The contract manufacturing industry has experienced robust expansion in recent years, driven by increasing demand for cost-efficient production, supply chain optimization, and the need for specialized manufacturing capabilities across sectors such as pharmaceuticals, electronics, and medical devices. According to a 2023 report by Mordor Intelligence, the global contract manufacturing market was valued at USD 789.4 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 9.2% from 2023 to 2028, reaching an estimated USD 1.35 trillion by the end of the forecast period. This growth is further supported by strategic outsourcing trends among OEMs and biopharma companies seeking scalability without heavy capital investment. As competition intensifies and technological advancements accelerate, identifying the top-performing contract manufacturing organizations (CMOs) becomes critical for businesses aiming to ensure quality, compliance, and timely delivery. Based on financial performance, production capacity, geographic reach, and industry specialization, the following 10 manufacturers stand out as leaders in the global contract manufacturing landscape.
Top 10 Contract Manufacturering Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Contract Manufacturing Company
Domain Est. 2005
Website: mplusa.com
Key Highlights: Medical Products Laboratories, Inc. is a full service pharmaceutical contract manufacturer serving the drug, medical device and dietary supplement industries….
#2 SMC
Domain Est. 1996
Website: smcltd.com
Key Highlights: With over 35 years of experience, SMC Ltd. offers industry-leading contract manufacturing for single-use medical devices and full-service pharma solutions….
#3 PLZ Corp
Domain Est. 2021
Website: plzcorp.com
Key Highlights: PLZ Corp is the leading manufacturer of specialty brand and private label aerosol and liquid products in North America. Contact us to get started!…
#4 . We are PCI Pharma Services. Let’s talk future
Domain Est. 1993
Website: pci.com
Key Highlights: PCI is your world leading CDMO, providing integrated end-to-end drug development, manufacturing, and packaging solutions to increase product speed to market and…
#5 Contract manufacturing
Domain Est. 1996
Website: novartis.com
Key Highlights: At Novartis, we partner with highly-regarded pharmaceutical and biotech companies globally as a contract development and manufacturing (CDMO) partner….
#6 Avara
Domain Est. 1996
Website: avara.com
Key Highlights: Avara is a pharmaceutical contract manufacturing organization committed to developing a collaborative relationship with clients to produce quality products….
#7 Contract Manufacturing
Domain Est. 1998
Website: carestream.com
Key Highlights: Our contract manufacturing services allow customers to leverage our lab services, precision coating equipment, manufacturing technologies, fully developed ……
#8 MICRO
Domain Est. 2006
Website: micro-co.com
Key Highlights: MICRO is your partner for End-to-End Medical Device Contract Manufacturing. Our expert engineers provide cutting-edge R&D solutions which support better health ……
#9 AGC Biologics
Domain Est. 2017
Website: agcbio.com
Key Highlights: AGC Biologics is a global CDMO providing development and manufacturing services for protein-based biologics and advanced therapies….
#10 Top Contract Manufacturing Companies
Website: contractmanufacturingcompanies.com
Key Highlights: Quickly connect with the top contract manufacturing companies in the United States who are ISO certified and offer premium products for affordable prices….
Expert Sourcing Insights for Contract Manufacturering

H2: 2026 Market Trends for Contract Manufacturing
As the global contract manufacturing landscape evolves toward 2026, several interrelated trends are reshaping the industry, driven by technological innovation, strategic repositioning, and shifting market demands. Companies across pharmaceuticals, electronics, medical devices, and consumer goods are increasingly relying on contract manufacturing organizations (CMOs) and contract development and manufacturing organizations (CDMOs), but with higher expectations for agility, integration, and value-added services.
H2: Accelerated Adoption of Smart Manufacturing and Digitalization
By 2026, digital transformation will be a cornerstone of competitive contract manufacturing. CMOs are investing heavily in Industry 4.0 technologies such as IoT-enabled equipment, AI-driven predictive maintenance, and real-time data analytics. Digital twins—virtual replicas of production systems—are becoming standard for optimizing throughput, reducing downtime, and ensuring product quality. Cloud-based manufacturing execution systems (MES) and enterprise resource planning (ERP) platforms enable seamless collaboration between clients and manufacturers, enhancing transparency and responsiveness. This digital backbone supports faster scale-up and regulatory compliance, especially in highly regulated sectors like biopharma.
H2: Geopolitical Realignment and Regionalization of Supply Chains
Ongoing geopolitical tensions, trade uncertainties, and recent supply chain disruptions have prompted a strategic shift toward regionalization. By 2026, there is a growing preference for nearshoring and friend-shoring, especially in North America, Europe, and parts of Southeast Asia. This trend is particularly evident in the pharmaceutical and semiconductor industries, where governments are incentivizing domestic and regional production to ensure supply security. Contract manufacturers are expanding facilities in Mexico, Eastern Europe, and India to serve as regional hubs, reducing dependency on single-source suppliers and minimizing logistical risks.
H2: Rise of Integrated CDMO Models in Life Sciences
In the pharmaceutical and biotech sectors, the traditional CMO model is rapidly evolving into the CDMO model, offering end-to-end services from drug development to commercial manufacturing. By 2026, CDMOs with strong R&D capabilities, regulatory expertise, and scalable manufacturing platforms (including mRNA and continuous bioprocessing) will dominate the market. The demand for personalized medicine and complex modalities like cell and gene therapies is fueling this integration, requiring CDMOs to offer flexible, adaptive production environments and faster time-to-market solutions.
H2: Sustainability as a Competitive Imperative
Environmental, social, and governance (ESG) criteria are increasingly influencing client selection. By 2026, leading contract manufacturers are expected to have robust sustainability roadmaps, including carbon footprint reduction, energy-efficient operations, waste minimization, and green chemistry practices. Clients—especially in consumer electronics and healthcare—are prioritizing partners with third-party verified green certifications. Water recycling, renewable energy integration, and circular economy principles in packaging and materials are becoming standard expectations.
H2: Talent Shortage and Workforce Transformation
As automation and advanced technologies grow, there is a widening skills gap in the contract manufacturing sector. By 2026, CMOs that invest in upskilling workforces, embracing human-machine collaboration, and fostering agile organizational cultures will have a strategic edge. Partnerships with technical institutions and apprenticeship programs are emerging as critical tools to secure talent in areas like data science, robotics, and advanced process engineering.
H2: Consolidation and Strategic Partnerships
The market is witnessing increased M&A activity among CMOs and CDMOs as companies seek scale, technological capabilities, and geographic reach. Smaller niche manufacturers are being acquired by larger players to enhance service portfolios. Simultaneously, long-term strategic partnerships—rather than transactional contracts—are becoming the norm, with clients co-investing in capacity expansion and technology upgrades to secure supply and drive innovation.
In conclusion, the 2026 contract manufacturing market will be defined by digital maturity, regional resilience, integration of services, sustainability, and strategic collaboration. Success will depend on a manufacturer’s ability to adapt quickly, deliver value beyond production, and align with the evolving priorities of global clients.
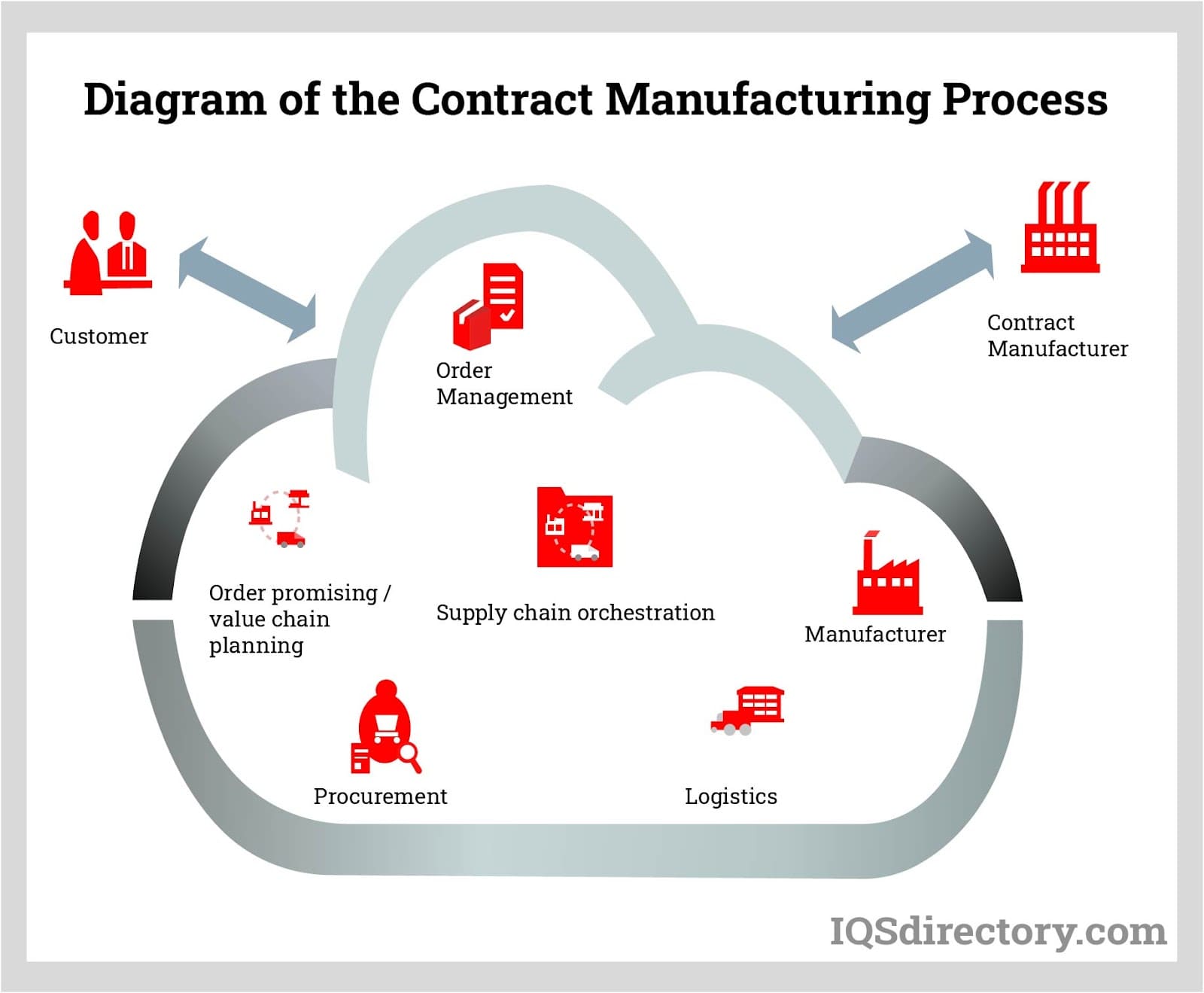
Common Pitfalls in Sourcing Contract Manufacturing: Quality and Intellectual Property Risks
When engaging a contract manufacturer (CM), companies often focus on cost and capacity while overlooking critical risks related to quality and intellectual property (IP). Failing to address these areas can lead to product failures, legal disputes, and reputational damage. Below are key pitfalls to avoid.
Quality Control Inadequacies
One of the most frequent issues in contract manufacturing is inconsistent product quality. This often stems from unclear specifications, insufficient oversight, or misaligned quality standards between the client and the CM. Without robust quality agreements, routine audits, and defined inspection protocols, defects may go undetected until products reach the market, resulting in recalls or customer dissatisfaction.
Lack of Transparency and Traceability
Many companies struggle with limited visibility into the CM’s production processes and supply chain. When materials or subcomponents are sourced from multiple tiers of suppliers, traceability becomes challenging. Without proper documentation and real-time monitoring, it’s difficult to ensure compliance with regulatory standards or to investigate quality issues effectively.
Inadequate Supplier Vetting and Due Diligence
Rushing into a partnership without thoroughly vetting a contract manufacturer can be disastrous. Overlooking certifications (e.g., ISO 13485 for medical devices or ISO 9001), audit histories, or past performance increases the risk of non-compliance and operational failures. Due diligence should include site visits, reference checks, and evaluation of the CM’s quality management system.
Weak or Absent Intellectual Property Protections
Sharing designs, formulations, or proprietary processes with a CM exposes sensitive IP. A common pitfall is relying on informal agreements or assuming that a standard NDA is sufficient. Without clearly defined IP ownership clauses in the manufacturing agreement, companies risk losing control over their innovations—especially in jurisdictions with lax IP enforcement.
Insufficient Control Over Manufacturing Know-How
Even when IP is legally protected, the CM may gain deep operational knowledge through repeated production. If not contractually restricted, the CM could use this know-how to serve competitors or develop similar products. Non-compete and non-circumvention clauses are essential but often neglected or poorly enforced.
Geographic and Jurisdictional Risks
Manufacturing in regions with lower costs often comes with higher IP and quality risks. Legal systems in some countries may not effectively uphold IP rights, and enforcement of contracts can be inconsistent. Companies must assess local regulatory environments and consider legal safeguards such as patent filings in the manufacturing country and escrow agreements for critical designs.
Poor Change Management and Communication
Unapproved process changes by the CM—such as substituting materials or altering production methods—can compromise product quality and safety. Without a formal change control process requiring client approval, these modifications may go unnoticed until problems arise, undermining consistency and compliance.
Over-Reliance on a Single Contract Manufacturer
Dependence on one CM increases exposure to both quality disruptions and IP misuse. If the relationship sours or the CM faces operational issues, the client may have limited recourse or alternatives. Diversifying manufacturing partners and maintaining in-house technical oversight can mitigate these risks.
By proactively addressing these pitfalls through comprehensive contracts, ongoing monitoring, and strong governance, companies can safeguard both product quality and intellectual property when sourcing contract manufacturing.

Logistics & Compliance Guide for Contract Manufacturing
Contract manufacturing involves outsourcing production to third-party manufacturers, making logistics and compliance critical for success. This guide outlines key considerations to ensure smooth operations, regulatory adherence, and supply chain efficiency.
Supply Chain Coordination
Effective logistics in contract manufacturing begins with seamless coordination across all parties. Establish clear communication channels between your organization, the contract manufacturer, and logistics providers. Define responsibilities for raw material procurement, component delivery, and finished goods transportation. Utilize integrated supply chain management (SCM) systems to track inventory levels, production schedules, and shipment statuses in real time.
Inventory Management & Warehousing
Maintain accurate inventory records to avoid overstocking or stockouts. Implement just-in-time (JIT) or vendor-managed inventory (VMI) models where appropriate to reduce holding costs. Ensure the contract manufacturer adheres to your inventory protocols and provides regular reporting. Secure warehousing solutions that meet product-specific requirements (e.g., temperature control, security, labeling standards).
Transportation & Distribution
Select reliable carriers with experience in your industry and geographic regions. Define service level agreements (SLAs) for delivery timelines, packaging standards, and freight handling. Optimize shipping routes and consolidate loads to reduce costs and environmental impact. Monitor transportation compliance, including customs documentation for international shipments and adherence to Incoterms® rules.
Regulatory Compliance
Ensure the contract manufacturer complies with all applicable local, national, and international regulations. This includes industry-specific standards such as FDA (food, pharmaceuticals), ISO certifications (e.g., ISO 9001, ISO 13485), REACH (chemicals), and RoHS (electronics). Verify that the manufacturer maintains proper documentation, conducts regular audits, and implements corrective actions as needed.
Quality Control & Product Standards
Establish clear quality specifications and inspection protocols in the manufacturing agreement. Require routine quality audits and batch testing. Define procedures for handling non-conforming products, including quarantine, rework, and disposal. Ensure traceability through lot numbering and batch tracking systems.
Import/Export Controls
For cross-border manufacturing, comply with export licensing requirements, import restrictions, and trade sanctions. Classify products accurately using Harmonized System (HS) codes and ensure proper documentation, including commercial invoices, packing lists, and certificates of origin. Stay updated on changes in trade agreements and tariffs that may impact costs and timelines.
Data Security & Intellectual Property Protection
Safeguard proprietary designs, formulas, and processes through legally binding non-disclosure agreements (NDAs) and intellectual property (IP) clauses. Limit access to sensitive data and ensure the manufacturer follows cybersecurity best practices. Audit data handling procedures regularly.
Sustainability & Ethical Sourcing
Align logistics and manufacturing practices with sustainability goals. Require suppliers and contract manufacturers to adhere to environmental standards (e.g., carbon footprint reduction, waste management) and ethical labor practices. Conduct supplier audits and consider certifications such as Fair Trade or Responsible Business Alliance (RBA) membership.
Risk Management & Business Continuity
Identify potential risks, including supply chain disruptions, geopolitical issues, and natural disasters. Develop contingency plans, such as dual sourcing or safety stock. Regularly review and update business continuity strategies with input from the contract manufacturer.
Audit & Performance Monitoring
Conduct regular audits of the contract manufacturer’s logistics and compliance practices. Use key performance indicators (KPIs) such as on-time delivery rate, defect rate, and compliance audit scores to assess performance. Maintain a collaborative relationship to drive continuous improvement.
By addressing these logistics and compliance areas, companies can mitigate risks, ensure product quality, and maintain regulatory adherence in contract manufacturing partnerships.
Conclusion for Sourcing Contract Manufacturing
In conclusion, sourcing contract manufacturing offers a strategic advantage for companies aiming to enhance operational efficiency, reduce production costs, and accelerate time-to-market. By leveraging the expertise, infrastructure, and scalability of specialized manufacturing partners, businesses can focus on core competencies such as product development, marketing, and customer engagement. However, successful implementation requires thorough due diligence in selecting the right partner, clear contractual agreements, effective communication, and ongoing quality oversight.
When managed effectively, contract manufacturing supports flexibility, geographic expansion, and access to advanced technologies—enabling organizations to remain competitive in dynamic global markets. As supply chains evolve and demand for customization grows, a well-structured contract manufacturing strategy will continue to be a critical component of sustainable growth and long-term success.