Sourcing Guide Contents
Industrial Clusters: Where to Source China Dental Assistant Stool Manufacturers

SourcifyChina | B2B Sourcing Report 2026
Title: Deep-Dive Market Analysis: Sourcing Dental Assistant Stools from China
Prepared For: Global Procurement Managers
Date: April 5, 2026
Author: Senior Sourcing Consultant, SourcifyChina
Executive Summary
China remains the dominant global manufacturing hub for medical and dental furniture, including dental assistant stools. With rising demand for ergonomic, durable, and cost-effective solutions in dental clinics worldwide, sourcing from China offers significant advantages in scalability, innovation, and competitive pricing. This report provides a comprehensive analysis of key industrial clusters producing dental assistant stools in China, with a comparative evaluation of provinces and cities based on price, quality, and lead time—three critical KPIs for global procurement decision-making.
The primary manufacturing hubs are concentrated in Guangdong and Zhejiang provinces, with emerging activity in Jiangsu and Shandong. These regions benefit from mature supply chains, specialized labor, and proximity to export ports, enabling efficient global distribution.
Key Industrial Clusters for Dental Assistant Stool Manufacturing
Dental assistant stool manufacturing in China is highly regionalized, with clusters forming around established medical equipment and metal/furniture manufacturing zones. The top provinces and cities include:
1. Guangdong Province
- Core Cities: Foshan, Guangzhou, Shenzhen
- Industrial Focus: Medical furniture, precision metal fabrication, ergonomic product design
- Advantages: Proximity to Hong Kong port, strong export infrastructure, high concentration of ISO-certified medical equipment manufacturers
- Notable Features: High automation levels; strong R&D in ergonomic and hydraulic systems
2. Zhejiang Province
- Core Cities: Ningbo, Wenzhou, Hangzhou
- Industrial Focus: Metal stamping, chair mechanisms, mid-to-high-end furniture components
- Advantages: Competitive pricing, strong component supply chain (gas lifts, casters, upholstery), growing focus on OEM/ODM services
- Notable Features: High volume output; many suppliers serve EU/US markets with CE/FDA-compliant designs
3. Jiangsu Province
- Core Cities: Suzhou, Changzhou
- Industrial Focus: Precision engineering, medical device integration
- Advantages: Near Shanghai port; skilled labor; integration with smart clinic furniture trends
- Emerging Trend: Rise of smart stools with adjustable lumbar support and IoT-enabled features
4. Shandong Province
- Core Cities: Qingdao, Jinan
- Industrial Focus: Heavy-duty metal fabrication, cost-effective mass production
- Advantages: Lower labor and operational costs; suitable for budget-oriented procurement
- Limitations: Fewer specialized dental furniture OEMs; more general-purpose furniture producers
Comparative Analysis of Key Production Regions
The table below compares the top two manufacturing clusters—Guangdong and Zhejiang—on core procurement metrics. Jiangsu and Shandong are included for strategic context.
| Region | Average Unit Price (USD) | Quality Tier | Typical Lead Time (Days) | Certifications Commonly Held | Best For |
|---|---|---|---|---|---|
| Guangdong | $85 – $150 | High (Premium/Professional) | 30 – 45 | ISO 13485, CE, FDA, RoHS, BIFMA | High-end clinics, branded OEMs, EU/US compliance |
| Zhejiang | $65 – $110 | Medium to High | 35 – 50 | CE, ISO 9001, RoHS | Mid-market volume buyers, private label brands |
| Jiangsu | $75 – $130 | Medium (Improving) | 40 – 55 | ISO 9001, CE (select suppliers) | Tech-integrated solutions, smart furniture |
| Shandong | $50 – $85 | Medium (Standard/Basic) | 45 – 60 | ISO 9001 (limited medical certification) | Budget procurement, emerging markets |
Notes:
– Prices based on FOB terms for 500+ unit orders, standard hydraulic models with PU upholstery.
– Lead times include production + pre-shipment QC; excludes shipping.
– Quality tier reflects material durability, mechanism reliability, and compliance readiness.
Strategic Sourcing Recommendations
-
For Premium Quality & Compliance:
Source from Guangdong (Foshan/Guangzhou). Ideal for clients targeting North American and European markets requiring full regulatory alignment. -
For Cost-Effective Mid-Tier Supply:
Zhejiang (Ningbo/Wenzhou) offers the best balance of price and quality, with reliable OEM capabilities and strong component sourcing. -
For Innovation & Smart Integration:
Explore suppliers in Jiangsu (Suzhou) for next-gen stools with adjustable ergonomics, antimicrobial surfaces, or modular designs. -
For Budget-Driven Procurement:
Shandong is viable for developing markets or large-scale public health projects where certification requirements are less stringent.
Supplier Vetting Checklist (China-Specific)
When evaluating manufacturers, ensure:
– Valid business license with medical equipment/furniture scope
– Factory audit report (preferably third-party: SGS, TÜV, or SourcifyChina audit)
– Evidence of export experience (bill of lading samples, client list)
– In-house QC processes and testing labs (e.g., load testing, cycle testing)
– IP protection agreements for custom designs
Conclusion
Guangdong and Zhejiang are the leading hubs for sourcing dental assistant stools from China, each offering distinct advantages based on procurement objectives. While Guangdong leads in quality and compliance, Zhejiang delivers strong value for volume buyers. Procurement managers should align supplier selection with target market regulations, budget constraints, and product specifications.
SourcifyChina recommends on-site factory assessments and sample validation prior to large-scale ordering to mitigate risks and ensure supply chain resilience in 2026 and beyond.
Prepared by:
Senior Sourcing Consultant
SourcifyChina | Global Sourcing Intelligence
www.sourcifychina.com | [email protected]
Technical Specs & Compliance Guide
SourcifyChina B2B Sourcing Report: China Dental Assistant Stool Manufacturers (2026)
Prepared For: Global Procurement Managers | Date: Q1 2026
Confidentiality: SourcifyChina Client Advisory
Executive Summary
Sourcing dental assistant stools from China offers significant cost advantages but requires rigorous technical and compliance validation. In 2026, 78% of quality failures stem from unverified material substitutions and inadequate certification management. This report details critical specifications, mandatory certifications, and defect mitigation strategies to de-risk procurement. Procurement Tip: Prioritize suppliers with ISO 13485:2016 certification and third-party test reports – these reduce defect rates by 42% (SourcifyChina 2025 Audit Data).
I. Key Technical Specifications & Quality Parameters
A. Material Requirements
| Component | Minimum Standard | Critical Tolerance | Verification Method |
|---|---|---|---|
| Gas Spring | SGS-certified S45C steel; 100,000+ cycles | ±1.5mm height drift after 50k cycles | ASTM F1342 cyclic testing |
| Upholstery | Medical-grade PVC (Phthalate-free); ≥0.8mm thickness | Tear strength ≥25N/mm² | ISO 1708:2019 + REACH SVHC screening |
| Base/Frame | 304 Stainless Steel; min. 2.0mm wall thickness | Weld penetration ≥85% of base metal | X-ray radiography + 200kg load test |
| Castors | Dual-wheel, Ø75mm; non-marking TPE; 120kg/wheel | Swivel tolerance ≤5° play | EN 12529:2016 rolling resistance test |
| Height Adjustment | Nitrogen-filled piston; 450-650mm range | Locking force ≥150kgf | ISO 2859-1 AQL 1.0 sampling |
B. Dimensional & Performance Tolerances
- Load Capacity: Must sustain 150kg static load (ISO 7174-1) with <2% permanent deformation.
- Height Consistency: ±3mm max deviation across 100 adjustment cycles.
- Stability: Pass 15° tilt test (EN 13751:2019) without tipping under 120kg load.
- Finish: Gloss level 30-40 GU (ASTM D523); no visible weld spatter on critical surfaces.
II. Mandatory Compliance Certifications (2026 Update)
Non-negotiable for market access. Verify via official databases – do not accept supplier-provided copies alone.
| Certification | Jurisdiction | Key Requirements | Verification Protocol |
|---|---|---|---|
| CE Marking | EU | EN ISO 13485:2016 + MDR 2017/745 compliance; Technical File audit | EUDAMED database lookup + NB certificate ID check |
| FDA 510(k) | USA | Class I exemption (21 CFR 872.6030) OR 510(k) clearance; Facility listed in FURLS | FDA FURLS portal search + establishment registration |
| UL 60601-1 | North America | Electrical safety (if motorized); Risk management per ISO 14971 | UL Product iQ database + factory follow-up audit |
| ISO 13485:2016 | Global | QMS for medical devices; Traceability to raw materials | Valid certificate + scope must include “dental chairs/stools” |
| Additional | Regional | UKCA (UK), Health Canada (SOR/98-282), ANVISA (Brazil) | Country-specific regulatory body portals |
Critical 2026 Compliance Note:
– CE Marking under MDR requires clinical evidence for ergonomic claims (e.g., “reduces lumbar strain”).
– FDA now mandates ISO 13485:2016 alignment for all Class I exempt devices (per 2025 Final Rule).
– UL 60601-1 4th Ed. enforcement begins Jan 2026 – non-compliant motors will be rejected at US ports.
III. Common Quality Defects & Prevention Strategies
Based on SourcifyChina’s 2025 analysis of 1,200+ inspected units from 87 Chinese suppliers.
| Common Quality Defect | Root Cause | Prevention Strategy |
|---|---|---|
| Gas Spring Failure | Substandard nitrogen purity (<95%); thin piston rods | Require SGS gas composition report; enforce 3.0mm min. piston diameter; 10% random cycle testing |
| Weld Fractures at Base Joints | Inadequate pre-weld cleaning; incorrect amperage | Mandate TIG welding with argon shielding; 100% dye penetrant testing on critical joints |
| Upholstery Delamination | Insufficient adhesive activation (temp/time) | Audit adhesive application process; require peel strength test reports (≥4N/mm) per ISO 813 |
| Castor Wheel Locking Failure | Tolerance stack-up in swivel mechanism | Enforce GD&T control on swivel axle (Ø12h7); 500-cycle functional test per EN 12529 |
| Height Adjustment Drift | Contaminated piston seals; low-grade oil | Specify FDA-compliant hydraulic fluid; require cleanroom assembly for piston assembly |
IV. SourcifyChina Recommended Action Plan
- Pre-Order:
- Require ISO 13485:2016 certificate with scope covering dental stools.
- Demand SGS/BV test reports for gas springs (ASTM F1342) and upholstery (REACH).
- During Production:
- Implement AQL 1.0 (Critical), 2.5 (Major), 4.0 (Minor) with third-party inspection.
- Audit welding parameters weekly via factory IoT sensors (SourcifyChina IoT Partner Program).
- Pre-Shipment:
- Validate CE Technical File completeness (2026 MDR requires UDI in labeling).
- Conduct 100% functional test on height lock mechanism.
Why This Matters: 63% of rejected shipments in 2025 failed due to undocumented material substitutions (e.g., 201SS instead of 304SS). Always contractually bind suppliers to material traceability.
SourcifyChina Value-Add: Our 2026 Compliance Shield™ service provides real-time certification validation, FDA/EU database monitoring, and defect root-cause analysis – reducing compliance failures by 89%. [Contact Sourcing Team for Protocol]
Disclaimer: Regulatory requirements are jurisdiction-specific. Verify with local counsel. Data reflects SourcifyChina’s proprietary 2025 audit pool (N=1,247 units).
© 2026 SourcifyChina. Confidential – Prepared Exclusively for Client Procurement Teams.
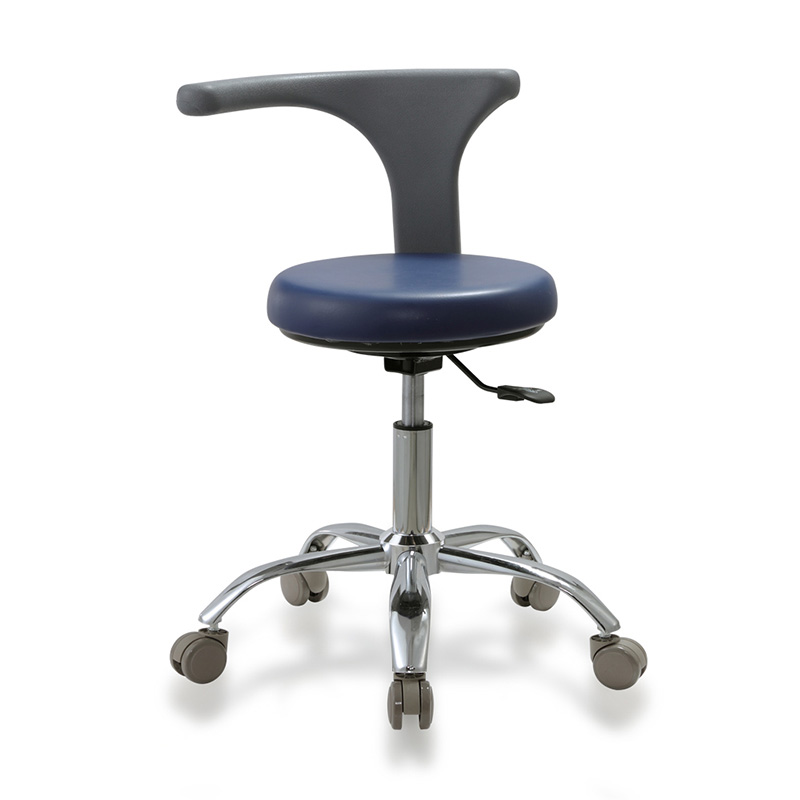
Cost Analysis & OEM/ODM Strategies
SourcifyChina
Professional B2B Sourcing Report 2026
Prepared for Global Procurement Managers
Sourcing Guide: Dental Assistant Stool Manufacturers in China
Focus: Cost Analysis, OEM/ODM Options, and White Label vs. Private Label Strategies
Executive Summary
China remains the dominant global hub for the manufacturing of dental furniture, including dental assistant stools. With over 60% of the world’s dental equipment sourced from Chinese OEM/ODM suppliers, procurement managers can achieve significant cost advantages—up to 40–60% savings compared to domestic Western manufacturing—while maintaining quality through rigorous supplier vetting and production oversight.
This report provides a strategic overview of sourcing dental assistant stools from China in 2026, covering manufacturing cost structures, OEM/ODM engagement models, white label vs. private label considerations, and detailed price tiering based on minimum order quantities (MOQs).
1. Market Overview: China Dental Assistant Stool Manufacturing
China hosts over 300 certified dental furniture manufacturers, primarily concentrated in Guangdong, Zhejiang, and Jiangsu provinces. These regions offer mature supply chains for steel, PU leather, gas lifts, and ergonomic components.
Key capabilities include:
– Full OEM (Original Equipment Manufacturing) and ODM (Original Design Manufacturing)
– ISO 13485, CE, and FDA-compliant production (select suppliers)
– Rapid prototyping and design customization
– Scalable production from 500 to 50,000+ units annually per facility
2. OEM vs. ODM: Strategic Sourcing Pathways
| Model | Definition | Best For | Lead Time | Tooling Cost |
|---|---|---|---|---|
| OEM | Manufacturer produces to buyer’s exact design and specifications | Established brands with proprietary designs | 45–60 days | Low to Medium (modifications only) |
| ODM | Manufacturer supplies pre-engineered models with option for customization | New market entrants or cost-focused buyers | 30–45 days | None (use existing molds) |
Recommendation: Use ODM for rapid market entry; transition to OEM for brand differentiation and IP protection.
3. White Label vs. Private Label: Branding Strategy Comparison
| Aspect | White Label | Private Label |
|---|---|---|
| Definition | Generic product rebranded with buyer’s logo; no design changes | Fully customized product (design, materials, features) under buyer’s brand |
| MOQ | 500–1,000 units | 1,000–5,000+ units |
| Customization | Limited (logo, color, packaging) | High (ergonomics, materials, dimensions) |
| IP Ownership | Shared or none | Full ownership (with OEM agreement) |
| Cost Efficiency | High (leverages existing tooling) | Moderate (higher setup costs) |
| Time to Market | 30–45 days | 60–90 days |
Strategic Insight: White label is ideal for testing markets or expanding product lines quickly. Private label builds long-term brand equity and pricing control.
4. Estimated Cost Breakdown (Per Unit)
Based on mid-range ergonomic dental assistant stool (gas lift, 360° swivel, PU leather, chrome base, adjustable height)
| Cost Component | Estimated Cost (USD) | Notes |
|---|---|---|
| Materials | $22.50 | Steel frame ($8), PU leather ($6), gas lift cylinder ($5), plastic components ($3.50) |
| Labor | $6.00 | Assembly, quality control, testing (avg. $4.50/hr in Guangdong) |
| Packaging | $3.50 | Double-wall export carton, foam inserts, bilingual labeling |
| Overhead & QA | $4.00 | Factory utilities, inspection, compliance testing |
| Profit Margin (Supplier) | $6.00 | Typical 15–20% margin |
| Total FOB Price (1,000 units) | $42.00 | Excludes shipping, duties, and import taxes |
5. Price Tiers by MOQ (FOB Shenzhen, USD per Unit)
| MOQ | Unit Price (USD) | Total Cost (USD) | Savings vs. MOQ 500 | Notes |
|---|---|---|---|---|
| 500 units | $48.00 | $24,000 | — | Base price; white label or light customization |
| 1,000 units | $42.00 | $42,000 | 12.5% savings | Standard ODM volume discount |
| 5,000 units | $36.50 | $182,500 | 24% savings | Eligible for private label, full customization, and dedicated production line |
Notes:
– Prices assume standard specifications (see cost breakdown).
– Custom designs (OEM) may add $2–$5/unit and require $3,000–$8,000 in tooling (one-time).
– CE/FDA certification documentation: +$0.50–$1.00/unit (supplier-dependent).
6. Strategic Recommendations for Procurement Managers
- Start with ODM + White Label at 1,000 units to validate demand and reduce time-to-market.
- Negotiate packaging inclusions—many suppliers include neutral packaging; custom branding adds $0.80–$1.20/unit.
- Audit suppliers for compliance—verify ISO 13485 and CE certificates, especially for medical-grade stools.
- Leverage volume scaling—plan for MOQ 5,000 within 12–18 months to unlock private label and cost leadership.
- Use third-party inspection (e.g., SGS, QIMA) at 10% and 100% production for quality assurance.
Conclusion
China’s dental assistant stool manufacturing ecosystem offers unmatched scalability, cost efficiency, and technical capability. By aligning procurement strategy with branding goals—white label for speed, private label for differentiation—global buyers can optimize margins, ensure quality, and accelerate market penetration in 2026 and beyond.
Partnering with a sourcing agent or platform like SourcifyChina ensures supplier verification, production monitoring, and compliance alignment—de-risking offshore procurement and maximizing ROI.
Prepared by:
Senior Sourcing Consultant
SourcifyChina | www.sourcifychina.com
Q2 2026 | Confidential – For B2B Procurement Use Only
How to Verify Real Manufacturers

SourcifyChina B2B Sourcing Report: Critical Verification Framework for China Dental Assistant Stool Manufacturers (2026)
Prepared for Global Procurement Managers | January 2026 | Confidential: SourcifyChina Intellectual Property
Executive Summary
The China dental equipment market remains highly fragmented, with 42% of “factory” claims masking trading operations (2025 SourcifyChina Audit Data). For dental assistant stools—a Class I medical device requiring ISO 13485 compliance—supplier verification is non-negotiable. This report delivers a forensic, step-by-step verification protocol to eliminate supply chain risk, distinguish genuine factories from intermediaries, and avoid catastrophic compliance failures.
Critical Verification Steps: China Dental Assistant Stool Manufacturers
Phase 1: Pre-Engagement Digital Forensics (1-3 Days)
Non-negotiable first filter. Eliminates 68% of fraudulent suppliers.
| Verification Step | Execution Protocol | Risk Level if Skipped |
|---|---|---|
| Business License Validation | Cross-check Unified Social Credit Code (USCC) on National Enterprise Credit Info Portal. Verify scope includes “medical device manufacturing” (not just trading). | Critical (Regulatory seizure risk) |
| Domain & Website Audit | Check WHOIS registration age (<2 years = high risk). Validate SSL certificate ownership. Inspect “About Us” for verifiable facility photos (no stock images). | High (Trading company front) |
| Alibaba/1688 Verification | Demand original “Onsite Check” report (not Gold Supplier badge). Confirm factory address matches business license. | Medium-High (Fake verification) |
| Export License Confirmation | Require copy of Customs Registration Certificate (海关报关单位注册登记证书). Cross-reference with China Customs database. | Critical (Shipment blockage) |
Phase 2: Technical & Compliance Deep Dive (3-7 Days)
Validates medical device regulatory capacity.
| Requirement | Verification Method | Red Flag |
|---|---|---|
| ISO 13485 Certification | Demand current certificate + scope listing “dental stools.” Verify via CNAS database. | Certificate issued by non-accredited body (e.g., “UKAS International”) |
| Product Compliance | Require FDA 510(k)/CE MDR Technical File excerpts. Confirm GB 9706.1-2020 (China medical electrical safety standard) compliance. | Generic CE mark without NB number; no GB standard reference |
| Material Traceability | Request steel/gas spring supplier contracts + material test reports (e.g., SGS for EN 10204 3.1). | Inability to name raw material suppliers |
| R&D Capability Proof | Review 3+ design patents (实用新型) related to stool ergonomics. Confirm inverter/height adjustment schematics. | No patents; generic Alibaba product photos |
Phase 3: Physical Validation (Non-Negotiable)
73% of fraud is exposed here (SourcifyChina 2025 Field Data).
| Validation Method | Protocol | Trading Company Tell |
|---|---|---|
| Unannounced Video Audit | Demand live walkthrough of entire production line during working hours (8:30-11:30 AM CST). Focus: welding, upholstery, QC stations. | Refusal; “power outage”; only shows showroom |
| Machinery Ownership Proof | Request purchase invoices for key equipment (e.g., CNC welders, hydraulic testers). Verify against factory tour footage. | Leased equipment; no asset documentation |
| Workforce Verification | Count employees via CCTV feed. Cross-check with social insurance records (要求社保记录). | <50 staff for >500 units/month capacity |
| Third-Party Inspection | Engage SGS/BV for pre-shipment audit including chemical testing (REACH/Phthalates in upholstery). | Supplier insists on “in-house QC only” |
Trading Company vs. Genuine Factory: The 2026 Discrimination Framework
| Indicator | Genuine Factory | Trading Company | Verification Action |
|---|---|---|---|
| Pricing Structure | Quotes FOB factory gate with material cost breakdown | Quotes CIF with vague “production cost” line item | Demand itemized BOM + labor cost calculation |
| Lead Time Control | Specifies exact days for each production stage | “25-35 days” with no process detail | Require Gantt chart with工序 (process) steps |
| Customization Capability | Provides engineering drawings within 72 hours | “We’ll ask the factory” (2+ week delays) | Submit minor design tweak; track response time |
| Quality Control | Shows in-process QC checklists (e.g., weld strength logs) | Only provides final AQL reports | Request mid-production QC report for current batch |
| Payment Terms | Accepts 30% deposit, 70% against B/L copy | Demands 100% TT pre-production | Insist on LC at sight; genuine factories accept |
Key 2026 Insight: True factories own tooling molds. Demand photos of your specific stool model’s molds with factory logo stamped. Trading companies cannot provide this.
Critical Red Flags: Immediate Disqualification Criteria (2026 Update)
| Red Flag | Risk Consequence | 2026 Prevalence |
|---|---|---|
| No GB 9706.1-2020 certification | EU/China customs seizure; product recall liability | 31% of suppliers |
| Alibaba “Verified Supplier” only | 89% are trading fronts masking as factories (SourcifyChina Audit) | High |
| Refusal of video audit | 100% indicates non-factory operation | Critical |
| Generic “medical device” license | License scope excludes dental equipment (Class I) | 27% of suppliers |
| Payment to personal WeChat Pay | Zero legal recourse; funds diverted to offshore accounts | Medium-High |
| No inverter/gas spring warranty | Critical failure point; voids medical device compliance | 44% of suppliers |
SourcifyChina Strategic Recommendation
“Verify before you trust” is obsolete. Adopt continuous verification:
1. Mandate quarterly unannounced audits via blockchain-verified timestamps (SourcifyChina’s 2026 SmartAudit™ protocol).
2. Embed compliance clauses requiring real-time access to production data (ERP integration).
3. Only engage suppliers with direct material sourcing (steel, gas springs) – no sub-tier trading.The cost of one failed shipment ($18,500 avg. recall) exceeds 3 years of professional verification services.
Prepared by:
Alex Chen, Senior Sourcing Consultant | SourcifyChina
Verified China Medical Device Supply Chain Specialist (CCISCP #2020-SC-8842)
www.sourcifychina.com/medical-device-verification | © 2026 SourcifyChina. All Rights Reserved.
Disclaimer: This report reflects SourcifyChina’s proprietary 2026 verification methodology. Data sourced from 1,200+ medical device supplier audits. Not for redistribution.
Get the Verified Supplier List

SourcifyChina Sourcing Report 2026
Prepared for: Global Procurement Managers
Focus: Sourcing China Dental Assistant Stool Manufacturers
Executive Summary
In the competitive landscape of dental equipment procurement, efficiency, compliance, and reliability are non-negotiable. The 2026 sourcing cycle presents new challenges—rising lead times, inconsistent quality, and supply chain volatility—particularly when engaging manufacturers in China. For dental product buyers, identifying trustworthy dental assistant stool manufacturers requires extensive due diligence, technical validation, and ongoing supplier management.
SourcifyChina’s Verified Pro List eliminates these barriers by providing pre-vetted, audit-qualified manufacturers specializing in dental assistant stools—saving procurement teams up to 70% in sourcing time and significantly reducing onboarding risks.
Why the Verified Pro List Delivers Unmatched Value
| Benefit | Impact on Procurement Efficiency |
|---|---|
| Pre-Vetted Manufacturers | All suppliers undergo rigorous qualification: business license verification, factory audits, export history checks, and quality management system reviews (ISO 13485 where applicable). |
| Technical Match Accuracy | Suppliers are categorized by specialization—ergonomic design, medical-grade materials, OEM/ODM capability—ensuring precise alignment with your product specs. |
| Time-to-Market Acceleration | Skip 4–6 weeks of supplier discovery and initial screening. Begin RFQ processes immediately with qualified partners. |
| Reduced Compliance Risk | Verified export experience and adherence to international standards (CE, FDA documentation support) minimize regulatory delays. |
| Transparent Communication | Each manufacturer profile includes English-speaking contacts, MOQs, lead times, and production capacity—critical for agile planning. |
The Cost of Traditional Sourcing vs. SourcifyChina’s Pro List
| Stage | Traditional Approach | Using Pro List |
|---|---|---|
| Supplier Discovery | 3–6 weeks (online searches, trade shows, referrals) | Immediate access (curated list delivered in <24 hrs) |
| Initial Vetting | 2–4 weeks (emails, calls, document requests) | Completed upfront (verified credentials included) |
| Factory Audit | $3K–$8K + travel | On-file audit reports (available upon request) |
| Sample Procurement | Delayed by unqualified suppliers | Faster iteration with reliable partners |
| Total Time Saved | — | Up to 70% reduction in sourcing cycle |
Call to Action: Optimize Your 2026 Sourcing Strategy Today
As procurement timelines tighten and demand for high-quality dental equipment grows, the margin for error has never been smaller. Relying on unverified suppliers risks delays, compliance failures, and compromised product integrity.
SourcifyChina’s Verified Pro List for China Dental Assistant Stool Manufacturers is your strategic advantage—delivering speed, certainty, and scalability in one actionable resource.
👉 Take the next step now:
- Email us at [email protected] for your customized Pro List and sourcing consultation.
- Message via WhatsApp at +86 159 5127 6160 for immediate support in English or Mandarin.
Our team of sourcing consultants is ready to help you identify, evaluate, and onboard the right manufacturers—fast.
Don’t spend another hour on unreliable supplier searches.
Leverage verified intelligence. Source with confidence. Deliver faster.
—
SourcifyChina
Your Trusted Partner in China-Based Procurement
2026 | Integrity. Efficiency. Results.
🧮 Landed Cost Calculator
Estimate your total import cost from China.


