The global dialysis machines market is experiencing robust growth, driven by the rising prevalence of chronic kidney disease (CKD), increasing geriatric population, and advancements in dialysis technologies. According to Mordor Intelligence, the dialysis equipment market was valued at approximately USD 13.5 billion in 2023 and is projected to grow at a compound annual growth rate (CAGR) of over 6.8% from 2024 to 2029. This expansion is further supported by growing healthcare expenditure, improved access to renal care in emerging economies, and a shift toward home-based dialysis solutions. As demand surges, a handful of key manufacturers are leading innovation and market share, shaping the future of renal replacement therapy. The following list highlights the top eight dialysis machine manufacturers that are at the forefront of this dynamic sector.
Top 8 Dialysis Machine Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Worldwide
Domain Est. 2019
Website: polletmedicalgroup.com
Key Highlights: Farmasol is a dialysis XPRT and manufacturer of Hemodialysis products. As one of the few companies, Farmasol produces almost all hemodialysis consumables ……
#2 Dialysis
Website: asahi-kasei.co.jp
Key Highlights: Dialysis products – We offer high-performance dialyzers that improve therapeutic effectiveness and contribute to patients’ quality of life….
#3 Hollow
Domain Est. 1995
Website: toray.com
Key Highlights: This is the products search page of Toray’s official website. You can search for products by application or business field….
#4 Products
Domain Est. 2000
Website: freseniusmedicalcare.com
Key Highlights: Fresenius Medical Care provides dialysis care products including dialysis machines and supplies, dialyzers and more….
#5 NIPRO Medical Corporation
Domain Est. 2008
Website: nipro-group.com
Key Highlights: Renal Care. We offer innovative, high-quality hemodialysis and dialysis solutions, ensuring patient safety and efficiency….
#6 A New Way of Experiencing Dialysis
Domain Est. 2014
Website: outsetmedical.com
Key Highlights: Outset Medical is reimagining dialysis for patients and providers, from the ICU to the home….
#7 SWS dialysis,medical,sws hemodialysis machine,CRRT,dialyzer
Domain Est. 2024
Website: swsdialysis.com
Key Highlights: Its branded products include:SWS dialysis,SWS medical,SWS dialysis machine,SWS hemodialysis … The official website of SWS company is:www.swsdialysis.com….
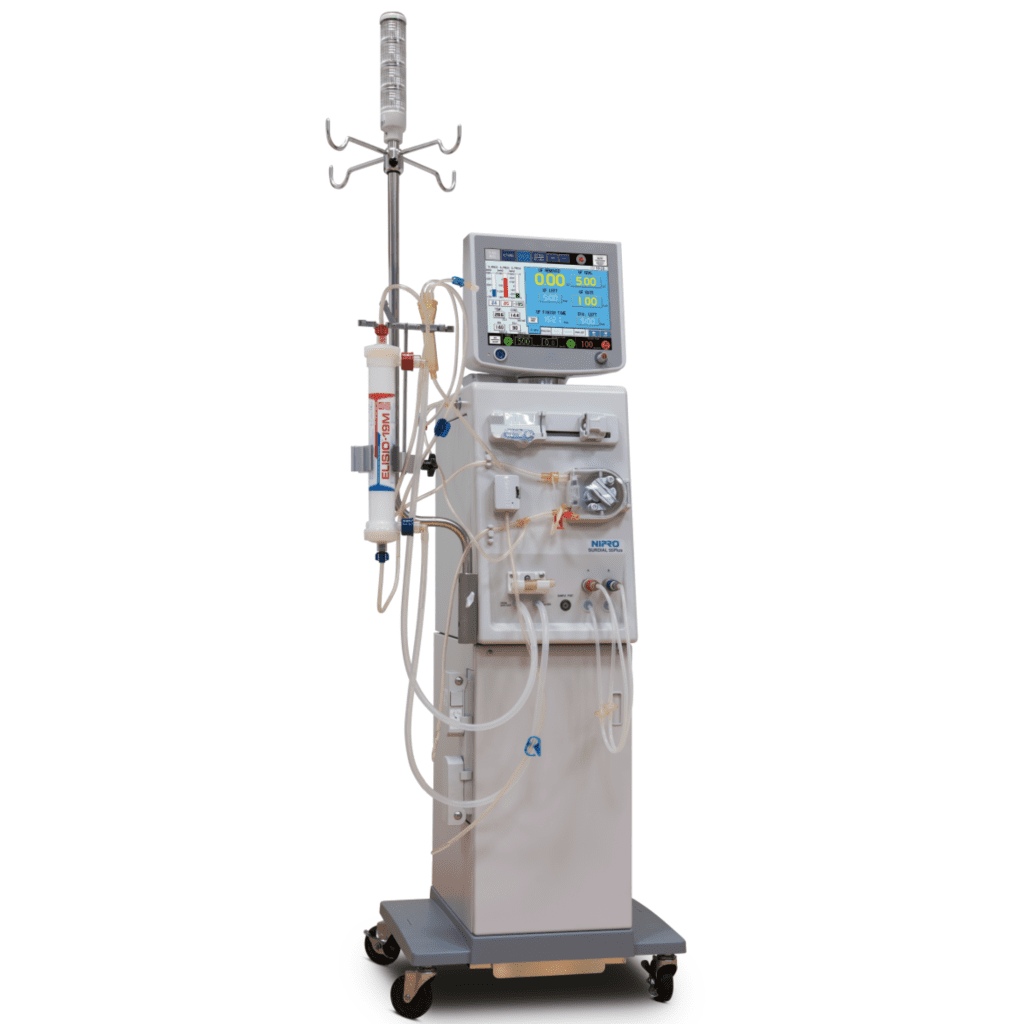
#8 Dialysis Machine
Website: nipro.co.jp
Key Highlights: Renal Products. Dialysis Machine. Surdial™ X. Equipped with various monitoring and profile functions to pursue optimized treatments. MORE(PDF:502KB) ……
Expert Sourcing Insights for Dialysis Machine
H2: 2026 Market Trends for Dialysis Machines
The global dialysis machine market is poised for significant transformation by 2026, driven by demographic shifts, technological advancements, healthcare policy changes, and increasing prevalence of chronic kidney disease (CKD). Below is an analysis of the key trends shaping the dialysis machine market in 2026:
-
Rising Prevalence of Chronic Kidney Disease
The growing incidence of diabetes, hypertension, and aging populations worldwide is fueling demand for dialysis treatments. According to the International Society of Nephrology, over 850 million people globally suffer from kidney diseases, with CKD ranking among the top 20 causes of death. By 2026, this expanding patient pool—especially in emerging economies such as India, China, and parts of Africa—is expected to drive sustained demand for both hemodialysis and peritoneal dialysis machines. -
Technological Innovation and Home-Based Dialysis
A major trend in 2026 is the shift toward portable and user-friendly dialysis machines that support home-based treatments. Innovations such as wearable artificial kidneys, compact hemodialysis units, and smart dialysis systems integrated with remote monitoring are gaining momentum. Companies like Baxter International and Fresenius Medical Care are investing heavily in home dialysis technologies, aiming to improve patient quality of life and reduce hospital congestion. The convenience and cost-effectiveness of home dialysis are expected to boost its adoption, particularly in North America and Western Europe. -
Digital Health Integration
By 2026, digital health technologies are becoming integral to dialysis care. Dialysis machines are increasingly equipped with IoT connectivity, AI-powered analytics, and cloud-based data platforms. These systems enable real-time monitoring of patient vitals, treatment efficacy, and machine performance. Predictive maintenance and personalized therapy adjustments enhance clinical outcomes and operational efficiency. Tele-nephrology and remote patient management are also expected to expand access to care in rural and underserved areas. -
Growth in Emerging Markets
While North America and Europe remain dominant in terms of market value, the fastest growth is anticipated in Asia-Pacific, Latin America, and the Middle East. Rising healthcare expenditure, improving medical infrastructure, and government initiatives to expand dialysis access are key drivers. For instance, India and China are launching national kidney disease programs and subsidizing dialysis treatments, creating substantial opportunities for medical device manufacturers. -
Focus on Cost Efficiency and Reimbursement Reforms
Healthcare systems worldwide are under pressure to manage costs, prompting a focus on value-based care models. In 2026, payers and providers are increasingly favoring reusable components, automated fluid management systems, and machines with lower maintenance requirements. In the U.S., the Centers for Medicare & Medicaid Services (CMS) continue to refine reimbursement models under the End-Stage Renal Disease (ESRD) Prospective Payment System, encouraging providers to adopt efficient and innovative technologies. -
Competitive Landscape and Strategic Collaborations
The dialysis machine market is highly consolidated, with key players including Fresenius Medical Care, Baxter International, B. Braun, Asahi Kasei, and Nikkiso. By 2026, strategic partnerships, mergers, and R&D collaborations are expected to intensify, particularly in emerging technologies such as bioartificial kidneys and regenerative medicine. Startups focusing on next-generation dialysis solutions are attracting venture capital, signaling a dynamic and evolving competitive environment. -
Sustainability and Environmental Concerns
Environmental sustainability is emerging as a concern in dialysis care due to the large volume of disposable supplies and water usage. By 2026, manufacturers are responding with eco-friendly designs, water recycling systems, and biodegradable components. Regulatory bodies and healthcare providers are also encouraging greener practices, influencing procurement decisions.
Conclusion
By 2026, the dialysis machine market will be characterized by innovation, decentralization of care, digital integration, and geographic expansion. As the global burden of kidney disease grows, the market will continue to evolve toward more patient-centric, efficient, and sustainable solutions. Stakeholders who embrace these trends—manufacturers, healthcare providers, and policymakers—will be best positioned to meet the escalating demand and improve outcomes for dialysis patients worldwide.
Common Pitfalls in Sourcing Dialysis Machines: Quality and Intellectual Property (IP) Concerns
Sourcing dialysis machines involves significant risks, particularly concerning product quality and intellectual property (IP) integrity. Overlooking these areas can lead to regulatory non-compliance, patient safety issues, legal disputes, and reputational damage. Below are key pitfalls to avoid:
Quality-Related Pitfalls
Inadequate Regulatory Compliance Verification
Failing to confirm that the dialysis machine meets essential regulatory standards—such as FDA 510(k) clearance, CE marking under the EU Medical Device Regulation (MDR), or local health authority approvals—can result in shipment rejections or legal penalties. Always verify up-to-date certifications and conduct audits of the manufacturer’s quality management system (e.g., ISO 13485).
Poor Manufacturing Standards and Process Controls
Sourcing from manufacturers with weak or uncertified production processes increases the risk of defective units. Substandard materials, inconsistent assembly, or lack of environmental controls can compromise device reliability. Conduct on-site manufacturing audits to assess adherence to Good Manufacturing Practices (GMP).
Insufficient Clinical Validation and Performance Data
Relying on marketing claims without reviewing clinical trial data, biocompatibility reports, or hemodialysis efficiency metrics (e.g., urea clearance, ultrafiltration accuracy) can lead to underperforming devices. Request and analyze third-party test reports and peer-reviewed studies.
Inadequate Post-Market Surveillance and Support
A lack of a robust post-market surveillance system means delayed responses to device malfunctions or adverse events. Ensure suppliers have active monitoring, timely software/firmware updates, and accessible technical support, especially for critical components like dialysate monitoring systems.
Counterfeit or Refurbished Units Misrepresented as New
Some suppliers may pass off refurbished or counterfeit dialysis machines as new. These units may lack proper safety testing or use substandard replacement parts. Verify serial numbers, conduct physical inspections, and source only from authorized distributors.
Intellectual Property-Related Pitfalls
Sourcing from IP-Infringing Manufacturers
Procuring machines from suppliers that replicate patented technology (e.g., proprietary blood pump mechanisms, control algorithms, or dialyzer designs) exposes buyers to legal liability. Conduct IP due diligence, including patent landscape analysis and supplier declaration of freedom-to-operate.
Lack of IP Ownership Transparency
Ambiguity around who owns the IP—especially for software, user interfaces, or connected health features—can lead to disputes. Clarify IP rights in contracts, including rights to use, modify, and service the device, particularly if customization is involved.
Unauthorized Modifications or Cloning
Some suppliers modify or clone original equipment to reduce costs, potentially violating patents and compromising safety. Ensure strict contractual clauses prohibiting unauthorized alterations and require compliance with original design specifications.
Inadequate Protection of Buyer’s Customizations
If a buyer commissions custom features (e.g., integration with hospital IT systems), failure to secure IP rights for these modifications can result in loss of control. Include clear IP assignment clauses in procurement agreements.
Risk of Trade Secret Leakage
Sharing technical requirements or integration plans with unvetted suppliers may expose sensitive information. Use non-disclosure agreements (NDAs) and limit data sharing to what is strictly necessary.
By proactively addressing these quality and IP pitfalls through rigorous supplier vetting, contractual safeguards, and regulatory validation, organizations can mitigate risks and ensure the safe, compliant, and legally sound procurement of dialysis machines.
Logistics & Compliance Guide for Dialysis Machines
Regulatory Compliance
Dialysis machines are classified as medical devices and are subject to strict regulatory oversight to ensure patient safety and device efficacy. Compliance with international and local regulations is mandatory throughout the product lifecycle.
FDA Requirements (U.S.)
In the United States, dialysis machines are regulated by the U.S. Food and Drug Administration (FDA) under Class II medical devices. Manufacturers must:
– Obtain 510(k) premarket notification clearance or Premarket Approval (PMA) if applicable.
– Register the device and list it in the FDA’s Device Registration and Listing database.
– Comply with Quality System Regulation (QSR) 21 CFR Part 820 for design, manufacturing, packaging, labeling, storage, and distribution.
– Implement post-market surveillance, including Medical Device Reporting (MDR) for adverse events.
CE Marking (European Union)
In the European Economic Area (EEA), dialysis machines must comply with the EU Medical Device Regulation (MDR) 2017/745. Key requirements include:
– Classification under Rule 10 (active therapeutic devices) typically as Class IIa or IIb.
– Conformity assessment involving a Notified Body.
– Technical documentation and risk management per ISO 14971.
– Affixing CE marking after successful assessment and issuing an EU Declaration of Conformity.
Other International Regulations
- Health Canada: Requires a Medical Device License (MDL) under the Medical Devices Regulations (SOR/98-282).
- Australia (TGA): Must be included in the Australian Register of Therapeutic Goods (ARTG).
- Japan (PMDA): Requires approval under the Pharmaceutical and Medical Device Act (PMDA).
- China (NMPA): Registration through the National Medical Products Administration with mandatory local testing and clinical evaluation.
Transportation & Handling
Due to their sensitive electronic components and mechanical parts, dialysis machines require careful handling and compliant logistics planning.
Packaging Requirements
- Use manufacturer-approved, shock-resistant packaging with internal immobilization.
- Include moisture barriers and desiccants to prevent condensation.
- Clearly label packages with “Fragile,” “This Side Up,” and “Medical Device” indicators.
- Provide tamper-evident seals to ensure integrity.
Shipping Conditions
- Maintain ambient temperatures between 10°C and 30°C during transit unless otherwise specified.
- Avoid exposure to extreme humidity, direct sunlight, or water.
- Utilize temperature and shock monitoring devices for high-value shipments.
- Ensure compliance with IATA and IMDG regulations if shipping internationally, especially with included batteries.
Transport Modes
- Air freight: Preferred for urgent deliveries; comply with IATA Dangerous Goods Regulations if lithium batteries are installed.
- Ground transport: Suitable for domestic or regional deliveries; vehicles must be climate-controlled and vibration-minimized.
- Sea freight: Cost-effective for large shipments; requires extended protective packaging and moisture control.
Import/Export Documentation
International movement of dialysis machines requires precise documentation to avoid customs delays.
Required Documents
- Commercial invoice (with HS code: typically 9018.90 for medical instruments).
- Packing list.
- Certificate of Conformity (CE, FDA, or other applicable).
- Export license (if required by the exporting country).
- Import permit (often required by health authorities in the destination country).
- Bill of Lading or Air Waybill.
- Manufacturer’s Declaration of Medical Device Use.
HS Code & Tariff Classification
- Harmonized System (HS) Code: 9018.90.6000 (for dialysis machines in many jurisdictions).
- Verify local tariff schedules as import duties and taxes vary by country.
Installation & Commissioning Compliance
Upon delivery, proper installation and validation are critical for regulatory and operational compliance.
Site Preparation
- Ensure facility meets electrical specifications (voltage, grounding, circuit capacity).
- Confirm availability of purified water and drainage for hemodialysis machines.
- Validate environmental conditions (temperature, humidity, space).
Installation Protocol
- Installation must be performed by trained biomedical technicians or authorized service personnel.
- Follow manufacturer’s installation manual and checklist.
- Perform initial safety and performance tests (e.g., leak tests, calibration).
Documentation & Training
- Complete and retain an Installation Qualification (IQ) report.
- Provide operator training and obtain sign-off.
- Register the device with local health authorities if required.
Post-Market Surveillance & Maintenance
Ongoing compliance requires monitoring and servicing of dialysis machines in clinical use.
Preventive Maintenance
- Adhere to manufacturer-recommended maintenance schedules.
- Keep detailed service logs and calibration records.
- Use only OEM or authorized replacement parts.
Incident Reporting
- Report malfunctions, adverse events, or patient injuries to relevant regulatory bodies (e.g., FDA MedWatch, EUDAMED).
- Conduct root cause analysis and implement corrective actions.
Software & Firmware Updates
- Apply updates as released by the manufacturer.
- Validate updates do not compromise device safety or performance.
- Document all updates in device history records.
Summary
Successful logistics and compliance for dialysis machines require adherence to global regulatory standards, careful transportation planning, accurate documentation, and rigorous post-market oversight. Collaboration between manufacturers, distributors, healthcare providers, and regulatory bodies ensures patient safety and uninterrupted clinical operations.
Conclusion:
After a thorough evaluation of potential suppliers for dialysis machines, it is evident that selecting the right partner is critical to ensuring high-quality patient care, operational efficiency, and regulatory compliance. Key factors such as product reliability, technical support, warranty terms, cost-effectiveness, delivery timelines, and after-sales service have been carefully assessed.
Based on the comparative analysis, Supplier [Insert Supplier Name] emerges as the most suitable choice due to their proven track record in delivering FDA/CE-certified dialysis machines, comprehensive training programs, responsive customer support, and competitive pricing. Additionally, their compliance with international quality standards and experience in serving healthcare institutions strengthen their credibility.
Proceeding with this supplier will not only enhance the dialysis unit’s service delivery but also support long-term sustainability and patient safety. It is recommended to formalize the procurement process through a detailed contract outlining service level agreements, maintenance schedules, and training sessions to ensure seamless integration into clinical operations.