The global lithium-ion battery market is experiencing unprecedented expansion, driven by rising demand for electric vehicles (EVs), renewable energy storage, and portable electronics. According to a 2023 report by Grand View Research, the market was valued at USD 53.1 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 16.4% from 2023 to 2030. Meanwhile, Mordor Intelligence forecasts a CAGR of over 14.1% during the same period, underscoring the robust momentum behind battery innovation and production scaling. At the heart of this transformation are the world’s leading cell manufacturers—companies that not only dominate production capacity but are also shaping the technological trajectory of energy storage. As competition intensifies and supply chain dynamics evolve, identifying the top players provides critical insight into the forces powering the clean energy transition.
Top 10 Cell Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 FuelCell Energy
Domain Est. 1999
Website: fuelcellenergy.com
Key Highlights: FuelCell Energy is an American clean technology and manufacturing company providing large-scale, always-on, power solutions and emissions management….
#2 Suniva
Domain Est. 2005 | Founded: 2007
Website: suniva.com
Key Highlights: Suniva is America’s oldest and largest monocrystalline solar cell manufacturer in North America. Suniva was founded in 2007….
#3 Cell Factory Systems
Domain Est. 2006
Website: thermofisher.com
Key Highlights: Nunc Cell Factory systems are a proven solution for large-scale production of cells, vaccines, and therapeutic proteins….
#4 Cell factory development and design
Domain Est. 2011
Website: vttresearch.com
Key Highlights: VTT develops optimised cell factories for sustainable bioproduction. Harness microbes and enzymes to produce proteins, chemicals, materials, and more….
#5 American Battery Factory
Domain Est. 2021
Website: americanbatteryfactory.com
Key Highlights: American Battery Factory (ABF) focuses exclusively on manufacturing and enhancing high-performance prismatic Lithium Iron Phosphate (LFP) batteries….
#6 NSF Engineering Research Center for Cell Manufacturing …
Domain Est. 2015
Website: cellmanufacturingusa.org
Key Highlights: Transforming the manufacture of cell-based therapeutics into a large-scale, lower-cost, reproducible, and high-quality engineered process….
#7 Cell Culture Company
Domain Est. 2015
Website: cellculturecompany.com
Key Highlights: Located in the U.S., Cell Culture Company specializes in cell culture, cell banking, protein production, and hollow-fiber perfusion systems….
#8 Cellares
Domain Est. 2019
Website: cellares.com
Key Highlights: The company is both developing and operating integrated technologies for cell therapy manufacturing to accelerate access to life-saving cell therapies….
#9 Ultium Cells LLC: Sustainability
Domain Est. 2020
Website: ultiumcell.com
Key Highlights: Discover how Ultium Cells is driving the future of electric mobility with advanced battery cell manufacturing, innovation, & sustainability….
#10 Amplify Cell Technologies
Website: amplifycelltech.com
Key Highlights: Amplify Cell Technologies will be the largest commercial battery cell manufacturing company for heavy and medium duty vehicles in the US….
Expert Sourcing Insights for Cell
H2: 2026 Market Trends for Cell-Based Technologies
As we approach 2026, the cell-based technology market—spanning cell therapy, cultured meat, regenerative medicine, and cell-line development—is poised for transformative growth, driven by advances in biotechnology, increasing investment, and evolving regulatory landscapes. Below is an analysis of key market trends expected to shape the cell-based sector in 2026:
-
Accelerated Growth in Cell and Gene Therapies
The global cell and gene therapy (CGT) market is projected to exceed $25 billion by 2026, fueled by FDA and EMA approvals of novel treatments for oncology (e.g., CAR-T therapies), rare genetic disorders, and autoimmune diseases. Personalized medicine platforms leveraging patient-derived cells will see broader clinical adoption, supported by improvements in viral vector manufacturing and gene editing tools like CRISPR-Cas9. -
Expansion of Cultured Meat and Cellular Agriculture
The cultivated meat sector is expected to reach $1.5–2 billion by 2026, as regulatory approvals expand beyond initial markets (e.g., U.S. and Singapore). Key trends include: - Cost reduction in growth media and bioreactor scalability.
- Strategic partnerships between biotech firms and traditional food companies.
- Consumer acceptance rising due to sustainability and animal welfare concerns.
-
Diversification beyond beef and chicken into seafood and exotic meats.
-
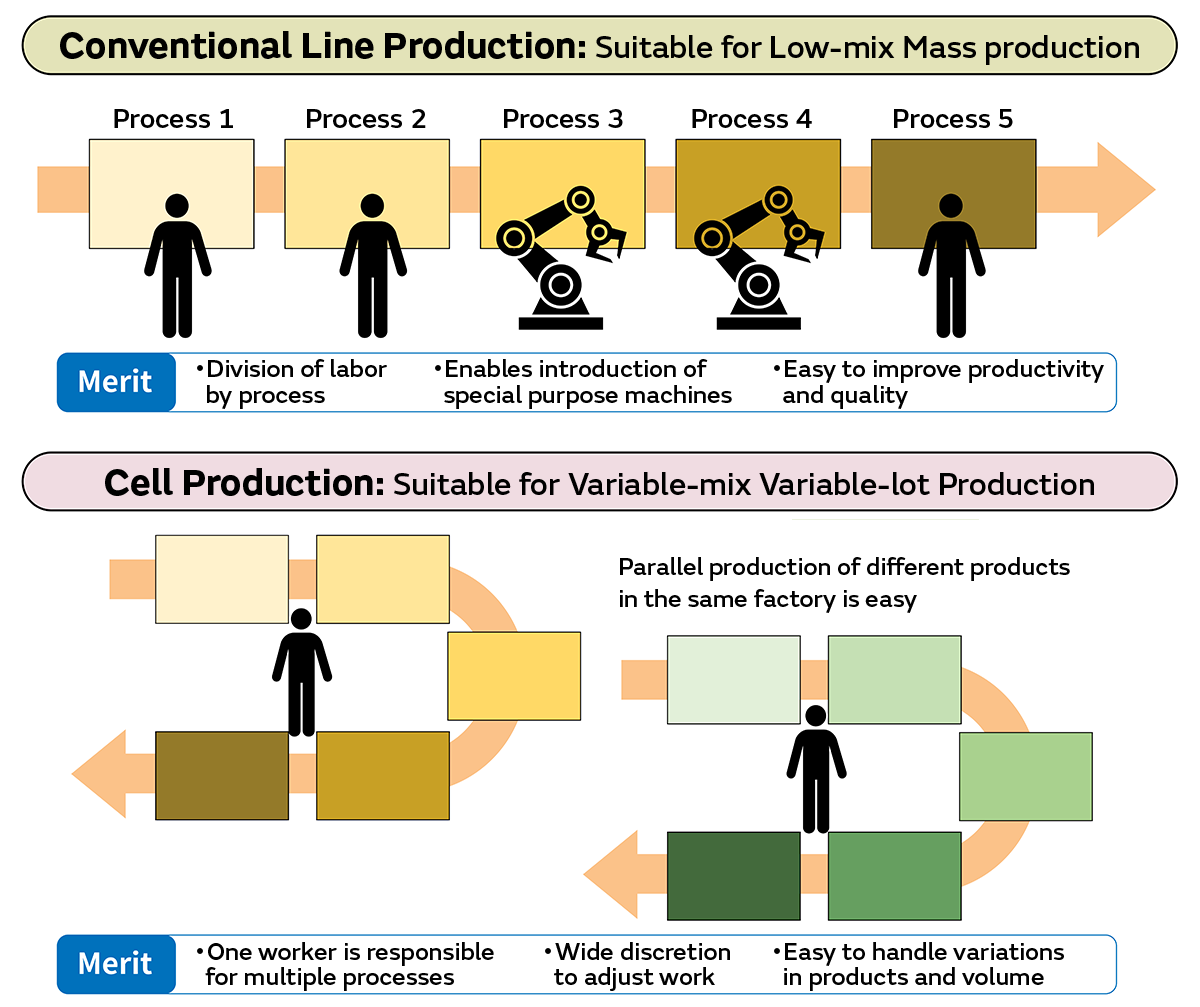
Automation and AI Integration in Cell Manufacturing
To address scalability and consistency challenges, the industry will increasingly adopt AI-driven process optimization, robotic automation, and real-time monitoring in cell culture. These technologies will enhance yield, reduce contamination risks, and lower production costs—critical for commercial viability in both therapeutics and food applications. -
Regulatory Harmonization and Standardization
By 2026, international regulatory bodies are expected to develop more cohesive frameworks for cell-based products. This includes standardized potency assays, release criteria, and guidelines for long-term safety monitoring—especially for allogeneic (“off-the-shelf”) cell therapies and novel food products. -
Rise of Allogeneic and Off-the-Shelf Therapies
While autologous therapies dominate today, allogeneic cell products are gaining momentum due to their scalability and lower costs. Advances in immune evasion technologies (e.g., gene editing to prevent rejection) will enable broader deployment of universal donor cells, particularly in immunotherapies and regenerative applications. -
Increased Investment and M&A Activity
Venture capital, pharma partnerships, and government grants will continue to fuel innovation. Expect heightened merger and acquisition (M&A) activity as large biopharma companies acquire startups with proprietary cell engineering platforms or scalable manufacturing capabilities. -
Focus on Sustainability and Ethical Sourcing
Sustainability will become a key market differentiator. In both therapeutics and food, companies will emphasize low-carbon footprints, serum-free culture systems, and ethical cell sourcing to appeal to ESG-conscious investors and consumers.
Conclusion:
By 2026, the cell-based market will be characterized by convergence across healthcare, food tech, and industrial biotechnology. Success will depend on overcoming manufacturing bottlenecks, achieving regulatory clarity, and building public trust. Companies that integrate innovation with scalable, sustainable practices are likely to lead the next wave of growth in the cell economy.

Common Pitfalls Sourcing Cells (Quality, IP)
When sourcing cells—whether for research, therapeutic development, or commercial applications—two critical risk areas are quality and intellectual property (IP). Overlooking these aspects can lead to scientific irreproducibility, regulatory setbacks, and legal liabilities. Below are common pitfalls in each domain.
Quality-Related Pitfalls
1. Lack of Authentication and Misidentification
A major issue in cell sourcing is receiving misidentified or cross-contaminated cell lines. For example, HeLa cells have contaminated numerous other lines. Without proper authentication (e.g., STR profiling), researchers risk invalid results and wasted resources.
2. Poor Viability and Contamination
Cells may arrive with low viability or be contaminated with mycoplasma, bacteria, or viruses. These issues compromise experimental outcomes and can spread contamination across lab cultures if not detected early.
3. Inconsistent Batch-to-Batch Quality
Suppliers may provide cells with variable characteristics due to differences in passage number, culture conditions, or cryopreservation methods. This variability affects reproducibility and data consistency.
4. Undefined or Inadequate Characterization
Some suppliers provide minimal data on cell phenotype, genotype, or differentiation status. Without comprehensive characterization, users cannot ensure the cells meet their experimental requirements.
5. Use of High-Passage Cells
Cells passaged excessively in culture may undergo genetic drift or senescence, altering their biological behavior. Sourcing cells without clear passage history increases the risk of using unreliable models.
Intellectual Property-Related Pitfalls
1. Unlicensed Use of Proprietary Cell Lines
Many cell lines (e.g., hybridomas, iPSCs, genetically modified lines) are protected by patents or material transfer agreements (MTAs). Using them without proper licensing can expose institutions or companies to legal action and financial penalties.
2. Ambiguous or Restrictive MTAs
Material Transfer Agreements may limit the use of cells to non-commercial research, prohibit sharing, or restrict publication. Overlooking these terms can delay collaborations or commercial development efforts.
3. Infringement of Background IP
Even if a cell line is obtained legally, using it in a specific application (e.g., a diagnostic or therapeutic) might infringe on third-party patents covering methods, genes, or uses—creating freedom-to-operate (FTO) risks.
4. Lack of Clear Ownership in Derived Products
When modifying sourced cells (e.g., creating new clones or engineered lines), ambiguity in IP ownership between the provider and recipient can lead to disputes over resulting inventions or commercial rights.
5. Sourcing from Non-Compliant or Unverified Providers
Using cells from suppliers that do not adhere to ethical sourcing standards (e.g., improper donor consent) or provide inadequate provenance documentation can jeopardize regulatory approval and public trust.
Mitigation Strategies
To avoid these pitfalls:
– Authenticate all incoming cell lines.
– Require certificates of analysis and contamination testing.
– Review MTAs and licensing terms carefully before use.
– Conduct IP landscape analyses when developing commercial products.
– Source cells from reputable, accredited repositories (e.g., ATCC, ECACC).
Proactively addressing quality and IP concerns ensures scientific rigor, regulatory compliance, and long-term project viability.

Logistics & Compliance Guide for Cells
This guide outlines key considerations for the safe, legal, and efficient transport and handling of cells (e.g., cell lines, primary cells, stem cells) in research, clinical, and commercial settings. Adherence to these guidelines ensures regulatory compliance, maintains cell viability, and protects personnel and the environment.
Regulatory Framework
Cells are subject to a variety of national and international regulations depending on their origin, type, and intended use. Key regulatory bodies include:
– FDA (U.S. Food and Drug Administration) – Regulates cells used in therapy, diagnostics, or as part of biologics under regulations such as 21 CFR Part 1271 (HCT/Ps).
– EMA (European Medicines Agency) – Oversees advanced therapy medicinal products (ATMPs), including cell-based therapies, under Regulation (EC) No 1394/2007.
– WHO (World Health Organization) – Provides global guidance on cell-based product safety and quality.
– CDC/USDA (U.S.) – Govern materials of animal or human origin that may pose infectious risks.
– IATA (International Air Transport Association) – Sets standards for the safe air transport of biological materials (IATA Dangerous Goods Regulations, Packing Instruction 650 for UN 3373).
Classification of Cells
Proper classification is essential for compliance:
– Biological Substance, Category B (UN 3373): Most diagnostic or research specimens containing viable cells, not known to cause disease in humans or animals.
– Exempt Human Specimens: Under certain conditions, human cells may be exempt from full dangerous goods regulations if there is minimal risk.
– Infectious Substances (Category A, UN 2814/2900): Cells known or expected to contain pathogens must be classified accordingly.
– Genetically Modified Organisms (GMOs): May require special permitting and documentation under environmental or biosafety regulations (e.g., NIH Guidelines, EU Directive 2001/18/EC).
Packaging & Labeling
All shipments must comply with IATA, national, and institutional requirements:
– Primary Container: Leak-proof, sealed tube or vial (e.g., cryovial) with internal closure.
– Secondary Container: Leak-proof and capable of containing all fluid if the primary container fails.
– Absorbent Material: Placed between primary and secondary containers to absorb any leaks.
– Outer Packaging: Rigid, durable box capable of withstanding normal handling.
– Labels: Include:
– “Biological Substance, Category B” (proper shipping name)
– UN 3373 diamond label
– Sender and recipient contact information
– Dry ice label (if applicable; UN 1845)
– Any required GMO or biohazard symbols
Cold Chain Management
Cells are often shipped under temperature-controlled conditions:
– Cryopreserved Cells: Shipped in vapor-phase liquid nitrogen dry shippers or dry ice (typically -80°C or below). Monitor temperature with data loggers.
– Refrigerated Cells: Transported at 2–8°C using validated cold boxes with phase-change materials.
– Ambient: Rare; only for short durations and robust cell types.
– Validation: Use qualified packaging systems and temperature monitoring devices. Maintain shipment records.
Documentation
Complete and accurate documentation is mandatory:
– Shipping Declaration: Required for dangerous goods (e.g., IATA Shipper’s Declaration for Dangerous Goods).
– Material Transfer Agreement (MTA): Governs ownership, use, and intellectual property.
– Customs Documentation: Include detailed description of contents, origin, and purpose (e.g., commercial invoice, certificate of origin).
– Ethical & Regulatory Approvals: Proof of IRB/ethics committee approval for human-derived cells, or IACUC approval for animal-derived cells.
– Permits: Import/export permits may be required (e.g., CITES for certain animal cells, USDA permits).
Import & Export Compliance
International shipments require special attention:
– Restricted Materials: Some countries restrict import of human or animal tissues.
– Dual-Use Concerns: Cells with potential biosecurity implications may require export licenses (e.g., under U.S. EAR or ITAR).
– Customs Delays: Avoid by ensuring complete, accurate documentation and proper classification.
Biosafety & Containment
- BSL Level: Handle and package cells according to appropriate biosafety level (BSL-1, BSL-2, etc.).
- Decontamination: Decontaminate packaging surfaces post-shipment if contamination is suspected.
- Training: Personnel must be trained in biosafety, shipping regulations (e.g., IATA certification), and emergency procedures.
Recordkeeping
Maintain detailed records for traceability and audits:
– Shipment logs (date, contents, sender, recipient, tracking)
– Temperature monitoring reports
– Copies of MTAs, permits, and regulatory approvals
– Training records for personnel
Emergency Procedures
- Leak or Spill: Isolate area, use PPE, decontaminate per institutional biosafety protocol.
- Exposure: Follow institutional medical response and reporting procedures.
- Regulatory Incident: Report breaches to appropriate authorities (e.g., FDA, EMA, IATA).
Conclusion
Proper logistics and compliance are critical for the responsible handling of cells. Always consult institutional biosafety officers, regulatory affairs departments, and the latest regulatory texts before shipping. Stay updated on evolving guidelines, especially for emerging cell therapies and cross-border collaborations.
Conclusion: Sourcing a Cell Factory
In conclusion, sourcing a cell factory is a strategic decision that plays a critical role in the successful development and commercialization of advanced therapies such as cell and gene treatments. The selection of the right manufacturing partner requires a comprehensive evaluation of technical capabilities, regulatory compliance, scalability, quality systems, and cost-efficiency. Whether opting for in-house development, a contract manufacturing organization (CMO), or a partnership with an established cell factory, the choice must align with the company’s long-term goals, pipeline complexity, and regulatory requirements.
A well-vetted and reliable cell factory ensures not only the consistent production of high-quality, clinically viable cellular products but also accelerates time-to-market and reduces operational risks. As the demand for personalized and regenerative medicines continues to grow, building resilient and flexible supply chains through strategic sourcing will be essential for innovation, compliance, and commercial success in the evolving biopharmaceutical landscape.