hydrogen production via water electrolysis. This analysis explores key 2026 market trends for bulk electrolyte powders, with a focus on their intersection with hydrogen (H₂) technologies.
1. Rising Demand in Green Hydrogen Production (H₂ via Electrolysis)
One of the most impactful trends shaping the electrolyte powder market in 2026 is its indirect but critical role in green hydrogen (H₂) generation.
- Proton Exchange Membrane (PEM) and Alkaline Electrolyzers require highly pure electrolytes (such as potassium hydroxide, KOH, or specialized ionic compounds) to facilitate water splitting into hydrogen and oxygen.
- Bulk electrolyte powders—particularly high-purity KOH and advanced ion-conductive salts—are increasingly in demand as countries scale up green H₂ infrastructure under decarbonization mandates.
- The global push for carbon-neutral energy (e.g., EU Green Deal, U.S. Inflation Reduction Act) is accelerating investments in hydrogen hubs, boosting procurement of industrial-grade electrolytes.
Trend Impact (2026):
The industrial segment will account for over 25% of high-purity electrolyte powder demand, up from <10% in 2020. Suppliers are adapting formulations for electrochemical stability and scalability.
2. Dual-Use Electrolyte Powders: Consumer Health + Energy Tech
By 2026, a new category of multi-functional electrolyte powders is emerging, designed for both human consumption and industrial use:
- Innovative formulations incorporating magnesium, sodium, potassium, and buffering agents (e.g., citrates) are being optimized not just for hydration, but also for catalytic support in low-temperature electrolysis.
- R&D efforts are exploring bio-derived electrolytes (e.g., from agricultural waste) that can serve in both sports nutrition and sustainable H₂ systems.
- Startups are developing modular electrolyte supply chains that serve both wellness and clean energy markets, reducing costs through economies of scale.
Trend Impact (2026):
Cross-sector formulations could represent 15–20% of new product launches in the bulk electrolyte space.
3. Supply Chain Shifts & Geopolitical Factors
The geopolitical drive for energy independence and control over critical minerals is reshaping sourcing:
- Countries investing heavily in H₂ (Germany, Japan, Australia) are securing long-term contracts for lithium, potassium, and rare earth-based electrolytes, often in powder form for ease of transport and storage.
- China and the U.S. dominate production of high-purity KOH and lithium salts, leading to regionalization of supply chains to avoid bottlenecks.
- Environmental regulations are pushing manufacturers toward closed-loop recycling of spent electrolytes from hydrogen plants.
Trend Impact (2026):
Regional production hubs in North America and Europe will grow, reducing reliance on Asian imports by ~30%.
4. Sustainability & ESG Pressures
Environmental, Social, and Governance (ESG) criteria are now central to bulk electrolyte procurement:
- Green hydrogen producers demand certified sustainable electrolytes with low carbon footprints.
- Leading suppliers are adopting carbon labeling and third-party verification for bulk powder products.
- Water usage in electrolyte production is under scrutiny, prompting innovations in dry synthesis methods and solvent-free processing.
Trend Impact (2026):
Over 60% of B2B contracts in the EU and North America will require ESG compliance documentation for electrolyte powders.
5. Technological Innovation: Solid-State & Composite Electrolytes
While liquid electrolytes dominate, 2026 sees progress in solid-state and composite electrolyte powders:
- Research into ceramic-polymer hybrid powders for high-efficiency electrolyzers is accelerating, supported by government grants.
- These powders offer safer handling, longer shelf life, and compatibility with modular H₂ generators.
- Though still in early adoption, pilot projects in Japan and Germany are testing powder-based solid electrolytes in next-gen PEM systems.
Trend Impact (2026):
Niche but growing market segment; expected to reach $120M in annual sales by 2026, with >40% CAGR from 2023.
6. Market Size & Growth Projections (2026 Outlook)
- Global Bulk Electrolyte Powder Market (2026): Estimated at $2.8–3.2 billion, with a CAGR of 8.7% (2021–2026).
- Breakdown by Application:
- Sports & Wellness Nutrition: 55%
- Pharmaceuticals & Clinical Hydration: 15%
- Industrial (incl. H₂ electrolysis): 25%
- Others (Agriculture, Electronics): 5%
Note: The industrial/H₂ segment is the fastest-growing, projected to grow at 14% CAGR through 2026.
Conclusion
By 2026, the bulk electrolyte powder market is no longer confined to health and fitness. The rise of the hydrogen (H₂) economy is fundamentally reshaping demand, quality standards, and innovation trajectories. Companies that position themselves at the intersection of human health and clean energy—offering high-purity, sustainable, and multi-application electrolyte powders—will lead the next phase of market growth.
Strategic Recommendation:
Suppliers should invest in dual-certification processes (food-grade + industrial-grade), expand into green hydrogen supply chains, and leverage AI-driven formulation optimization to meet the dual demands of hydration and H₂ production.
Note: “H₂” in this context refers to molecular hydrogen and its role in the energy transition, not a company or product named H2.
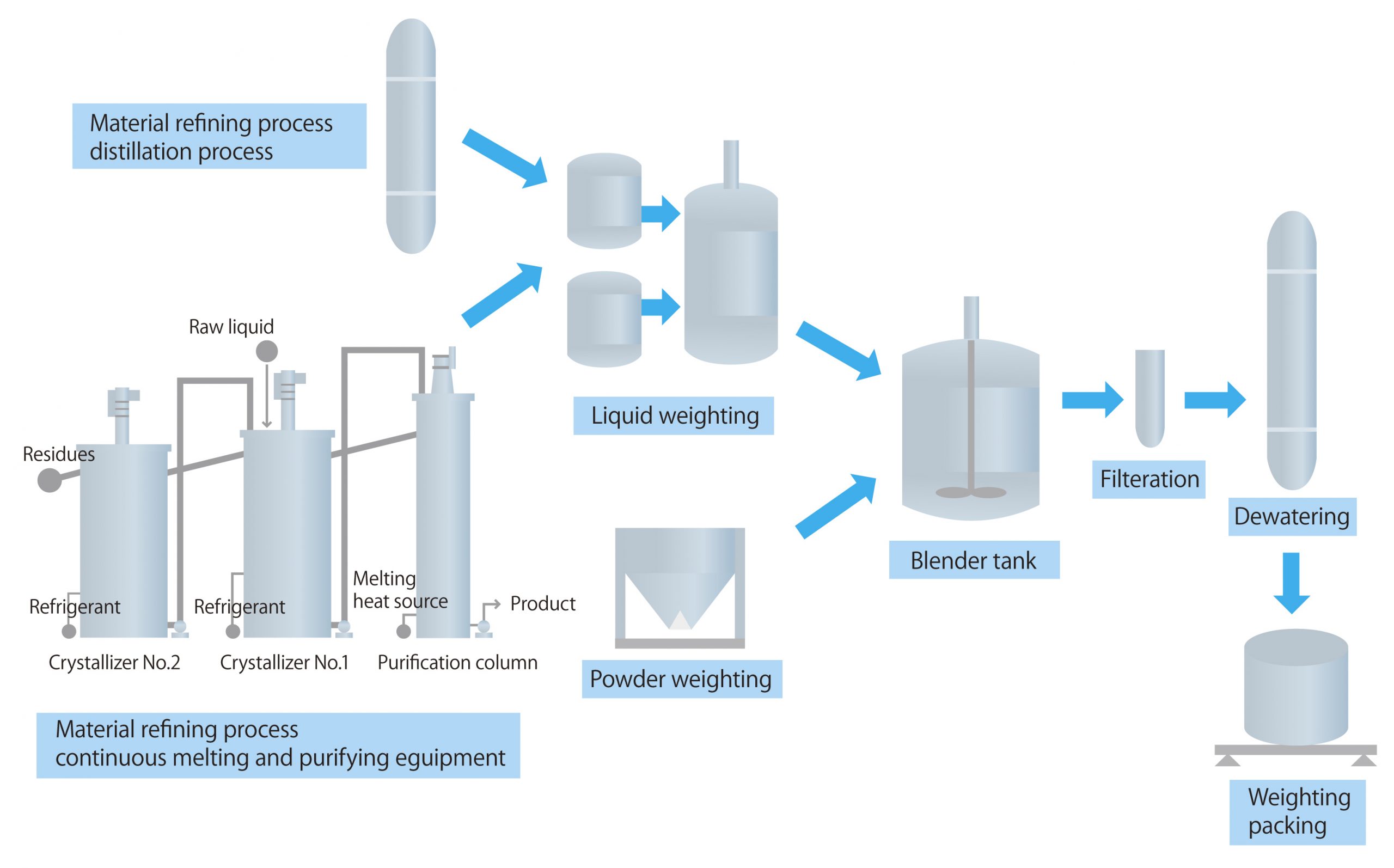
H2: Common Pitfalls When Sourcing Electrolyte Powder in Bulk – Quality & Intellectual Property (IP) Risks
Sourcing electrolyte powder in bulk offers significant cost advantages, but it comes with critical risks, particularly concerning quality consistency and intellectual property (IP) protection. Overlooking these pitfalls can lead to product failures, regulatory issues, brand damage, and legal disputes.
H3: Quality Pitfalls in Bulk Electrolyte Powder Sourcing
-
Inconsistent Raw Material Sourcing & Quality:
- Pitfall: Suppliers may source key electrolytes (sodium, potassium, magnesium, calcium, chloride, citrate, etc.) from multiple, unvetted sub-suppliers with varying purity standards, geographical origins, and processing methods. This leads to batch-to-batch variability in potency, solubility, taste, and color.
- Consequence: Your final product’s performance (hydration efficacy, taste profile) becomes unreliable, damaging consumer trust and potentially leading to complaints or returns.
-
Inadequate or Non-Standardized Manufacturing Processes:
- Pitfall: Lack of strict adherence to GMP (Good Manufacturing Practices) or cGMP (for regulated products like supplements/OTC drugs). Poor hygiene, cross-contamination risks, inconsistent mixing times/temperatures, or inadequate drying can compromise product safety and uniformity.
- Consequence: Risk of microbial contamination, uneven distribution of active ingredients (leading to under/over-dosing), clumping, or poor solubility. This can trigger recalls or regulatory action.
-
Insufficient or Inaccurate Testing & Certification:
- Pitfall: Relying solely on supplier-provided Certificates of Analysis (CoA) without verification. CoAs might be generic, outdated, or falsified. Lack of comprehensive testing for heavy metals (lead, arsenic, cadmium, mercury), microbial load (aerobic plate count, yeast/mold, pathogens), residual solvents, or identity/potency of all declared ingredients.
- Consequence: Introduction of harmful contaminants into your product, failure to meet regulatory requirements (e.g., FDA, EFSA, Health Canada), and potential health risks to consumers.
-
“Formula Drift” and Unapproved Substitutions:
- Pitfall: Suppliers may silently substitute ingredients (e.g., changing the source of citrate, using a different anti-caking agent, altering ratios) to cut costs or due to supply chain issues, without informing you or seeking approval.
- Consequence: Significant changes in taste, solubility, stability, efficacy, and potentially allergenicity (e.g., switching to an ingredient derived from a common allergen). Your product becomes inconsistent with its original specification and marketing claims.
-
Poor Packaging and Stability Issues:
- Pitfall: Inadequate packaging (e.g., moisture-permeable bags, lack of oxygen barriers) or storage conditions (high heat/humidity) during transit or at the supplier’s facility, leading to moisture absorption, clumping, degradation of sensitive ingredients (like certain vitamins often added), or loss of potency.
- Consequence: Shorter shelf life, product spoilage, customer dissatisfaction, and increased waste.
H3: Intellectual Property (IP) Pitfalls in Bulk Electrolyte Powder Sourcing
-
Lack of Clear IP Ownership and Confidentiality Agreements:
- Pitfall: Failing to establish upfront, legally binding agreements (like a robust Manufacturing Agreement or Supply Agreement with strong IP clauses and a comprehensive NDA) defining who owns the final product formula, any proprietary processes used, and how confidential information (your specific blend ratios, unique ingredient combinations, branding, customer data) will be protected.
- Consequence: The supplier may claim co-ownership of the formula, use your confidential blend to make identical or similar products for your competitors, or sell the information to others. You lose control over your unique selling proposition (USP).
-
Supplier Replication and “Knock-Off” Competition:
- Pitfall: Once a supplier manufactures your specific electrolyte powder blend, they possess the knowledge and capability to replicate it exactly or create a very similar product (“private label” or “white label”) for other buyers, including your direct competitors, often at lower prices.
- Consequence: Erosion of your market share, price competition on your own product, dilution of your brand’s uniqueness, and loss of competitive advantage. This is a major risk with “contract manufacturers” who also serve multiple clients.
-
Ambiguity in “Private Label” vs. “Custom” Manufacturing:
- Pitfall: Assuming a “custom” blend automatically grants exclusivity. Many suppliers offer “custom blends” but retain the right to produce the same custom blend for other customers unless explicitly prohibited by contract.
- Consequence: Your “custom” formula isn’t unique, negating the intended benefit of differentiation. The market becomes flooded with identical products.
-
Failure to Secure Process IP:
- Pitfall: If your product relies on a unique manufacturing process (e.g., a specific drying technique for better solubility, a proprietary coating for stability) developed or specified by you, failing to ensure the supplier cannot use or disclose that process for others.
- Consequence: Loss of a key technological advantage that competitors can quickly replicate.
Mitigation Strategies:
- Rigorous Due Diligence: Audit potential suppliers (onsite if possible), verify certifications (GMP, ISO, organic, non-GMO, etc.), and check references.
- Comprehensive Contracts: Use detailed agreements clearly defining:
- IP Ownership: Explicitly state that all IP (formula, process, branding) developed for or provided to the supplier belongs solely to you.
- Exclusivity/Non-Compete: Prohibit the supplier from manufacturing the exact same formula for any other party.
- Confidentiality: Enforce strict NDA terms covering all aspects of the formulation and business.
- Quality Specifications: Define exact ingredient specifications, tolerances, testing protocols (including required tests and frequency), and CoA requirements for every batch.
- Change Control: Require formal notification and your prior written approval for any change in raw materials, sources, or manufacturing processes.
- Audit Rights: Reserve the right to conduct regular audits of the supplier’s facility and records.
- Independent Testing: Implement a robust batch testing program using independent, accredited laboratories to verify CoAs and ensure ongoing compliance.
- Diversify Supply (Cautiously): While relying on a single supplier increases IP risk, having multiple pre-qualified suppliers (with identical, protected specs) can mitigate quality risks. Ensure each has the same stringent IP protections.
By proactively addressing these quality and IP pitfalls through careful supplier selection, robust legal agreements, and ongoing verification, you can secure a reliable, high-quality bulk electrolyte powder supply while protecting your brand’s value and competitive edge.

H2: Logistics & Compliance Guide for Electrolyte Powder (Bulk)
Handling and transporting bulk electrolyte powder requires strict adherence to logistics best practices and regulatory compliance to ensure product safety, quality, and legal conformity. This guide outlines key steps and requirements for the safe and compliant shipment, storage, and handling of bulk electrolyte powder.
H2.1: Product Classification & Regulatory Overview
Electrolyte powders typically contain ingredients such as sodium chloride, potassium chloride, magnesium citrate, calcium carbonate, and flavoring agents. While generally recognized as safe (GRAS) for human consumption, they must still comply with relevant regulations:
- FDA Compliance (U.S.): Subject to 21 CFR regulations for food or dietary supplements, depending on labeling and claims. If marketed as a supplement, must comply with DSHEA (Dietary Supplement Health and Education Act).
- EU Regulations: Must comply with EU Regulation (EC) No 1924/2006 (nutrition and health claims) and food safety standards under Regulation (EC) No 178/2002.
- Customs & Import: Accurate HS Code classification is essential (e.g., 2106.90 for food preparations not elsewhere specified). Provide certificates of analysis (CoA), certificates of free sale, and ingredient declarations where required.
- REACH/CLP (EU): If any component is classified as hazardous, SDS (Safety Data Sheet) under CLP regulation may be required, even for non-hazardous mixtures.
H2.2: Packaging & Labeling Requirements
Proper packaging ensures product integrity and regulatory compliance:
- Primary Packaging: Use food-grade, moisture-resistant, multi-wall poly-lined bags (e.g., 25–50 lb sacks) or bulk FIBCs (Flexible Intermediate Bulk Containers) rated for food contact (FDA 21 CFR 177.1520 compliant).
- Labeling:
- Product name and batch/lot number
- Net weight
- Manufacturer and distributor information
- Storage instructions (e.g., “Store in a cool, dry place”)
- Allergen statements (if applicable)
- Nutrition facts panel (if sold as food/supplement)
- Country of origin
- Bulk Shipping Labels: Include handling symbols (e.g., “Keep Dry,” “Do Not Stack”), hazard communication if applicable, and proper shipping name.
H2.3: Storage Conditions
To maintain quality and shelf life (typically 12–24 months):
- Environment: Store in a dry, temperature-controlled warehouse (15–25°C / 59–77°F recommended).
- Humidity: Maintain relative humidity below 60% to prevent caking or clumping.
- Pest & Contamination Control: Implement GMP (Good Manufacturing Practices) and HACCP protocols. Store off the floor on pallets and away from walls.
- Segregation: Keep separate from strong-smelling or hazardous materials.
H2.4: Transportation & Shipping
- Mode of Transport:
- Truck (Domestic): Use enclosed, dry vans with clean flooring. Avoid mixed loads with chemicals or perishables.
- Sea Freight (International): Use containerized shipping with desiccants to control moisture. Ensure containers are sealed and inspected for leaks.
- Air Freight: Possible for smaller volumes; ensure compliance with IATA regulations (usually non-hazardous, but verify classification).
- Documentation:
- Commercial invoice
- Packing list
- Bill of Lading (B/L) or Air Waybill (AWB)
- CoA and nutritional information
- Import permits (if required by destination country)
- Temperature Control: Not typically required unless specified, but avoid extreme temperatures during transit.
H2.5: Customs & Import Compliance
- HS Code: Confirm correct classification (e.g., 2106.90.9999 for other food preparations).
- FDA Prior Notice (U.S.): Required for food imports. Submit via FDA’s Prior Notice System Gateway (PNSG).
- EU Entry Requirements: Use the TRACES NT system for notifications if applicable.
- Labeling at Destination: Ensure final packaging meets local language and regulatory requirements.
H2.6: Safety & Handling
- Worker Safety: Provide PPE (gloves, masks) during handling to minimize inhalation of fine particles.
- SDS Availability: Maintain a Safety Data Sheet even if not classified as hazardous, for transparency and emergency response.
- Spill Management: In case of spills, collect dry powder carefully (avoid water if hygroscopic); dispose of per local waste regulations.
H2.7: Quality Assurance & Traceability
- Batch Tracking: Implement a system to track raw materials and finished batches for full traceability.
- Testing: Conduct routine microbiological and heavy metal testing as per internal quality standards or customer requirements.
- Audits: Prepare for third-party audits (e.g., BRCGS, SQF, or FDA inspections).
H2.8: Sustainability & Disposal
- Recyclable Packaging: Use recyclable or biodegradable materials where feasible.
- Waste Management: Unused or expired product should be disposed of in accordance with local environmental regulations. Do not dump in waterways.
Conclusion
Proper logistics and compliance for bulk electrolyte powder involve coordinated efforts across packaging, labeling, storage, transport, and regulatory documentation. Adhering to food safety standards and international trade requirements ensures smooth operations and protects brand integrity. Always consult legal and regulatory experts for destination-specific compliance.
— End of Guide —
Conclusion: Sourcing Electrolyte Powder in Bulk
Sourcing electrolyte powder in bulk offers significant advantages for businesses, institutions, and organizations looking to meet high-volume demand efficiently and cost-effectively. By purchasing in bulk, buyers benefit from reduced per-unit costs, consistent supply, and improved operational scalability. Key considerations such as product quality, ingredient transparency, compliance with regulatory standards (e.g., FDA, cGMP, NSF), and supplier reliability are essential to ensuring a safe and effective final product.
Additionally, evaluating customization options, packaging flexibility, lead times, and logistical support from suppliers enhances the overall value of bulk sourcing. Building strong relationships with reputable manufacturers or distributors not only ensures a steady supply chain but also supports long-term growth and brand integrity.
In conclusion, with diligent supplier vetting and strategic planning, sourcing electrolyte powder in bulk is a smart investment that supports quality, economies of scale, and customer satisfaction—making it an ideal solution for hydration-focused products in the sports, wellness, and healthcare markets.