The global orthopedic braces and supports market, which includes drop foot braces and specialized footwear, is experiencing steady growth driven by rising incidences of neurological disorders, musculoskeletal conditions, and an aging population. According to Mordor Intelligence, the global orthopedic braces and supports market was valued at USD 3.3 billion in 2023 and is projected to grow at a CAGR of 5.8% from 2024 to 2029. This growth is further fueled by increasing demand for mobility-assistive devices, advancements in lightweight orthotic materials, and rising awareness about early rehabilitation. As foot drop—a common gait disorder caused by nerve injury, stroke, or multiple sclerosis—affects millions worldwide, the need for effective, comfortable, and biomechanically sound braces and compatible footwear has intensified. In response, manufacturers are innovating with ergonomic designs, customizable features, and integrated shoe-brace systems. Based on market presence, product innovation, and clinical adoption, the following nine companies have emerged as leading manufacturers of drop foot braces and orthopedic footwear, shaping the future of mobility solutions.
Top 9 Drop Foot Braces Shoes Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 AFO Foot Drop Brace, Empowering A New Experience In Gait …
Domain Est. 2022
Website: bose-grow.com
Key Highlights: On December 8, 2025, BOSEGROW MEDICAL, a orthopedic brace manufacturer in China, formally unveiled its brand-new FS-05 AFO Foot Drop Brace….
#2 Foot-Up®
Domain Est. 1996
Website: ossur.com
Key Highlights: Foot-Up® is a lightweight ankle drop foot brace that offers dynamic support for drop foot or related disorders that require dorsiflexion support….
#3 Cascade Dafo
Domain Est. 2005
Website: cascadedafo.com
Key Highlights: Makers of Dynamic Ankle Foot Orthoses for improved mobility, including Fast Fit Chipmunk shoe inserts. JumpStart AFOs, DAFO 4 and DAFO Tami2 custom ……
#4 Allard USA
Domain Est. 2005
Website: allardusa.com
Key Highlights: ToeUP™ Textile Foot Drop Orthosis. ToeUP is a textile ankle-foot orthosis (AFO) designed for patients with milder forms of drop foot. It features various ……
#5 Push ortho Ankle Foot Orthosis (AFO)
Domain Est. 2011
Website: bracelab.com
Key Highlights: With a new, innovative drop foot brace design that allows for a natural gait, the Push AFO brace makes safe and efficient walking possible again….
#6 The Elevate 360°® Drop Foot Brace
Domain Est. 2011
Website: footscientific.com
Key Highlights: The Elevate Drop Foot Brace was developed to create a drop foot brace that is discrete yet versatile, and stable yet comfortable….
#7 Elevate Movement
Domain Est. 2017
Website: elevatemovement.com
Key Highlights: Our products combine decades of orthopedic bracing expertise with cutting edge design to enable drop foot users to live healthier lives, one step at a time….
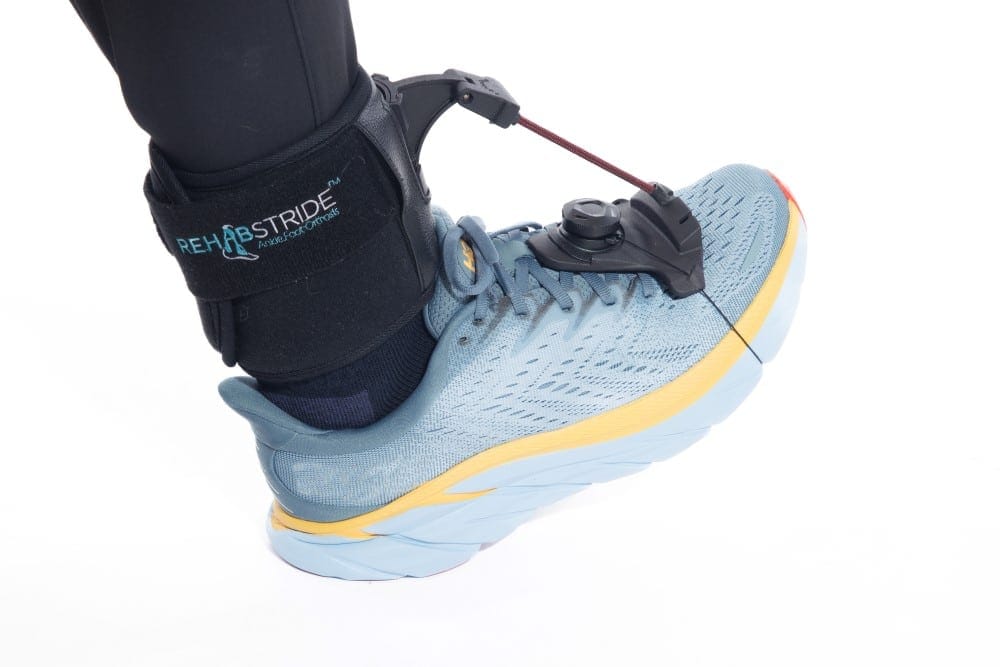
#8 RehabStride™ AFO Brace for Foot Drop
Domain Est. 2023
Website: rehabstride.com
Key Highlights: RehabStride AFO brace for foot drop – seamlessly adapts to all shoes and utilizes a personalized tension-adjustable cable system, enabling normalized gait….
#9 AFO Ankle Foot Orthotics, Best Drop Foot Braces, StepSmart Brace
Domain Est. 2006
Website: insightfulproducts.com
Key Highlights: Free delivery 30-day returnsStarting at $199 · Fits inside shoe · Standard Calf Section · Top Cover Available · Hip Square Available · Compatible with Cuffed Calf Section….
Expert Sourcing Insights for Drop Foot Braces Shoes
2026 Market Trends for Drop Foot Braces Shoes
Rising Prevalence of Neurological and Musculoskeletal Disorders
The global market for drop foot braces shoes is expected to grow significantly by 2026, driven primarily by the increasing incidence of neurological conditions such as stroke, multiple sclerosis, cerebral palsy, and peripheral nerve injuries. An aging population, particularly in North America and Europe, contributes to higher rates of stroke and spinal cord injuries, both common causes of foot drop. Additionally, rising diabetes prevalence globally leads to more cases of diabetic neuropathy, further expanding the target patient pool. As healthcare systems improve diagnosis and long-term rehabilitation strategies, demand for assistive devices like drop foot braces embedded in shoes will continue to grow.
Advancements in Smart and Wearable Technology Integration
By 2026, smart technology integration is poised to revolutionize drop foot brace shoes. Manufacturers are increasingly incorporating sensors, microprocessors, and AI-driven algorithms to create responsive, gait-adaptive footwear. These intelligent systems detect the user’s gait phase in real time and deliver timely dorsiflexion assistance via lightweight actuators or Functional Electrical Stimulation (FES). Brands are partnering with med-tech firms to offer apps that track mobility metrics, monitor compliance, and allow remote adjustments by clinicians. This shift toward personalized, data-driven rehabilitation enhances user outcomes and is expected to dominate premium segments of the market.
Shift Toward Aesthetic and Lifestyle-Oriented Designs
Consumer demand for medical devices that blend seamlessly into daily life is reshaping product development. In 2026, drop foot braces shoes are increasingly designed to resemble conventional athletic or casual footwear, moving away from bulky, clinical appearances. Leading companies are investing in ergonomic, lightweight materials such as carbon fiber and advanced polymers to improve comfort and discretion. Collaborations with fashion designers and footwear brands are anticipated to expand, offering stylish options in various sizes, colors, and fits. This trend addresses psychological barriers to adoption, especially among younger users and those concerned with social stigma.
Growth in Home-Based Rehabilitation and Telehealth
The expansion of telehealth services and home-based rehabilitation programs will influence how drop foot braces shoes are prescribed and monitored. By 2026, more patients are expected to receive remote consultations and digital prescriptions, supported by virtual physical therapy sessions. Integrated wearables in brace shoes will transmit gait data directly to healthcare providers, enabling timely interventions. This trend supports early adoption, improves adherence, and reduces hospital visits—making treatment more accessible, especially in rural or underserved areas.
Regulatory and Reimbursement Landscape Evolution
Regulatory approvals and insurance coverage will play a critical role in market accessibility. By 2026, streamlined FDA and CE certification processes for Class I and II medical devices may accelerate product launches. Simultaneously, increasing advocacy and clinical evidence supporting the efficacy of drop foot braces could lead to broader insurance reimbursement, particularly in the U.S. and parts of Europe. However, disparities in healthcare coverage across regions may still limit market penetration in low- and middle-income countries, creating opportunities for cost-effective, simplified models.
Expansion of Emerging Markets
While North America and Europe remain dominant markets, Asia-Pacific and Latin America are expected to witness the fastest growth by 2026. Rising healthcare spending, improving awareness, and expanding rehabilitation infrastructure in countries like China, India, and Brazil will drive adoption. Local manufacturing and partnerships with global brands will help reduce costs and tailor products to regional needs, such as climate-appropriate materials and culturally acceptable designs.
Sustainability and Customization Trends
Environmental consciousness is influencing material choices, with a growing preference for recyclable, biodegradable, or sustainably sourced components. Additionally, 3D printing technologies are enabling highly customized orthotic insoles and braces that match individual biomechanics, enhancing comfort and effectiveness. By 2026, on-demand manufacturing and digital fitting tools are likely to become standard, reducing waste and improving patient satisfaction.
In summary, the 2026 market for drop foot braces shoes will be shaped by technological innovation, demographic shifts, patient-centric design, and evolving healthcare delivery models—positioning the sector for robust, human-centered growth.
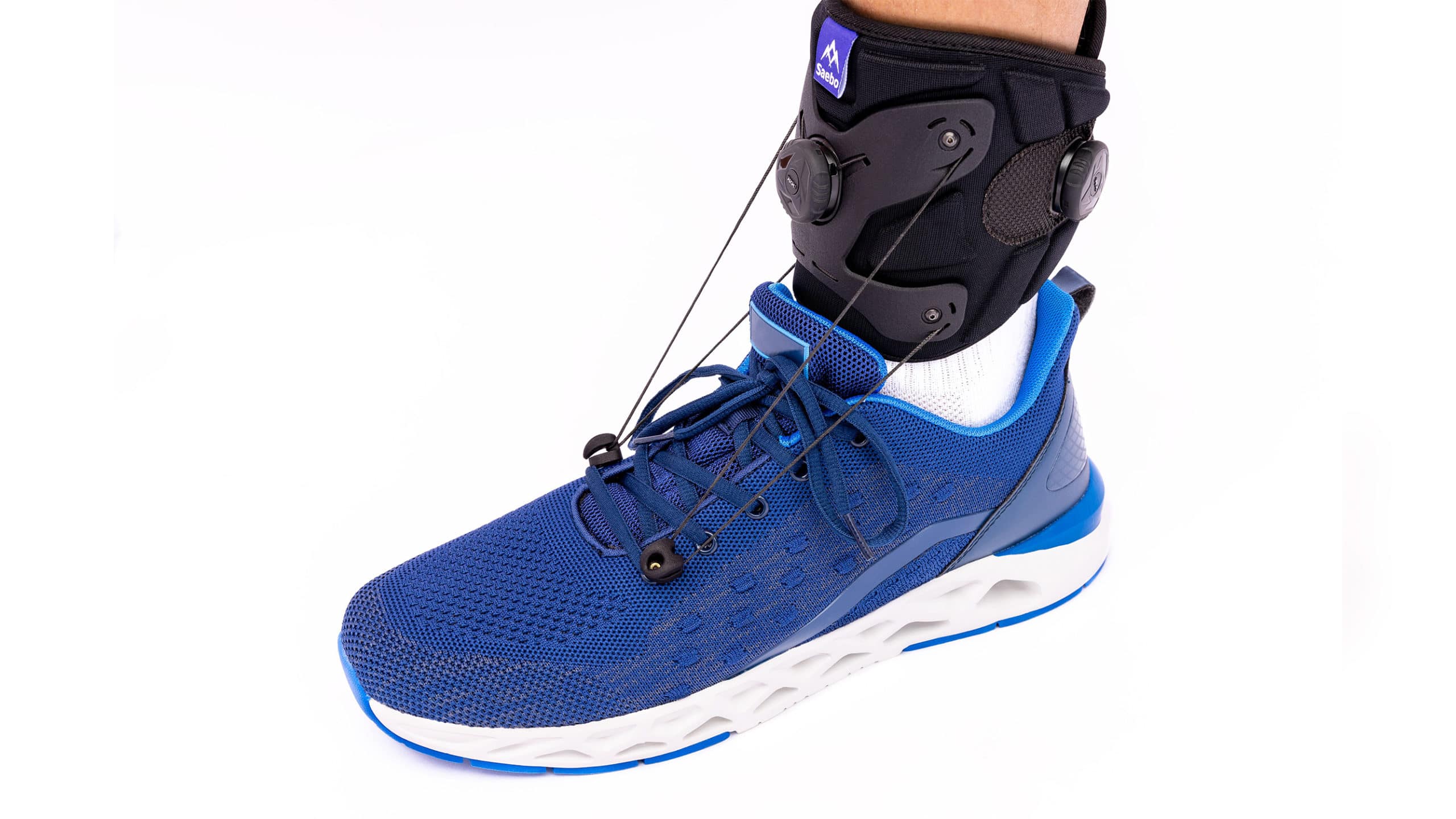
Common Pitfalls When Sourcing Drop Foot Braces Shoes (Quality, Intellectual Property)
Sourcing drop foot brace shoes—also known as AFO (Ankle-Foot Orthosis) shoes—requires careful attention to both product quality and intellectual property considerations. Failure to address these areas can lead to safety risks, regulatory non-compliance, legal liabilities, and reputational damage. Below are key pitfalls to avoid.
Poor Material and Construction Quality
One of the most significant risks in sourcing drop foot brace shoes is receiving products made with substandard materials or poor craftsmanship. Low-quality plastics, weak hinges, or inadequate support can compromise patient safety and therapeutic effectiveness. Sourcing from manufacturers with lax quality control processes may result in inconsistent sizing, breakage under stress, or discomfort during prolonged use.
To mitigate this, ensure suppliers adhere to medical device standards such as ISO 13485 and conduct third-party testing for durability, load-bearing capacity, and biocompatibility. Request product samples and conduct wear-testing before mass production.
Lack of Customization and Proper Fit
Drop foot patients often require highly personalized solutions depending on the severity of their condition, gait pattern, and foot anatomy. A common pitfall is sourcing shoes with rigid, one-size-fits-all designs that fail to accommodate individual needs. Poor fit can lead to skin irritation, pressure sores, or reduced mobility.
Ensure suppliers offer adjustable features—such as modular AFOs, cushioned insoles, and adaptable fastening systems. Work with manufacturers who support customization or partner with clinics to validate fit and function.
Inadequate Compliance with Medical Device Regulations
Drop foot brace shoes may be classified as medical devices depending on jurisdiction (e.g., Class I or II under FDA or EU MDR). A major sourcing risk is partnering with suppliers who do not comply with regional regulatory requirements. Non-compliant products can be blocked at customs, recalled, or result in legal penalties.
Verify that suppliers have necessary certifications (e.g., CE marking, FDA registration) and maintain technical documentation, including risk assessments and clinical evaluations, where required.
Intellectual Property Infringement
Many established designs for AFO shoes and integrated orthotic systems are protected by patents, trademarks, or design rights. Sourcing from manufacturers who copy patented mechanisms (e.g., specific hinge systems, shoe-orthosis integration techniques) exposes your business to IP litigation.
Conduct thorough IP due diligence before finalizing a supplier. Search patent databases (e.g., USPTO, WIPO) and consider consulting an IP attorney to assess freedom-to-operate. Avoid suppliers who cannot provide proof of IP ownership or licensing for critical components.
Hidden Costs from Design and Tooling Ownership
Some suppliers may offer low unit prices but retain ownership of molds, tooling, or product designs. This creates dependency and limits your ability to switch manufacturers or iterate on the design. It can also complicate IP protection and scalability.
Always negotiate clear terms regarding design ownership and tooling rights in your contract. Ensure you retain full rights to product designs and manufacturing tools to maintain control and flexibility.
Insufficient Post-Market Surveillance and Support
Medical devices require ongoing monitoring for safety and performance. Sourcing from suppliers without post-market surveillance processes increases the risk of undetected defects or failure to respond to adverse events.
Choose partners who provide traceability, warranty support, and mechanisms for reporting and resolving issues. This is critical for regulatory compliance and patient trust.
By addressing these quality and intellectual property pitfalls proactively, businesses can ensure they source safe, effective, and legally compliant drop foot brace shoes that meet both clinical and commercial objectives.

Logistics & Compliance Guide for Drop Foot Braces Shoes
Drop Foot Braces Shoes are classified as medical devices designed to assist individuals with foot drop, a condition affecting gait. Proper logistics and compliance are essential to ensure patient safety, regulatory adherence, and smooth supply chain operations. This guide outlines key considerations for manufacturers, distributors, and importers.
Regulatory Classification and Registration
Drop Foot Braces Shoes are typically classified as Class I or Class II medical devices, depending on design, functionality, and risk level. In the United States, they must be registered with the U.S. Food and Drug Administration (FDA). Most models fall under 21 CFR 890.3825 (Ankle-Foot Orthoses). Manufacturers must:
- Register their facility and list devices with the FDA.
- Comply with Quality System Regulation (QSR) under 21 CFR Part 820.
- Obtain a Medical Device Listing (MDL) number.
- Ensure devices have proper labeling, including product name, model number, manufacturer details, and intended use.
In the European Union, these devices require CE marking under Regulation (EU) 2017/745 on Medical Devices (MDR). Key steps include:
- Determine the correct classification (usually Class I or IIa).
- Appoint an EU Authorized Representative if the manufacturer is outside the EU.
- Prepare a Technical File and conduct conformity assessment.
- Issue an EU Declaration of Conformity.
- Register devices in the EUDAMED database.
Other regions (e.g., Canada, Australia, UK) have similar requirements through Health Canada (MDL), TGA (ARTG listing), and UK MHRA (UKCA marking post-Brexit).
Labeling and Packaging Requirements
Proper labeling is critical for compliance and patient safety. Labels must include:
- Device name and model number.
- Manufacturer name, address, and contact information.
- Intended use and indications for use.
- Contraindications and warnings.
- Size specifications and fitting instructions.
- Lot number or serial number.
- Expiration date, if applicable.
- Symbols per ISO 15223-1 (e.g., sterile, single-use, do not reuse).
- UDI (Unique Device Identification) in markets requiring it (e.g., U.S., EU).
Packaging should protect the device during transit and storage. Use tamper-evident seals and materials resistant to moisture and physical damage. Include user manuals in applicable languages, especially for international shipments.
Import and Export Compliance
When shipping internationally, comply with customs and import regulations:
- Obtain an HS (Harmonized System) code. Common codes include:
- 9021.39 (Other orthopedic appliances) for most braces.
- 6402.99 (Other footwear with outer soles of rubber, plastics, leather) if integrated with shoe components.
- Provide accurate commercial invoices, packing lists, and certificates of origin.
- Include regulatory documentation (e.g., CE Certificate, FDA registration letter, Certificate of Free Sale).
- Verify import permits or notifications required by destination countries.
- Comply with U.S. Customs and Border Protection (CBP) and FDA import alerts.
For exports from the U.S., file Electronic Export Information (EEI) via the Automated Export System (AES) if the shipment value exceeds $2,500 or requires a license.
Storage and Distribution Logistics
- Store devices in a clean, dry environment with controlled temperature (typically 15–30°C).
- Avoid direct sunlight and high humidity to prevent material degradation.
- Follow FIFO (First In, First Out) inventory practices.
- Monitor shelf life for any components with expiration dates.
- Use validated shipping methods that maintain product integrity.
- Partner with logistics providers experienced in medical device handling.
- Implement temperature monitoring for sensitive materials during transit, if needed.
Post-Market Surveillance and Reporting
Manufacturers must establish a post-market surveillance (PMS) system to monitor device performance and safety:
- Collect and analyze complaints and adverse events.
- Report serious incidents to regulatory authorities within required timelines (e.g., 30 days for FDA, 15 days for EU under MDR).
- Conduct periodic safety update reports (PSURs) for higher-risk devices.
- Maintain records for at least 10 years post-device discontinuation.
Unique Device Identification (UDI) Compliance
In the U.S. and EU, Drop Foot Braces Shoes must carry a UDI:
- Assign a Device Identifier (DI) and Production Identifier (PI).
- Label devices with UDI in both human-readable and machine-readable (e.g., barcode, Data Matrix) formats.
- Submit UDI data to the FDA’s GUDID and EUDAMED.
- Ensure UDI remains legible throughout the device’s lifecycle.
Conclusion
Successfully managing logistics and compliance for Drop Foot Braces Shoes requires adherence to global regulatory standards, accurate documentation, proper labeling, and robust supply chain practices. Staying current with evolving regulations (e.g., EU MDR, FDA updates) and conducting regular internal audits will help ensure market access and patient safety. Engaging regulatory consultants or third-party compliance experts is recommended for complex international rollouts.
Conclusion on Sourcing Drop Foot Braces and Shoes
Sourcing drop foot braces and specialized footwear requires a careful balance between medical effectiveness, comfort, durability, and cost. After evaluating various suppliers, product types (such as AFOs – Ankle-Foot Orthoses), and integrated brace-shoe systems, it is clear that the best outcomes arise from a personalized, patient-centered approach. Custom orthotics from reputable medical device manufacturers offer superior fit and support, particularly for long-term use, while off-the-shelf options provide a cost-effective and timely solution for temporary or mild cases.
Key factors in successful sourcing include compliance with medical standards (e.g., FDA, CE marking), access to certified orthotists for fitting and adjustments, and the availability of breathable, lightweight materials that enhance user comfort and mobility. Additionally, integrating patient feedback and ensuring compatibility with daily lifestyle needs—such as ease of wear, shoe style, and adaptability to different terrains—significantly impacts adherence and rehabilitation success.
In conclusion, a strategic sourcing plan should involve collaboration between healthcare providers, orthotic specialists, and patients to select the most appropriate drop foot brace or brace-integrated shoe. Prioritizing quality, customization, and long-term usability not only improves mobility and safety for patients but also reduces the risk of secondary complications, ultimately leading to better clinical and quality-of-life outcomes.