The global creatine market has experienced steady growth over the past decade, driven by rising consumer awareness of sports nutrition, expanding fitness industries, and increased demand for performance-enhancing supplements. According to Mordor Intelligence, the global creatine market was valued at approximately USD 266 million in 2023 and is projected to grow at a compound annual growth rate (CAGR) of over 6.8% from 2024 to 2029. This growth is further supported by rising participation in athletic and recreational fitness activities, particularly across North America and Asia-Pacific regions. As demand surges, innovation in formulation—such as creatine monohydrate, hydrochloride, and buffered variants—has intensified competition among manufacturers. Supply chain consolidation, quality certifications, and research-backed production standards now play a pivotal role in market differentiation. In this evolving landscape, the top eight creatine manufacturers have emerged as leaders through scale, scientific rigor, and global distribution networks—setting the benchmark for purity, efficacy, and compliance in the sports nutrition sector.
Top 8 Creatine Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 NSF Product and Service Listings
Domain Est. 1996
Website: info.nsf.org
Key Highlights: Trade Designation, Product ID, Product Form, Manufacturer’s Recommended Daily Serving Size ; Creatine ; Creatine HMB Black Cherry, All, Powder, 1 scoop (9.5 g)….
#2 CreaBev®
Domain Est. 2002
Website: glanbianutritionals.com
Key Highlights: Achieve advanced creatine performance with less degradation. From ready-to-mix powders, acidic through neutral pH ready-to-drink beverages, gummies and gels….
#3 Creatine Manufacturer
Domain Est. 2021
Website: nutracoast.com
Key Highlights: Partner with Nutra Coast — a trusted USA-based creatine manufacturer offering custom, high-quality supplements with cGMP & FDA-compliant production….
#4 Creapure
Domain Est. 1998
Website: creapure.com
Key Highlights: Creapure® – Pure Creatine. Made in Germany. More energy. Shorter recovery. Faster muscle growth. Better physical well-being….
#5 Creatine MagnaPower®
Domain Est. 1999
Website: balchem.com
Key Highlights: Creatine MagnaPower® is a unique blend of magnesium and creatine. It helps deliver ATP to muscle cells, increasing anaerobic performance….
#6 Creatine [+]
Domain Est. 2002
Website: optimumnutrition.com
Key Highlights: Optimum Nutrition Creatine Plus, 0.79lb container, Orange Passionfruit flavor, enhanced 3-in-1 formula for strength and recovery Optimum Nutrition Creatine ……
#7 Creapure®
Domain Est. 2006
Website: alzchem.com
Key Highlights: Creapure® is the premium brand for creatine worldwide – Made in Germany. You can find more information here….
#8 Creatine monohydrate (Creapure®) powder
Domain Est. 2020
Website: protealpes.com
Key Highlights: In stock Rating 5.0 (54) Creapure®-certified creatine made in Germany, packaged by Protéalpes in Albertville. Normally free from doping substances, it improves physical performan…
Expert Sourcing Insights for Creatine

2026 Market Trends for Creatine: Key Developments and Outlook
As we approach 2026, the global creatine market is poised for continued growth and transformation, driven by evolving consumer preferences, scientific advancements, and expanding applications beyond traditional sports nutrition. Here are the major trends shaping the creatine landscape:
Rising Demand Across Diverse Consumer Segments
While athletes and bodybuilders remain core users, creatine is experiencing significant adoption among broader demographics. Older adults are increasingly incorporating creatine to support muscle preservation, cognitive function, and healthy aging. Additionally, interest is growing among women seeking performance enhancement and general wellness, as well as recreational gym-goers focused on overall fitness. This demographic expansion is broadening the market base and driving product innovation tailored to different lifestyles and health goals.
Product Innovation and Formulation Advancements
The market is moving beyond basic creatine monohydrate powders. By 2026, expect to see a surge in advanced delivery formats such as chewables, gummies, effervescent tablets, and ready-to-drink (RTD) beverages to improve convenience and palatability. Enhanced formulations combining creatine with synergistic ingredients—like beta-alanine, electrolytes, plant-based proteins, and nootropics—are becoming popular for targeted benefits in energy, endurance, and mental focus. Flavored and unflavored options catering to clean-label and natural product preferences are also on the rise.
Expansion into Cognitive and Longevity Health
Scientific research continues to validate creatine’s role beyond physical performance, particularly in brain health. Studies highlighting its potential to support memory, reduce mental fatigue, and mitigate age-related cognitive decline are fueling its positioning in the nootropic and longevity markets. By 2026, creatine is likely to be more commonly marketed as a dual-purpose supplement for both physical and mental performance, attracting consumers interested in holistic wellness and preventive health.
Sustainability and Ethical Sourcing Gaining Importance
As with other supplement categories, environmentally conscious consumers are demanding transparency in sourcing and manufacturing. Brands are responding by emphasizing sustainable production practices, vegan/vegetarian certification (especially for synthetic creatine), recyclable packaging, and third-party testing for purity and contaminants. Companies that prioritize sustainability and ethical claims are expected to gain competitive advantage and consumer trust.
Regulatory Scrutiny and Quality Assurance
With market growth comes increased regulatory attention. In regions like the EU and North America, authorities are tightening controls on supplement labeling, health claims, and ingredient safety. By 2026, compliance with evolving regulations—such as the EU’s Novel Food regulations for new creatine derivatives—will be critical. Consumers are also becoming more educated, favoring brands that provide third-party certifications (e.g., NSF, Informed-Sport) to ensure product quality and safety, especially in professional and collegiate sports.
Digital Influence and Direct-to-Consumer Growth
E-commerce and digital marketing remain powerful drivers of creatine sales. Social media platforms, influencer endorsements, and personalized nutrition apps are shaping consumer choices and driving brand discovery. Direct-to-consumer (DTC) brands are leveraging data analytics to offer tailored creatine regimens and subscription models, enhancing customer retention and brand loyalty in a competitive marketplace.
Regional Market Dynamics
North America continues to dominate the creatine market, but Asia-Pacific is expected to register the highest growth rate by 2026, fueled by rising fitness awareness, urbanization, and disposable income in countries like China, India, and South Korea. Emerging markets in Latin America and the Middle East are also showing increasing demand, supported by expanding health club infrastructure and digital access to fitness content.
In summary, the 2026 creatine market reflects a maturing industry characterized by innovation, diversification, and a shift toward science-backed, consumer-centric products. As understanding of creatine’s benefits deepens and delivery methods evolve, its role is expanding from a niche sports supplement to a mainstream wellness staple.

Common Pitfalls When Sourcing Creatine: Quality and Intellectual Property (IP) Concerns
Sourcing creatine, especially for supplement manufacturers, requires careful attention to both product quality and intellectual property rights. Overlooking these aspects can lead to regulatory issues, reputational damage, and legal liabilities. Below are the key pitfalls to avoid.
Quality-Related Pitfalls
-
Inadequate Purity and Contaminant Testing
Low-quality creatine may contain impurities such as dicyandiamide, cyanamide, or heavy metals due to poor manufacturing processes. Always require third-party Certificates of Analysis (CoA) verifying purity (ideally ≥99.9%) and absence of contaminants. -
Misrepresentation of Form (e.g., Creatine Monohydrate vs. Alternatives)
Some suppliers falsely market inferior or synthetic analogs as premium forms like micronized creatine monohydrate. Confirm the exact form and verify it through independent lab testing. -
Inconsistent Particle Size and Solubility
Poorly processed creatine may have inconsistent particle size, affecting solubility and consumer experience. This is especially critical for powdered formulations. Request specifications on mesh size and dissolution profile. -
Lack of GMP or ISO Certification
Sourcing from facilities without Good Manufacturing Practice (GMP) or ISO 22716 certification increases the risk of inconsistent quality and non-compliance with regulatory standards (e.g., FDA, EFSA). -
Unreliable Supply Chain Transparency
Hidden subcontractors or opaque sourcing can compromise traceability. Ensure full visibility into the manufacturing and supply chain, including raw material origins.
Intellectual Property (IP)-Related Pitfalls
-
Infringement of Patented Creatine Forms or Processes
While creatine monohydrate itself is off-patent, certain delivery systems (e.g., buffered creatine, creatine salts like Creapure®), stabilization methods, or production processes may be protected. Using patented technologies without licensing can lead to legal action. -
Trademark Violations (e.g., Creapure®)
“Creapure®” is a registered trademark of AlzChem for high-purity creatine monohydrate. Using this name—or packaging that mimics it—without authorization constitutes trademark infringement, even if the product is chemically identical. -
False “Patent-Pending” or “Proprietary” Claims
Misrepresenting a formulation as patented or proprietary when it is not can mislead consumers and violate advertising standards (e.g., FTC guidelines), leading to enforcement actions. -
Failure to Conduct IP Due Diligence
Not performing a freedom-to-operate (FTO) analysis before launching a product can result in costly litigation. Work with legal counsel to assess existing patents related to creatine formulations and delivery methods. -
Sourcing from Unlicensed or Grey-Market Suppliers
Some suppliers may offer “Creapure®-equivalent” creatine sourced from unauthorized channels. These materials may not meet quality standards and could expose your brand to IP risks if the trademark is implied or misused.
Best Practices to Mitigate Risks
- Verify Supplier Credentials: Audit suppliers for GMP, ISO, and relevant certifications.
- Demand Comprehensive Testing: Require CoAs for every batch, including purity, heavy metals, and microbial content.
- Protect Your IP: Consider trademarking your brand name and securing patents for novel formulations.
- Consult Legal Experts: Engage IP attorneys to review formulations, packaging, and marketing claims.
- Use Authorized Distributors: For branded ingredients like Creapure®, source only through licensed channels to ensure authenticity and compliance.
By addressing both quality and IP concerns proactively, businesses can safeguard product integrity, comply with regulations, and protect their brand reputation in the competitive supplement market.

Logistics & Compliance Guide for Creatine
Product Classification & Regulatory Status
Creatine is classified as a dietary supplement ingredient in most major markets, including the United States, European Union, Canada, and Australia. As such, it is subject to dietary supplement regulations rather than pharmaceutical requirements. In the U.S., the FDA regulates creatine under the Dietary Supplement Health and Education Act (DSHEA), requiring adherence to Good Manufacturing Practices (GMPs) outlined in 21 CFR Part 111. In the EU, creatine falls under the EU Food Supplements Directive (2002/46/EC), with member states implementing national rules based on harmonized standards.
Manufacturing & Quality Control
Manufacturers must comply with cGMP (current Good Manufacturing Practices) to ensure product identity, purity, strength, and composition. Key requirements include:
– Raw material testing for authenticity and contaminants (e.g., heavy metals, microbial load)
– In-process and finished product testing
– Stability and shelf-life studies
– Batch record documentation
Creatine monohydrate should meet pharmacopeial standards such as USP-NF or Ph. Eur., specifying criteria for assay, impurity profile, and particle size.
Labeling Requirements
Labels must comply with regional mandates:
– U.S. (FDA): Supplement Facts panel, ingredient list, net quantity, manufacturer/distributor information, and a disclaimer: “This statement has not been evaluated by the FDA. This product is not intended to diagnose, treat, cure, or prevent any disease.”
– EU: Nutrition declaration, list of ingredients in descending order, recommended daily dose, warning if applicable, and statement that supplements should not replace a balanced diet.
– Other Regions: Canada (Natural Product Number required), Australia (AUST L listing), each with specific labeling templates and health claim restrictions.
Health claims must be substantiated and compliant—e.g., in the EU, only the claim “creatine increases physical performance in successive bursts of short-term, high-intensity exercise” is authorized under EFSA Regulation (EC) No 432/2011.
Import/Export Documentation
International trade requires:
– Certificate of Analysis (CoA) for each batch
– Certificate of Free Sale (CFS) issued by the exporting country
– Commercial invoice, packing list, and bill of lading/airway bill
– Import permits, if required (e.g., some countries regulate creatine as a novel food or controlled substance)
HS Code: Typically 2924.19 (other cyclic amides) or 3824.90 (prepared binders for foundry molds), depending on form and purity. Verify with local customs authorities.
Storage & Transportation
- Storage: Keep in a cool, dry, well-ventilated area away from moisture and direct sunlight. Ideal conditions: 15–25°C with <60% relative humidity.
- Packaging: Use food-grade, moisture-resistant containers (e.g., multi-wall poly-lined bags or sealed drums). Ensure tamper-evident packaging for retail units.
- Transportation: Comply with IATA (air) or IMDG (sea) guidelines if shipping in bulk. While creatine is non-hazardous, proper handling prevents contamination and degradation. Monitor temperature and humidity during transit.
Regulatory Notifications & Approvals
- U.S.: Notify FDA within 75 days of marketing a new dietary ingredient (NDI), if applicable. Most creatine forms are grandfathered.
- EU: Notify national authorities before placing product on the market; some countries require pre-market approval.
- China & Brazil: Creatine may require pre-market registration as a health food or supplement. Consult local regulatory agencies (e.g., ANVISA, NMPA).
Adverse Event Reporting
Establish a system to collect, evaluate, and report adverse events:
– In the U.S., submit Serious Adverse Event (SAE) reports to FDA within 15 days via the Safety Reporting Portal.
– In the EU, report through national pharmacovigilance systems or the EudraVigilance database when required.
Maintain records for a minimum of 6 years.
Conclusion
Successfully navigating the logistics and compliance landscape for creatine requires strict adherence to regional regulatory frameworks, robust quality systems, and accurate documentation. Proactive monitoring of evolving regulations—especially in emerging markets—is essential for uninterrupted global distribution.
Conclusion: Sourcing a Creatine Manufacturer
In conclusion, selecting the right creatine manufacturer is a critical decision that directly impacts product quality, regulatory compliance, cost-efficiency, and brand reputation. After evaluating key factors such as manufacturing standards (e.g., GMP, ISO certification), raw material sourcing, product purity (verified through third-party testing), scalability, and regulatory compliance (including FDA, EU, or other regional requirements), it becomes evident that partnering with a reliable, transparent, and experienced manufacturer is essential.
A reputable creatine supplier should offer consistent batch-to-batch quality, comprehensive certification documentation (e.g., COA, MSDS), and the capability to meet specific formulation needs—whether creatine monohydrate, micronized, or alternative forms. Additionally, strong communication, ethical sourcing practices, and sustainable production methods further enhance long-term partnership viability.
Ultimately, thorough due diligence, including on-site audits or virtual assessments and sample testing, will ensure that the chosen manufacturer aligns with your quality standards and business objectives. By prioritizing quality, compliance, and reliability, companies can secure a competitive advantage in the growing dietary supplements market while building consumer trust and brand integrity.


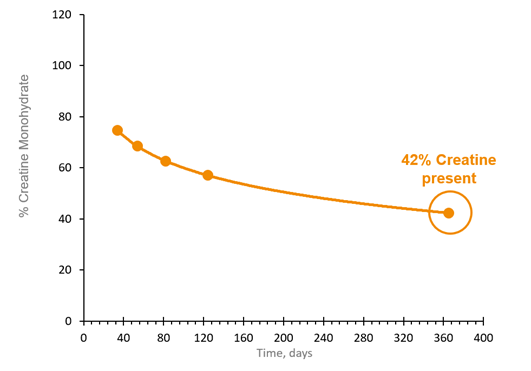


![Creatine [+]](https://www.sohoinchina.com/wp-content/uploads/2026/01/creatine-785.jpg)