The global cervical spine traction devices market is experiencing steady growth, driven by the rising prevalence of neck pain, degenerative spinal conditions, and an aging population. According to Mordor Intelligence, the market was valued at approximately USD 285 million in 2023 and is projected to grow at a CAGR of over 5.8% during the forecast period from 2024 to 2029. This expansion is further supported by increasing adoption of non-invasive treatment modalities and growing awareness about at-home physical therapy solutions. As demand for effective, user-friendly cervical traction devices rises, manufacturers are investing in ergonomic designs, technological integration, and clinical validation to differentiate their offerings. In this evolving landscape, identifying leading players with a proven track record in innovation, product quality, and global distribution is critical for healthcare providers, distributors, and end users alike. Here, we spotlight the top 7 cervical spine traction machine manufacturers shaping the industry’s future through data-backed performance and strategic advancements.
Top 7 Cervical Spine Traction Machine Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
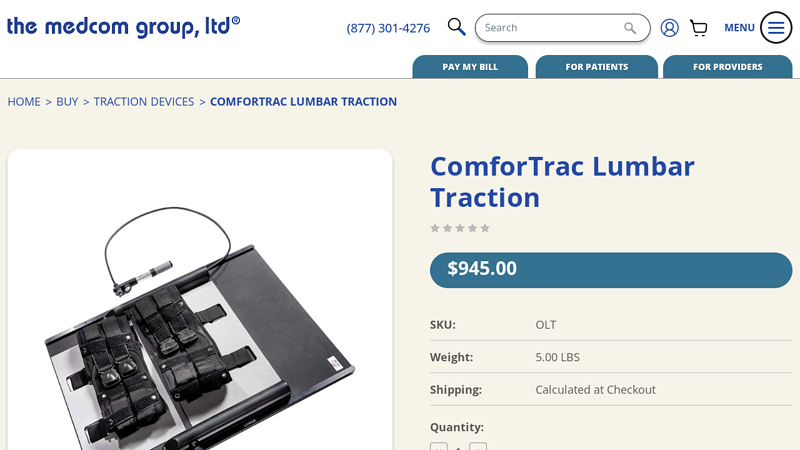
#1 ComforTrac Lumbar Traction
Domain Est. 1996
Website: medcomgroup.com
Key Highlights: This Lumbar Traction Device is designed to help separate the spaces between your vertebrae to help in relieving lower back and sciatica pain….
#2 ComforTrac Cervical Traction
Domain Est. 2000
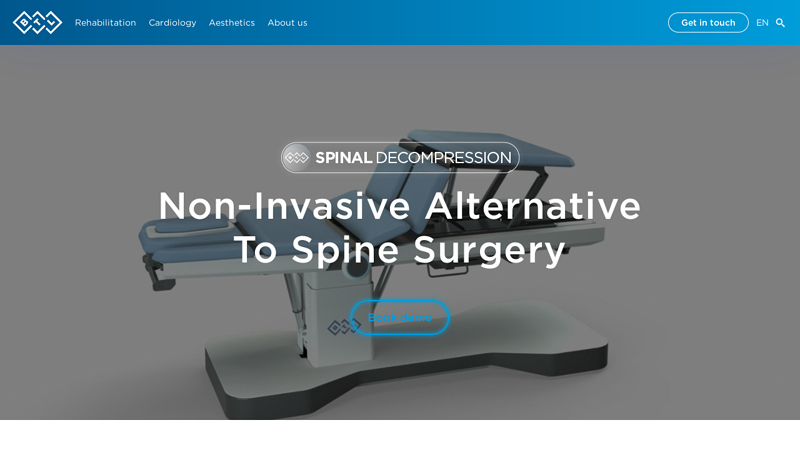
#3 BTL Spinal Decompression Machine
Domain Est. 2000
Website: btlnet.com
Key Highlights: Professional spinal decompression equipment for clinics and medical practices. Automated back decompression machine for cervical, thoracic and lumbar segments….
#4 Saunders Cervical Traction Device with Case
Domain Est. 2012
Website: sourceortho.com
Key Highlights: In stock Rating 4.8 (155) This compact and lightweight neck traction device effectively stretches and realigns the neck and spine with the assistance of a pump to achieve the per…
#5 Saunders Cervical Traction Device
Domain Est. 2017
Website: kdimfg.com
Key Highlights: A safe, use-at-home traction device for treating neck injuries. In orthopedics, traction is the practice of relieving pressure on the skeletal system….
#6 Saunders® Cervical Traction Device
Domain Est. 2019
Website: chattanoogarehab.com
Key Highlights: The Saunders Cervical Traction Device is designed to provide traction (stretching) to the cervical region (neck). The device requires no assembly….
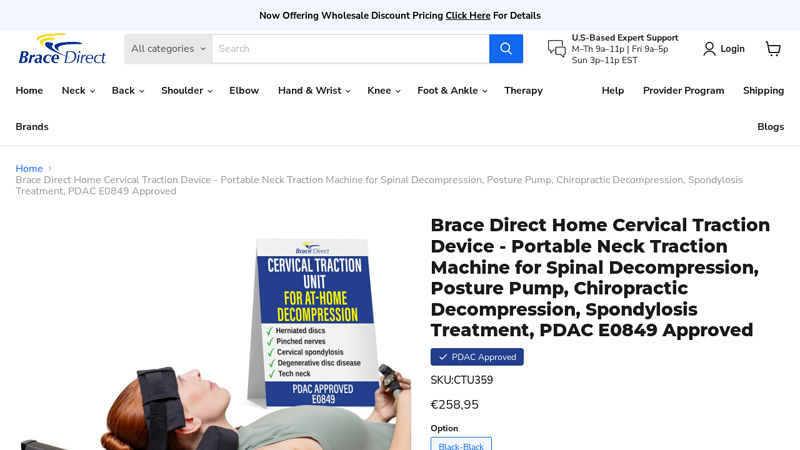
#7 Brace Direct Cervical Traction Device
Domain Est. 2019
Expert Sourcing Insights for Cervical Spine Traction Machine
H2: 2026 Market Trends for Cervical Spine Traction Machines
The global market for cervical spine traction machines is projected to experience steady growth through 2026, driven by rising prevalence of cervical spine disorders, increasing adoption of non-invasive pain management therapies, and technological advancements in home healthcare devices. Key trends shaping the market include:
-
Growing Incidence of Cervical Spinal Disorders: With sedentary lifestyles, prolonged screen time, and an aging population, conditions such as cervical spondylosis, herniated discs, and chronic neck pain are on the rise. This surge in musculoskeletal disorders is fueling demand for non-surgical treatment options like cervical traction therapy.
-
Shift Toward Home-Based Care: There is a growing preference for at-home medical devices due to convenience, cost-effectiveness, and post-pandemic healthcare trends. Portable and user-friendly cervical traction units—such as over-the-door traction devices and inflatable neck traction collars—are gaining popularity among consumers seeking self-managed pain relief.
-
Technological Innovation and Smart Devices: Manufacturers are integrating smart technology into traction machines, including app connectivity, adjustable tension controls, and real-time feedback. These advancements enhance treatment precision, user compliance, and data tracking, appealing to tech-savvy patients and healthcare providers alike.
-
Expansion of Telehealth and Remote Monitoring: The integration of cervical traction devices with telehealth platforms enables remote monitoring by physical therapists. This trend supports personalized treatment plans and improves patient outcomes, further boosting market adoption.
-
Regulatory Approvals and Reimbursement Policies: Increasing regulatory clearances (e.g., FDA, CE marking) and improved insurance coverage for non-invasive spinal therapies are reducing barriers to access and encouraging wider use of cervical traction machines.
-
Emerging Markets Driving Growth: Regions such as Asia-Pacific and Latin America are witnessing rapid growth due to improving healthcare infrastructure, rising awareness, and expanding middle-class populations. Local manufacturing and distribution partnerships are enabling market penetration in these areas.
-
Focus on Ergonomic and Wearable Designs: Companies are investing in compact, lightweight, and ergonomic designs that improve user comfort and portability. Wearable traction devices represent a niche but growing segment with strong potential by 2026.
In conclusion, the cervical spine traction machine market is poised for significant expansion by 2026, supported by medical, technological, and socio-economic drivers. Stakeholders who prioritize innovation, user experience, and accessibility are likely to capture substantial market share in this evolving landscape.
Common Pitfalls When Sourcing Cervical Spine Traction Machines: Quality and Intellectual Property Risks
Sourcing cervical spine traction machines, especially from international or unfamiliar suppliers, presents significant risks related to product quality and intellectual property (IP) infringement. Overlooking these pitfalls can result in regulatory non-compliance, patient safety issues, legal liabilities, and reputational damage.
Quality-Related Pitfalls
Inadequate Regulatory Compliance
Many suppliers, particularly in low-cost manufacturing regions, may claim their devices meet international standards but fail to provide verifiable certification. Machines might not comply with FDA 510(k), CE marking under the Medical Device Regulation (MDR), or other regional requirements. Lack of proper documentation such as Technical Files, ISO 13485 certification, or test reports increases the risk of customs delays or market bans.
Substandard Materials and Manufacturing
To reduce costs, some manufacturers use inferior materials—such as low-grade plastics or non-medical-grade metals—that compromise durability and patient safety. Poor assembly practices can lead to mechanical failures, inconsistent traction force delivery, or discomfort during use. Without on-site audits or third-party inspections, these flaws may not be detected until after shipment.
Inaccurate or Unreliable Performance
Cervical traction devices require precise control over force, angle, and duration. Inferior models may lack calibrated tension mechanisms or fail to maintain consistent settings, leading to ineffective or potentially harmful treatments. Units without proper safety stops or overload protection pose significant clinical risks.
Poor Design for Clinical Use
Some machines are designed with aesthetics or cost in mind rather than ergonomics or therapeutic efficacy. Issues include uncomfortable headrests, non-adjustable components, or setups that don’t accommodate diverse patient anatomies. These design flaws reduce clinical utility and may deter adoption by healthcare providers.
Intellectual Property-Related Pitfalls
Risk of IP Infringement
Many low-cost traction machines are direct copies or close imitations of patented designs from established medical device companies. Sourcing such products—even unknowingly—can expose buyers to legal action for contributory infringement. This is especially prevalent with popular models featuring unique adjustment mechanisms, control systems, or frame geometries.
Lack of IP Verification
Suppliers may not disclose the original design ownership or provide misleading claims about patent status. Without conducting due diligence—such as patent landscape searches or legal opinions—buyers risk importing devices that violate existing patents, leading to product seizures, fines, or litigation.
Voided Warranties and Limited Support
IP-infringing devices often come without legitimate warranties or technical support. If a machine fails or requires servicing, the supplier may disappear or refuse assistance, leaving the buyer with unusable equipment and no recourse.
Brand and Reputational Damage
Being associated with counterfeit or copied medical devices can damage a distributor’s or clinic’s reputation. Healthcare providers value trust and quality; selling or using a device proven to violate IP rights can undermine credibility and lead to loss of business.
Mitigation Strategies
To avoid these pitfalls, conduct thorough supplier vetting, request verifiable regulatory documentation, perform design and IP audits, and consider third-party quality inspections. Partnering with legally compliant manufacturers and investing in original, certified devices ensures patient safety, regulatory adherence, and long-term business integrity.
Logistics & Compliance Guide for Cervical Spine Traction Machine
Product Classification and Regulatory Status
Cervical Spine Traction Machines are typically classified as medical devices. In the United States, the FDA categorizes most traction units as Class I or Class II devices, depending on their design and intended use. Class I devices are subject to general controls (e.g., labeling, registration, and good manufacturing practices), while Class II devices may require 510(k) premarket notification to demonstrate substantial equivalence to a predicate device. In the European Union, these devices fall under the Medical Devices Regulation (MDR) (EU) 2017/745 and generally require CE marking, often via conformity assessment by a Notified Body. Manufacturers and distributors must verify the classification in each target market to ensure compliance with applicable regulatory frameworks.
Import and Export Requirements
Importing or exporting Cervical Spine Traction Machines requires adherence to international trade regulations. Key considerations include accurate HS (Harmonized System) code classification — typically under 9019.10 (apparatus for massage or physiotherapy) or 9021.10 (orthopedic appliances), depending on device features. Exporters must comply with destination country regulations, including documentation such as a Certificate of Free Sale, Certificate to Foreign Government (CFG), or Certificate of Conformity. Additionally, shipments must include proper commercial invoices, packing lists, and, where required, import licenses or permits. Dual-use considerations or technology transfer restrictions should be evaluated, particularly when shipping to sanctioned countries.
Labeling and Packaging Compliance
Labeling must meet the requirements of the destination market. FDA-regulated devices require labels showing the device name, intended use, manufacturer details, UDI (Unique Device Identifier), and any applicable warnings. EU labeling under MDR must include the CE mark, UDI, manufacturer’s name and address, and authorized representative information. Multilingual labeling may be required for EU and other international markets. Packaging must ensure product integrity during transit and comply with environmental and hazardous material regulations (e.g., no restricted substances per RoHS). Sterile devices, if applicable, must indicate sterility status and expiry date.
Shipping and Transportation Logistics
Cervical Spine Traction Machines should be shipped using carriers experienced in handling medical equipment. Devices must be securely packaged to prevent damage, with shock and moisture protection as needed. Temperature control is typically not required unless components are sensitive. For air freight, compliance with IATA Dangerous Goods Regulations is essential if batteries (e.g., in motorized units) are included — lithium batteries must be properly classified, labeled, and packed. Ground and sea shipments should follow IMDG (for maritime) or ADR (for European road) regulations when applicable. Real-time tracking and insurance are recommended for high-value shipments.
Customs Clearance and Duties
Customs clearance requires submission of accurate documentation, including the commercial invoice, bill of lading/airway bill, packing list, and any required certificates (e.g., CE, FDA listing, certificate of origin). Duties and VAT rates vary by country and classification. Preferential tariff treatment may apply under free trade agreements if rules of origin are met. Customs brokers should be engaged to ensure compliance and expedite clearance. Importers must also comply with post-market surveillance requirements, such as maintaining records and reporting adverse events per local regulations.
Post-Market Compliance and Reporting
After distribution, manufacturers and importers must comply with post-market obligations. In the U.S., this includes adherence to FDA’s Medical Device Reporting (MDR) requirements for adverse events and device malfunctions. Under EU MDR, manufacturers must appoint a Person Responsible for Regulatory Compliance (PRRC), conduct post-market surveillance (PMS), and submit Periodic Safety Update Reports (PSURs) for higher-risk devices. Vigilance reporting timelines vary (10–15 days for serious incidents). Maintaining a Quality Management System (QMS) compliant with ISO 13485 is strongly recommended to support ongoing compliance and audits.
Environmental and Disposal Regulations
End-of-life disposal of Cervical Spine Traction Machines must comply with environmental directives such as the EU WEEE (Waste Electrical and Electronic Equipment) Directive, which mandates proper recycling and labeling of electronic components. Batteries must be handled according to local battery disposal regulations (e.g., EU Battery Directive). Manufacturers may be responsible for take-back programs in certain jurisdictions. Clear instructions for decommissioning and disposal should be included in user manuals to support environmentally responsible practices.
Conclusion on Sourcing a Cervical Spine Traction Machine
Sourcing a cervical spine traction machine requires careful consideration of clinical needs, patient safety, equipment quality, and cost-effectiveness. After evaluating various suppliers, models, and features, it is evident that selecting the right device involves balancing advanced functionality—such as adjustable traction force, ease of use, portability, and patient comfort—with compliance with medical standards and regulatory requirements.
Clinics and healthcare providers should prioritize devices backed by clinical evidence, manufacturer support, and warranties to ensure long-term reliability. Whether opting for a manual or motorized unit, the decision should align with the specific therapeutic goals and patient population being served. Additionally, supplier reputation, after-sales service, and training availability are critical factors that contribute to successful implementation.
In conclusion, a well-informed sourcing decision ensures effective treatment outcomes, enhances patient satisfaction, and supports efficient clinical operations. Investing in a high-quality cervical spine traction machine from a reputable supplier ultimately contributes to improved musculoskeletal care and reinforces the standards of professional rehabilitation practice.