The global computed tomography (CT) market is experiencing robust growth, driven by rising demand for early disease diagnosis, technological advancements in imaging, and increasing prevalence of chronic diseases. According to Grand View Research, the global CT imaging market was valued at USD 5.7 billion in 2023 and is projected to expand at a compound annual growth rate (CAGR) of 5.8% from 2024 to 2030. Similarly, Mordor Intelligence forecasts a CAGR of approximately 5.6% over the same period, citing the growing adoption of advanced imaging systems in emerging economies and continuous innovations such as photon-counting CT and AI-enhanced image reconstruction. As healthcare systems prioritize precision diagnostics and minimally invasive procedures, the role of leading CT manufacturers in shaping the future of radiology has become increasingly critical. This list highlights the top 10 computed tomography manufacturers leading the market through innovation, global reach, and technological excellence.
Top 10 Computed Tomography Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Lumafield
Domain Est. 2020
Website: lumafield.com
Key Highlights: Lumafield’s advanced 3D X-ray technology is taking manufacturing into new frontiers of speed, experience, and possibility….
#2 Computed tomography equipment
Domain Est. 1997
Website: planmed.com
Key Highlights: Planmed Verity utilizes CBCT technology (Cone Beam Computed Tomography) that provides high resolution 3D images of the extremities and maxillofacial area….
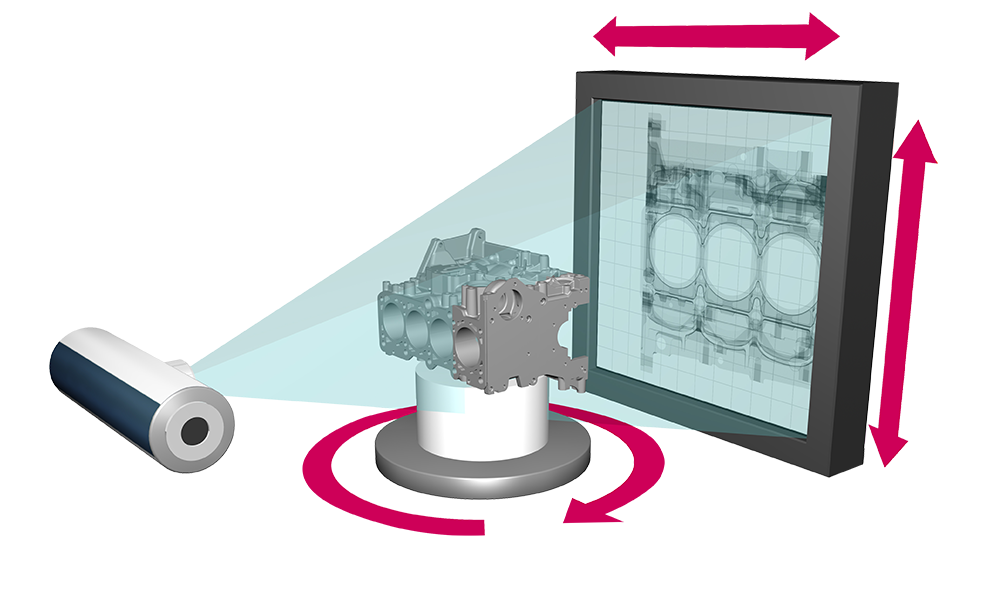
#3 Industrial CT Systems & X
Domain Est. 2003
Website: 4nsi.com
Key Highlights: Enhance your quality control, detect internal defects, and ensure precise internal measurements with our industrial CT systems and X-ray scanning services….
#4 CT systems
Domain Est. 1987
Website: usa.philips.com
Key Highlights: Discover our range of CT systems that can help keep you ahead in radiology, cardiology, oncology, neurology, trauma, interventional radiology and radiation ……
#5 Analogic
Domain Est. 1994
Website: analogic.com
Key Highlights: We develop and deliver enabling technologies used in computed tomography (CT), digital mammography (DM), and magnetic resonance imaging (MRI)….
#6 Computed Tomography
Domain Est. 1999
Website: gehealthcare.com
Key Highlights: Our family of innovative CT products let you match a system to your exact imaging needs—so you can focus on better patient care….
#7 NeuroLogica
Domain Est. 1999
Website: neurologica.com
Key Highlights: We transform fixed imaging technologies into point-of-care platforms, bringing the power of innovative imaging to your patients….
#8 MARS Bioimaging
Domain Est. 2007
Website: marsbioimaging.com
Key Highlights: High-resolution color CT imaging for preclinical research, complete with advanced imaging analysis tools….
#9 Computed Tomography
Domain Est. 2016
Website: siemens-healthineers.com
Key Highlights: Siemens Healthineers strives to deliver CT innovation that matters, through our trusted SOMATOM portfolio of CT scanners, and now the NAEOTOM Alpha….
#10 Computed Tomography
Domain Est. 2017
Website: global.medical.canon
Key Highlights: Stand out with a CT system that optimizes your workflow, improves patient experience, and helps you to keep the dose low and the image quality high….
Expert Sourcing Insights for Computed Tomography

2026 Market Trends for Computed Tomography
The global computed tomography (CT) market is poised for significant transformation by 2026, driven by technological innovation, evolving clinical demands, and shifting healthcare dynamics. Key trends shaping the landscape include:
Artificial Intelligence Integration and Intelligent Imaging
AI is transitioning from a supplementary tool to a core component of CT systems. By 2026, AI-powered applications will be standard across the imaging workflow—enabling automated protocol selection, real-time motion correction, advanced image reconstruction (reducing radiation dose by up to 50–70%), and AI-assisted diagnostics such as nodule detection in lung cancer screening. Vendors are increasingly embedding AI directly into scanner hardware and cloud platforms, accelerating image processing and improving diagnostic confidence.
Expansion of Photon-Counting CT (PCCT) Technology
Photon-counting CT, once limited to research and elite institutions, is expected to gain broader clinical adoption by 2026. PCCT offers superior spatial resolution, improved contrast-to-noise ratio, and the ability to perform multi-energy imaging without spectral switching. Early applications in cardiovascular imaging, oncology, and musculoskeletal diagnostics are demonstrating clinical value, prompting major manufacturers to scale production and reduce costs—making PCCT more accessible to large hospitals and academic centers.
Focus on Dose Reduction and Patient-Centric Design
Patient safety remains a priority, pushing manufacturers to innovate in low-dose imaging. Advances in detector efficiency, iterative reconstruction algorithms, and AI-driven denoising are enabling high-quality diagnostics at significantly reduced radiation exposure. Concurrently, ergonomic scanner designs with wider bores, quieter operation, and ambient lighting are improving patient comfort—especially beneficial for pediatric, claustrophobic, and obese populations.
Growth in Point-of-Care and Compact CT Systems
The demand for rapid diagnostics in emergency departments, intensive care units, and outpatient clinics is fueling the growth of compact and mobile CT scanners. By 2026, specialized low-cost, low-footprint systems—particularly head and extremity CTs—are expected to expand into ambulatory surgery centers and rural healthcare settings. These systems support stroke triage, trauma assessment, and orthopedic imaging outside traditional radiology departments.
Consolidation of Hybrid Imaging and Workflow Integration
Integration of CT with other modalities such as PET-CT and emerging intraoperative CT systems will continue to grow, particularly in oncology and interventional radiology. Furthermore, seamless integration of CT data into electronic health records (EHRs), picture archiving and communication systems (PACS), and clinical decision support platforms will enhance workflow efficiency and enable longitudinal patient monitoring.
Emerging Markets and Infrastructure Development
Regions including Asia-Pacific, Latin America, and parts of Africa are expected to see accelerated CT adoption due to rising healthcare investments, expanding insurance coverage, and growing burdens of chronic diseases. Governments and private providers are prioritizing diagnostic infrastructure, creating opportunities for cost-effective, service-oriented CT solutions tailored to diverse economic environments.
In summary, by 2026, the CT market will be defined by smarter, safer, and more accessible imaging technologies, with AI and photon-counting detectors leading a new era of precision diagnostics. Success will depend on balancing innovation with cost-effectiveness and clinical integration across global healthcare ecosystems.

Common Pitfalls in Sourcing Computed Tomography: Quality and Intellectual Property Concerns
Sourcing computed tomography (CT) services—whether for industrial inspection, medical applications, or research—can introduce significant challenges related to data quality and intellectual property (IP) protection. Organizations often overlook these critical aspects, leading to compromised results or legal exposure. Below are the most common pitfalls to avoid.
Inadequate Quality Assurance and Validation Processes
One of the foremost risks in sourcing CT scanning is receiving data that does not meet the required quality standards. This often stems from insufficient validation procedures by the service provider. Pitfalls include inconsistent scan resolution, misalignment of reconstructed images, or artifacts due to improper scanning parameters. Without clear service level agreements (SLAs) defining minimum quality metrics—such as spatial resolution, signal-to-noise ratio, or dimensional accuracy—clients may receive unusable or misleading data. Additionally, third-party providers may use outdated equipment or software, further degrading output quality.
Lack of Standardized Protocols and Documentation
Different CT service providers may employ varying scanning protocols, reconstruction algorithms, and calibration methods. When these are not standardized or fully documented, it becomes difficult to reproduce results or compare data across different batches or suppliers. This lack of transparency can undermine confidence in the data, especially in regulated industries such as aerospace or medical device manufacturing where traceability is essential.
Insufficient Expertise and Training of Service Providers
CT scanning and data interpretation require specialized technical knowledge. Sourcing from providers lacking experienced personnel can lead to misinterpretation of scan results, missed defects, or incorrect dimensional measurements. This is particularly problematic when automated defect recognition (ADR) software is used without proper validation by a trained radiographer or metrology expert. Clients may unknowingly accept flawed analyses due to overreliance on provider expertise.
Ambiguous Ownership and Usage Rights for Scanned Data
A major IP-related pitfall is the failure to clearly define who owns the CT data and derived insights. Some providers retain rights to scanned datasets or use them for benchmarking, marketing, or training AI models without client consent. Without a comprehensive contract specifying data ownership, usage limitations, and confidentiality, companies risk losing control over sensitive design information, especially when scanning proprietary components or pre-release products.
Inadequate Data Security and Confidentiality Measures
CT data often contains detailed geometric and material information that can reveal trade secrets. If the service provider lacks robust cybersecurity protocols—such as encrypted data transfer, secure storage, and restricted access—there is a heightened risk of data breaches or unauthorized sharing. Furthermore, providers operating in jurisdictions with weaker data protection laws may expose clients to compliance risks under regulations like GDPR or ITAR.
Unapproved Data Retention and Secondary Use
Many CT service agreements do not explicitly address data retention policies. Providers may store scanned datasets indefinitely or reuse them for internal purposes unless contractually prohibited. This creates potential IP leakage, especially when scanning competitive or innovative products. Clients should ensure contracts include clauses mandating data deletion after project completion and prohibit any secondary use of the data.
Failure to Address Regulatory and Compliance Requirements
In highly regulated sectors, such as healthcare or defense, the use of external CT services must comply with industry-specific standards (e.g., FDA 21 CFR Part 11, ISO 13485, or AS9100). Sourcing from non-compliant providers can invalidate certifications or audit trails. Clients must verify that the provider adheres to relevant regulatory frameworks and maintains proper documentation for quality management systems.
Conclusion
To mitigate these pitfalls, organizations should establish clear contractual terms covering data quality specifications, IP ownership, confidentiality, and compliance. Conducting due diligence on providers’ technical capabilities, security practices, and regulatory adherence is essential. Proactive management of both quality and IP concerns ensures that outsourced CT services deliver reliable results while protecting valuable intellectual assets.
Logistics & Compliance Guide for Computed Tomography
Equipment Procurement and Installation
When acquiring a computed tomography (CT) scanner, institutions must follow strict procurement protocols to ensure regulatory compliance and technical suitability. This includes verifying that the equipment meets FDA 510(k) clearance or PMA approval in the United States, or equivalent CE marking in the European Union. Site planning must account for radiation shielding requirements, electrical infrastructure, cooling systems, and space for ancillary equipment. A qualified medical physicist should be involved early in the installation process to conduct radiation safety assessments and room design validation.
Regulatory Compliance and Licensing
CT operations are subject to federal, state, and local regulations. In the U.S., facilities must comply with regulations from the Food and Drug Administration (FDA), the Centers for Medicare & Medicaid Services (CMS), and state radiation control programs. Facilities participating in Medicare must meet Conditions of Participation and be accredited by an approved body such as the American College of Radiology (ACR). All CT scanners must be registered with the appropriate state radiation protection agency, and operators must hold valid certifications (e.g., ARRT in radiography or radiologic technology).
Radiation Safety and Dose Monitoring
Strict radiation safety protocols are required to protect patients, staff, and the public. Facilities must appoint a Radiation Safety Officer (RSO) and implement an ALARA (As Low As Reasonably Achievable) program. Regular dose audits using standardized metrics (e.g., CTDIvol, DLP) should be conducted, and dose tracking software is recommended to monitor patient exposure. All CT protocols must be optimized for patient size and clinical indication, and routine quality control tests—such as CT number accuracy, uniformity, and spatial resolution checks—must be performed according to ACR or AAPM guidelines.
Personnel Training and Credentialing
All personnel operating CT equipment must undergo comprehensive training in radiation safety, scanner operation, emergency procedures, and patient care. Technologists should hold current certification from recognized bodies (e.g., ARRT, NMTCB). Continuing education is mandatory to maintain licensure and stay current with technological advances. Physicians interpreting CT studies must be board-certified in radiology or a relevant specialty and maintain ongoing credentialing through their institution’s medical staff office.
Data Management and HIPAA Compliance
CT imaging generates large volumes of sensitive patient data that must be protected under HIPAA and other data privacy regulations. Facilities must implement secure Picture Archiving and Communication Systems (PACS) with encrypted storage and transmission. Access to imaging data should be role-based and logged for audit purposes. Disaster recovery and data backup plans are essential to ensure data integrity and availability. All staff must complete HIPAA training and adhere to institutional privacy policies.
Maintenance and Quality Assurance
A formal quality assurance (QA) program is critical for maintaining CT system performance and patient safety. Daily, weekly, monthly, and annual QA tests must be documented and reviewed by a medical physicist. Preventive maintenance schedules provided by the manufacturer should be strictly followed. Service contracts should include response time guarantees and regulatory compliance support. Records of all repairs, calibrations, and performance evaluations must be retained for audit and accreditation purposes.
Accreditation and Continuous Improvement
Facilities should pursue accreditation through recognized organizations such as the ACR or Intersocietal Accreditation Commission (IAC). Accreditation requires documentation of policies, personnel qualifications, dose management, QA results, and peer review processes. Participating in national dose registries (e.g., ACR Dose Index Registry) supports benchmarking and continuous quality improvement. Regular internal audits and staff feedback mechanisms help identify areas for operational and clinical enhancement.
In conclusion, sourcing computed tomography (CT) manufacturers requires a strategic and comprehensive approach to ensure the acquisition of high-quality, reliable, and cost-effective imaging systems. It is essential to evaluate manufacturers based on key criteria such as technological innovation, regulatory compliance (e.g., FDA, CE marking), product reliability, service and support infrastructure, and total cost of ownership. Leading global manufacturers like Siemens Healthineers, GE Healthcare, Philips, Canon Medical Systems, and United Imaging offer advanced CT solutions with varying strengths in image resolution, scanning speed, radiation dose management, and AI integration.
Conducting thorough due diligence—including site visits, customer references, and performance evaluations—helps in selecting a partner aligned with clinical needs, budget constraints, and long-term healthcare goals. Additionally, considering local service support, training availability, and upgrade paths is crucial for maximizing uptime and system longevity. As healthcare continues to evolve with digital transformation and precision medicine, partnering with a reputable CT manufacturer not only enhances diagnostic capabilities but also supports improved patient outcomes and operational efficiency.