The global cold storage market is experiencing robust expansion, driven by rising demand for temperature-controlled supply chains across the food & beverage, pharmaceutical, and healthcare sectors. According to Grand View Research, the market was valued at USD 238.0 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 15.5% from 2023 to 2030. This surge is fueled by increasing perishable goods trade, stricter food safety regulations, and the rapid growth of e-commerce for refrigerated products. Additionally, Mordor Intelligence forecasts a CAGR of over 17% during the period 2023–2028, highlighting the critical role of advanced cold chain infrastructure in emerging economies. As demand escalates, leading cold storage manufacturers are innovating with energy-efficient, automated, and IoT-enabled solutions to meet scalability and sustainability needs. In this dynamic landscape, the top 10 cold storage companies stand out through technological leadership, global reach, and comprehensive service offerings that support resilient cold chain ecosystems.
Top 10 Cold Storage Companies Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Cold Storage Solutions
Domain Est. 1997
Website: thermoking.com
Key Highlights: Thermo King cold storage solutions offer various customizations and upgrades on our Magnum Plus and SuperFreezer units….
#2 to LaCold
Domain Est. 1998
Website: lacold.com
Key Highlights: Los Angeles Cold Storage offers a variety of inspection services including FDA, USDA and USDC, in our modern, state of the art facilities….
#3 United States Cold Storage
Domain Est. 1999
Website: uscold.com
Key Highlights: As industry leaders, we provide unparalleled cold storage solutions and logistics services, positioning ourselves at the forefront of the cold chain….
#4 Cold Chain Technologies
Domain Est. 2001
Website: coldchaintech.com
Key Highlights: Cold Chain Technologies is a provider of insulated packaging solutions ensuring product safety for the life sciences industry….
#5 Cold chain logistics & Advanced automated warehouse
Domain Est. 2009
Website: newcold.com
Key Highlights: NewCold offers food companies highly automated and sustainable cold storage and cold chain logistics solutions. Explore our global network….
#6 Arctic Cold
Domain Est. 2016
Website: arcticllc.com
Key Highlights: With decades of experience in distribution and storage, we specialize in handling everything from frozen to fresh commodities with precision and care….
#7 RealCold
Domain Est. 2019
Website: realcold.com
Key Highlights: Explore RealCold’s cold chain storage facilities, offering flexible, multi-temperature storage and value added services….
#8 Agile Cold Storage
Domain Est. 2019
Website: agilecoldstorage.com
Key Highlights: Discover automated, multi-temperature and conventional warehousing services at Agile Cold Storage. Get customized cold storage solutions for your products….
#9 Vertical Cold Storage
Domain Est. 2021
Website: verticalcold.com
Key Highlights: Vertical Cold Storage specializes in innovative cold storage and logistics solutions, offering services like blast freezing, import/export handling, ……
#10 Arcadia Cold Storage & Logistics
Domain Est. 2021
Website: arcadiacold.com
Key Highlights: Arcadia is a 3PL powered by years of experience. Our modern facilities and focus on service represents the new age of cold chain….
Expert Sourcing Insights for Cold Storage Companies
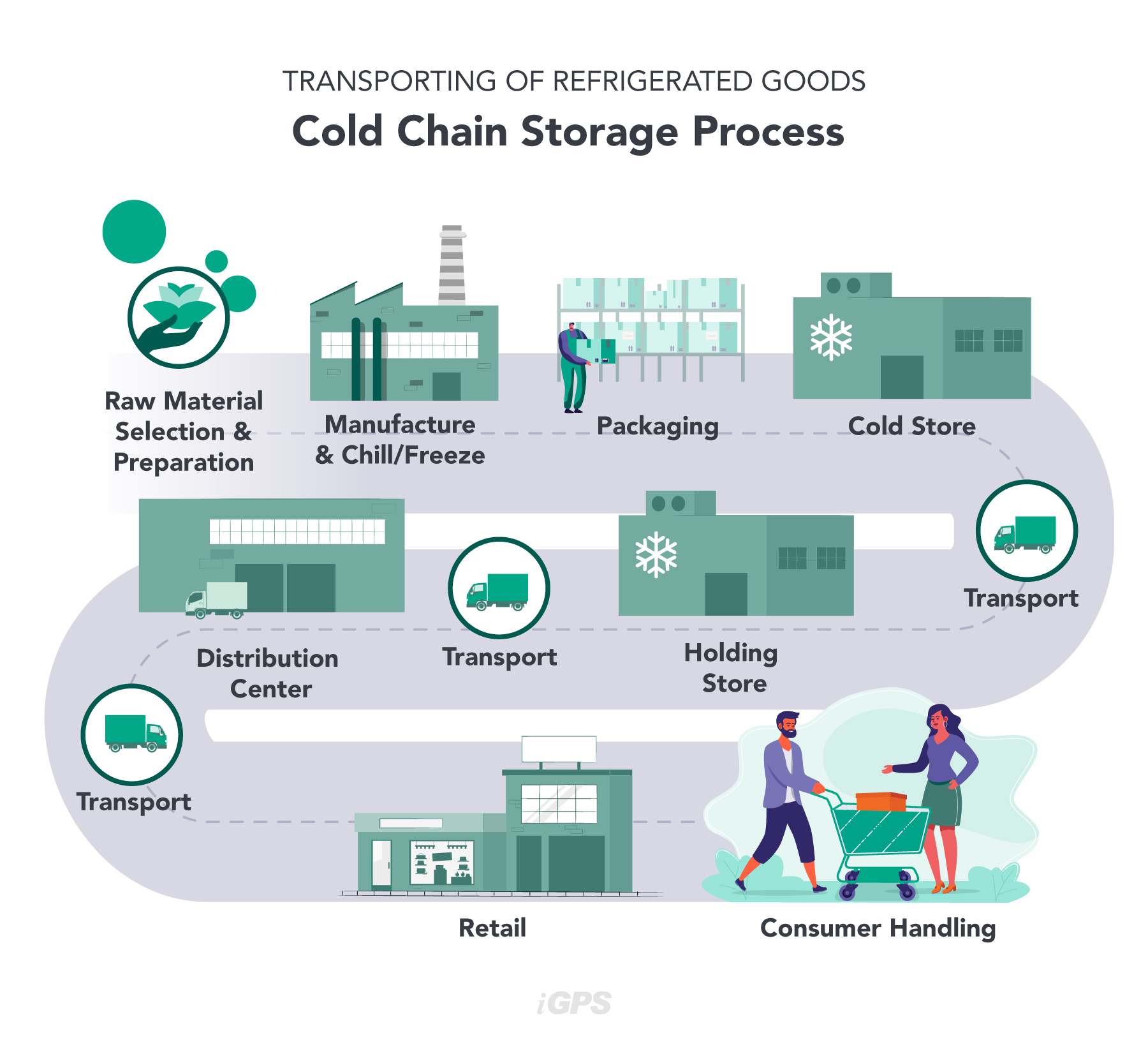
H2: Cold Storage Market Trends in 2026
By 2026, the global cold storage market is poised for significant transformation, driven by evolving consumer behaviors, technological innovation, and increasing regulatory demands. Here’s a strategic analysis of the key trends shaping the industry:
1. Exponential Growth in Demand
The cold chain infrastructure will continue to expand rapidly. Driven by rising demand for perishable goods—including fresh produce, dairy, meat, pharmaceuticals (especially biologics and vaccines), and e-grocery—global cold storage capacity is projected to grow at a CAGR of 15–18% through 2026. Emerging markets in Asia-Pacific, Latin America, and Africa will lead expansion due to urbanization and improving supply chains.
2. Digitalization and Smart Cold Storage
Cold storage facilities will increasingly adopt IoT-enabled sensors, AI-driven analytics, and blockchain for real-time temperature monitoring, predictive maintenance, and supply chain transparency. By 2026, over 60% of large-scale cold storage operators are expected to implement integrated digital platforms to optimize energy use, reduce spoilage, and ensure compliance.
3. Sustainability and Energy Efficiency
Environmental regulations and ESG (Environmental, Social, and Governance) pressures will push companies toward energy-efficient technologies. Adoption of natural refrigerants (e.g., CO₂, ammonia), solar-powered facilities, and advanced insulation materials will become standard. Facilities achieving LEED or BREEAM certifications will gain competitive advantage.
4. Last-Mile Cold Chain Innovation
With the e-commerce grocery sector projected to exceed $1 trillion globally by 2026, last-mile delivery solutions—such as insulated electric delivery vans, smart lockers with refrigeration, and drone deliveries in select regions—will become critical. Cold storage providers will increasingly partner with logistics firms to offer end-to-end cold chain solutions.
5. Pharmaceutical Cold Chain Expansion
The growth of mRNA vaccines, cell and gene therapies, and biologics will require ultra-low temperature (ULT) storage (-70°C or lower). This will drive demand for specialized cold storage infrastructure, including GMP-compliant warehouses and temperature-controlled distribution hubs, especially near research centers and hospitals.
6. Consolidation and Strategic Partnerships
Market consolidation will accelerate as large players acquire regional cold storage operators to expand geographic reach and enhance service offerings. Strategic alliances between cold storage providers, food retailers, and pharma companies will foster vertically integrated cold chain ecosystems.
7. Regulatory and Compliance Pressures
Stringent food safety (e.g., FDA FSMA, EU Food Law) and pharmaceutical regulations (e.g., GDP guidelines) will mandate higher traceability and audit readiness. Cold storage companies will invest in automated compliance systems and digital documentation to meet these requirements efficiently.
8. Labor Automation and Robotics
Labor shortages and rising wages will drive investment in automation. Automated Storage and Retrieval Systems (AS/RS), robotic palletizers, and autonomous guided vehicles (AGVs) will become common in new cold storage warehouses, improving throughput and reducing human error.
Conclusion:
By 2026, cold storage companies that embrace digital transformation, prioritize sustainability, and adapt to pharmaceutical and e-commerce logistics demands will lead the market. Success will hinge on agility, technological integration, and the ability to deliver secure, transparent, and efficient cold chain solutions across diverse verticals.

Common Pitfalls When Sourcing Cold Storage Companies (Quality, IP)
Sourcing cold storage companies for temperature-sensitive products—such as pharmaceuticals, biologics, or perishable goods—requires careful due diligence. Overlooking key risks can lead to compromised product integrity, regulatory non-compliance, and intellectual property (IP) exposure. Below are common pitfalls related to quality and IP that organizations should avoid:
Inadequate Temperature Monitoring and Validation
Many cold storage providers claim compliance with temperature requirements but lack robust monitoring systems. A common mistake is relying on spot checks or infrequent data logging instead of continuous, real-time monitoring with automated alerts. Without proper validation of storage conditions (e.g., thermal mapping of warehouses), companies risk product degradation due to undetected temperature excursions.
Lack of Regulatory Compliance and Audit Trail Gaps
Choosing a provider without adherence to industry standards—such as GDP (Good Distribution Practice), GMP (Good Manufacturing Practice), or FDA 21 CFR Part 11—can result in audit failures and product recalls. Poor documentation practices, including incomplete temperature logs or missing chain-of-custody records, undermine quality assurance and regulatory readiness.
Insufficient Contingency Planning
Power outages, equipment failures, or natural disasters can disrupt cold chain integrity. Sourcing a provider without tested backup systems (e.g., redundant refrigeration, emergency generators, or disaster recovery plans) exposes products to spoilage. Evaluating business continuity plans during due diligence is essential to ensure operational resilience.
Overlooking Staff Training and Qualification
Human error is a significant risk factor. Cold storage facilities must have trained personnel who understand handling procedures, emergency protocols, and regulatory requirements. Providers with high staff turnover or inadequate training programs increase the likelihood of mishandling, compromising product quality.
Insecure Data Handling and Intellectual Property Risks
Cold storage partners often have access to sensitive shipment data, batch records, and product specifications. A major IP pitfall arises when providers lack secure data systems or clear confidentiality agreements. Unauthorized access, data breaches, or weak cybersecurity protocols can lead to IP theft or competitive exposure.
Ambiguous Contracts and IP Ownership Clauses
Contracts that fail to define ownership of process data, storage methodologies, or custom handling procedures may inadvertently grant the provider rights to proprietary information. Without explicit IP protection clauses, companies risk losing control over innovations developed during the partnership.
Geographic and Logistical Vulnerabilities
Selecting a facility based solely on cost or proximity without evaluating local infrastructure (e.g., grid reliability, transportation networks) can compromise quality. Additionally, storing IP-sensitive materials in jurisdictions with weak IP enforcement increases legal and operational risks.
Failure to Conduct On-Site Audits
Paper certifications and self-reported audits are insufficient. Not performing physical audits to verify equipment calibration, storage conditions, and data security measures leaves critical quality and IP risks undetected. On-site assessments are vital to confirm compliance and operational integrity.
Avoiding these pitfalls requires a structured sourcing process that includes technical evaluations, legal review of contracts, cybersecurity assessments, and ongoing performance monitoring. Prioritizing quality systems and IP protection safeguards both product integrity and competitive advantage.

Logistics & Compliance Guide for Cold Storage Companies
Cold storage companies play a critical role in the global supply chain, ensuring the safe handling, storage, and transportation of temperature-sensitive goods such as food, pharmaceuticals, and chemicals. To maintain product integrity, customer trust, and legal compliance, these businesses must adhere to rigorous logistics and regulatory standards. This guide outlines key areas every cold storage provider should address.
Temperature Control & Monitoring
Maintaining precise temperature environments is the cornerstone of cold storage operations. Deviations can lead to spoilage, safety hazards, and regulatory violations.
- Calibrated Monitoring Systems: Install reliable, calibrated temperature monitoring devices in all storage zones (e.g., freezer, chill, ambient). Use real-time digital data loggers with remote access.
- Alarm Systems: Implement automated alerts for temperature excursions, power outages, or equipment failure. Ensure 24/7 monitoring and response protocols.
- Temperature Mapping: Conduct regular temperature mapping studies to identify hot or cold spots within storage areas and validate uniform conditions.
Facility Design & Maintenance
Facility infrastructure must support consistent cold chain integrity and hygiene.
- Insulation & Sealing: Use high-quality insulation materials and ensure door seals, walls, and roofs are properly maintained to minimize heat transfer.
- Refrigeration Systems: Maintain redundant or backup refrigeration systems to prevent total failure. Schedule preventive maintenance quarterly.
- Zoning & Segregation: Separate storage areas by temperature requirements and product type (e.g., raw vs. cooked, pharmaceutical vs. food). Prevent cross-contamination.
Cold Chain Transportation
Transit is a high-risk phase in the cold chain. Logistics must ensure seamless temperature control from warehouse to delivery.
- Reefer Compliance: Use refrigerated transport units (reefers) that meet industry standards (e.g., ATP in Europe, FDA requirements in the U.S.). Validate performance regularly.
- Pre-Trip Inspections: Confirm equipment is pre-cooled and functioning before loading. Document pre-trip checklists.
- In-Transit Monitoring: Equip vehicles with GPS-enabled temperature loggers. Share real-time data with clients and internal teams.
- Loading Protocols: Minimize door-open time during loading/unloading. Use dock shelters and levelers to reduce ambient exposure.
Regulatory Compliance
Cold storage operations are subject to multiple local, national, and international regulations.
- Food Safety Modernization Act (FSMA): Comply with preventive controls, sanitary transportation rules, and recordkeeping requirements (U.S.).
- HACCP & HARPC: Implement hazard analysis plans tailored to your operations. Document critical control points and corrective actions.
- FDA & USDA Regulations: Adhere to labeling, inspection, and reporting obligations for food and animal products.
- Pharmaceutical Standards (if applicable): Follow GDP (Good Distribution Practices), 21 CFR Part 11 (electronic records), and EU GDP guidelines for temperature-sensitive medicines.
- International Standards: For cross-border operations, comply with ATP (Accord Relatif aux Transports Internationaux de denrées Périssables) and Codex Alimentarius.
Sanitation & Pest Control
Cleanliness is essential to prevent contamination and meet health code standards.
- Scheduled Cleaning: Establish SOPs for routine cleaning and sanitization of storage areas, equipment, and transport units.
- Pest Management: Partner with licensed pest control providers. Conduct regular inspections and maintain logs.
- Allergen Control: Prevent cross-contact, especially in food storage. Clearly label allergen-containing products.
Documentation & Recordkeeping
Accurate documentation supports traceability, compliance audits, and incident investigations.
- Temperature Logs: Maintain automated, tamper-proof logs with timestamps for at least two years.
- Maintenance Records: Document all equipment servicing, repairs, and calibration results.
- Training Records: Keep records of employee training in hygiene, safety, and compliance procedures.
- Audit Trails: Preserve records of internal and third-party audits.
Staff Training & Safety
Employees are vital to maintaining compliance and operational excellence.
- Compliance Training: Train staff annually on FSMA, HACCP, hygiene, allergen control, and emergency procedures.
- Cold Stress Awareness: Educate workers on the risks of prolonged exposure to cold environments and provide appropriate protective gear.
- GMP & SOP Adherence: Reinforce proper handling techniques, handwashing, and use of PPE.
Emergency Preparedness
Develop plans to respond to disruptions that could compromise product safety.
- Power Outage Plan: Include backup generators, emergency contacts, and protocols for relocating high-risk inventory.
- Recall Procedures: Establish a rapid product recall process with traceability systems (e.g., lot tracking, barcoding).
- Disaster Recovery: Prepare for natural disasters, fires, or cyberattacks with business continuity plans.
Sustainability & Energy Efficiency
Cold storage is energy-intensive. Operational improvements can reduce costs and environmental impact.
- Energy Audits: Conduct regular audits to identify inefficiencies in lighting, refrigeration, and insulation.
- LED Lighting & Motion Sensors: Reduce energy use in storage areas.
- Heat Recovery Systems: Capture waste heat from refrigeration units for other uses (e.g., heating office spaces).
- Renewable Energy: Consider solar or geothermal options where feasible.
By following this comprehensive guide, cold storage companies can ensure product safety, meet regulatory obligations, and deliver reliable, efficient logistics services in an increasingly demanding market. Regular audits, staff engagement, and investment in technology are key to long-term success.
In conclusion, sourcing cold storage companies requires a strategic and comprehensive approach that balances cost, reliability, technological capability, and geographic coverage. Key factors to consider include temperature control accuracy, compliance with food safety and regulatory standards (such as FDA, HACCP, or GMP), facility location and distribution network efficiency, scalability, and the provider’s track record in maintaining product integrity. Additionally, evaluating the level of customer service, transparency, and integration capabilities with your supply chain systems can significantly impact operational success.
Partnering with a reputable cold storage provider not only ensures the preservation of product quality and safety but also enhances supply chain resilience and responsiveness. Conducting thorough due diligence, requesting site visits, and obtaining references or case studies can help in making an informed decision. Ultimately, the right cold storage partner should act as an extension of your business, supporting your growth, compliance, and customer satisfaction goals in the temperature-sensitive supply chain.