The global compression therapy devices market, driven by rising awareness of circulatory health and increasing prevalence of conditions like deep vein thrombosis (DVT) and lymphedema, is projected to grow at a CAGR of 7.2% from 2023 to 2030, according to Grand View Research. With this upward trajectory, demand for advanced calf pressure machines—critical tools in both clinical rehabilitation and athletic recovery—has surged among healthcare providers, physical therapists, and sports medicine professionals. As innovation accelerates in pneumatic compression technology, manufacturers are differentiating through enhanced pressure modulation, portability, and data-driven recovery tracking. Based on market presence, product performance, technological innovation, and clinical validation, the following eight companies represent the forefront of calf pressure machine manufacturing in today’s competitive landscape.
Top 8 Calf Pressure Machine Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
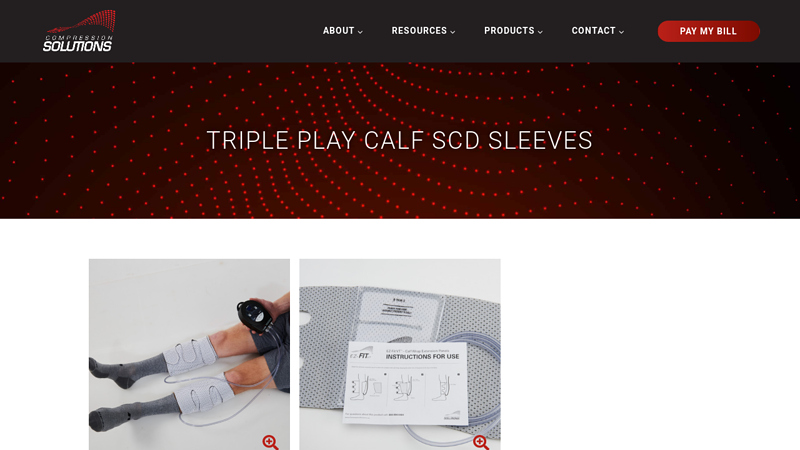
#1 Triple Play Calf SCD Sleeves
Domain Est. 2009
Website: compressionsolutions.us
Key Highlights: Add pneumatic compression for calves into post-surgical recovery in order to prevent DVT or blood clots after surgery.Missing: pressure manufacturer…
#2 VALD Performance
Domain Est. 2014
Website: valdperformance.com
Key Highlights: VALD technology is used to measure, monitor and manage physical performance in dozens of different court, field, track, individual and team sports….
#3 AIROS Medical
Domain Est. 2016
Website: airosmedical.com
Key Highlights: Our compression therapy device technology helps thousands of people battling lymphatic and venous disorders live better….
#4 SD
Domain Est. 1996
Website: truefitness.com
Key Highlights: The Leg/Calf Press machine from the FORCE strength series enhances any facility with its ultra-sleek and stylish appearance and will exude sophistication in ……
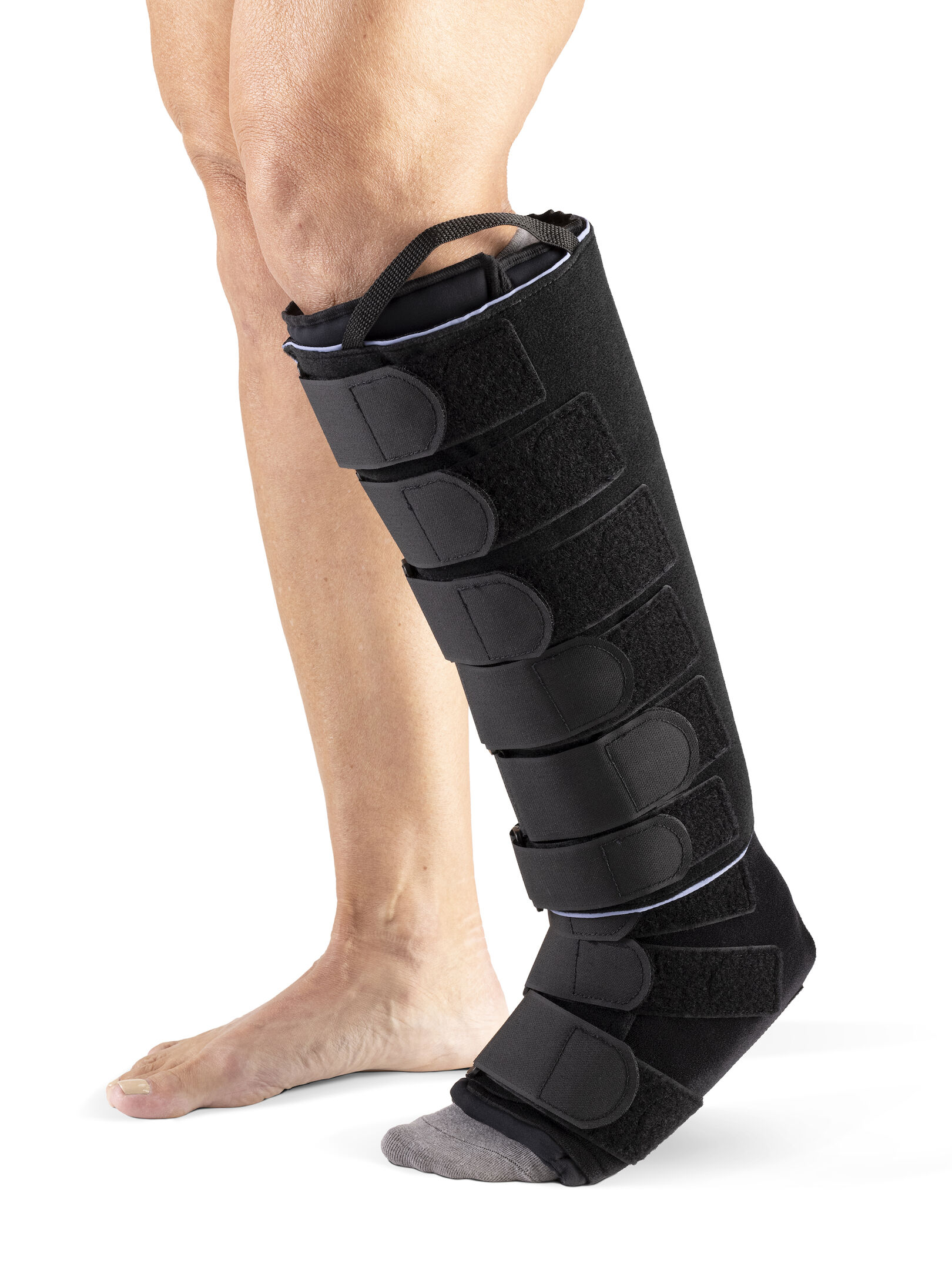
#5 Medaform Standard Calf and Foot
Domain Est. 1996
Website: sigvaris.com
Key Highlights: The Medaform Standard Calf and Foot is an inelastic compression device, paired with a non-compressive, Basic Liner….
#6 Normatec 3 Legs: Complete Leg Recovery Suite
Domain Est. 2007
Website: hyperice.com
Key Highlights: In stock Rating 5.0 (334) Experience leg recovery like never before with Normatec 3 Legs. Tailored pressures, dynamic cycles. Dive into a world of rapid relief and relaxation….
#7 Recovery Systems
Domain Est. 2016
Website: recoverysystemssport.com
Key Highlights: 9-day delivery 14-day returnsAtom is the most powerful calf air compression sleeve pump on the market, with Four pressures to match all recovery needs and a type C rechargeable bat…
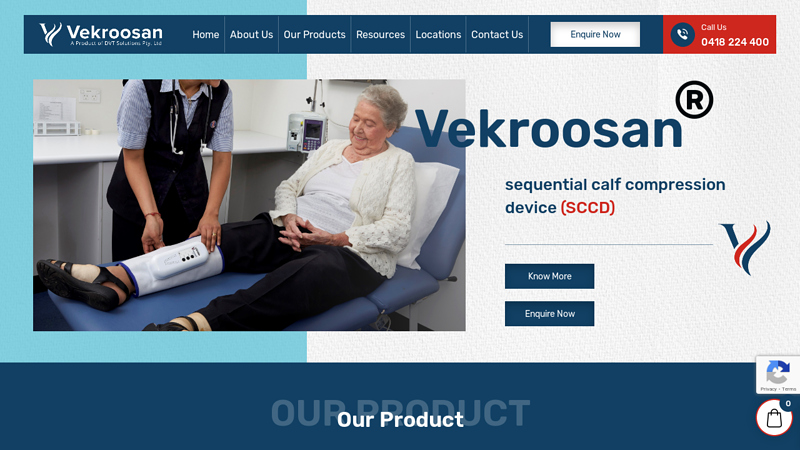
#8 Vekroosan
Website: vekroosan.com.au
Key Highlights: Vekroosan is an easy-to-use, mobile calf compression device designed to prevent and treat Deep Vein Thrombosis, Chronic Venous Disorder and related vein ……
Expert Sourcing Insights for Calf Pressure Machine

H2: 2026 Market Trends for Calf Pressure Machines
The global market for calf pressure machines—also known as compression therapy devices or pneumatic compression devices—is projected to experience significant growth and transformation by 2026, driven by rising awareness of circulatory health, increasing prevalence of chronic conditions, and advancements in wearable medical technology. Key trends shaping the market include:
-
Growing Demand in Preventive and Post-Surgical Care
By 2026, calf pressure machines are expected to play a more prominent role in preventive healthcare, particularly in reducing the risk of deep vein thrombosis (DVT) and venous insufficiency. Hospitals, outpatient clinics, and home healthcare settings are increasingly adopting these devices for post-surgical recovery, especially after orthopedic and cardiovascular procedures. The aging global population and rising obesity rates are contributing to higher DVT incidence, fueling demand. -
Expansion of Home Healthcare Segment
The shift toward decentralized care models is accelerating the adoption of portable and user-friendly calf pressure devices for home use. By 2026, manufacturers are focusing on lightweight, app-connected devices that allow patients to manage therapy remotely. This trend is supported by favorable reimbursement policies in regions like North America and Europe and growing consumer comfort with self-administered treatments. -
Technological Integration and Smart Devices
A major trend is the integration of smart technology into calf pressure machines. Devices equipped with sensors, Bluetooth connectivity, and mobile apps are gaining traction. These features enable real-time monitoring of therapy sessions, adherence tracking, and data sharing with healthcare providers. Artificial intelligence (AI) is also being explored to personalize compression therapy based on patient-specific metrics. -
Rise in Sports and Wellness Applications
Beyond clinical use, calf pressure machines are increasingly popular among athletes and fitness enthusiasts for recovery and performance enhancement. By 2026, the sports medicine and wellness sectors are expected to represent a growing share of the market. Brands are launching stylish, portable compression boots marketed for muscle recovery, reduced soreness, and improved athletic performance. -
Increased Competition and Innovation
The market is becoming more competitive, with both established medical device companies and new entrants launching innovative products. Key players are investing in R&D to improve comfort, reduce noise, and enhance portability. Additionally, disposable and reusable sleeve designs are being refined for hygiene and convenience. -
Regulatory and Reimbursement Landscape
Regulatory approvals and insurance coverage will continue to influence market access. By 2026, clearer guidelines around the use of compression therapy for various indications may expand insurance reimbursement, especially in emerging markets like Asia-Pacific and Latin America. -
Geographic Market Expansion
While North America currently dominates the market due to advanced healthcare infrastructure, the Asia-Pacific region is expected to register the highest growth rate by 2026. Increasing healthcare spending, urbanization, and rising awareness of vascular health are key drivers in countries such as China, India, and South Korea.
In conclusion, the calf pressure machine market in 2026 will be characterized by technological innovation, expanding applications beyond traditional medical use, and a strong push toward patient-centric, at-home solutions. Stakeholders who invest in smart, accessible, and evidence-based devices are likely to gain a competitive advantage in this evolving landscape.

Common Pitfalls When Sourcing Calf Pressure Machines (Quality and IP)
Sourcing calf pressure machines—commonly used in medical rehabilitation, sports therapy, or lymphedema treatment—requires careful attention to both product quality and intellectual property (IP) concerns. Overlooking these aspects can lead to operational inefficiencies, legal risks, and compromised patient safety. Below are key pitfalls to avoid:
Poor Build Quality and Inadequate Safety Standards
Many low-cost suppliers, especially in competitive international markets, may offer calf pressure machines with subpar materials, inconsistent pressure delivery, or insufficient safety certifications. Machines lacking compliance with medical device regulations (e.g., FDA, CE marking, ISO 13485) risk patient injury and expose buyers to liability. Always verify conformity with region-specific medical standards and request third-party test reports.
Lack of Calibration and Pressure Accuracy
A critical function of calf pressure devices is consistent and accurate compression. Inexpensive or poorly engineered units may deliver erratic pressure levels, undermining therapeutic effectiveness. Be cautious of suppliers who cannot provide calibration documentation, pressure range specifications, or data on cycle durability. Test units before bulk purchase to verify performance consistency.
Incomplete or Counterfeit Technical Documentation
Some suppliers may provide incomplete user manuals, missing maintenance guides, or falsified compliance documentation. This not only affects usability but also complicates regulatory approval in your target market. Ensure all documentation is comprehensive, in the correct language, and verifiable with certification bodies.
Intellectual Property Infringement Risks
Sourcing from manufacturers that copy patented designs, software interfaces, or proprietary technology can expose your business to IP litigation. Avoid suppliers offering “branded” features or design elements that closely resemble leading market products without authorization. Conduct due diligence on the supplier’s R&D capabilities and request proof of IP ownership or licensing agreements.
No Clear Warranty or After-Sales Support
Low-cost machines often come with limited or ambiguous warranty terms and minimal after-sales service. If the supplier lacks local support or spare parts availability, downtime and repair costs can outweigh initial savings. Confirm warranty duration, service response times, and availability of technical support before finalizing procurement.
Misrepresentation of Intended Use or Certification Scope
Some suppliers claim their devices are “medical-grade” without proper certification for clinical use. Be wary of ambiguous claims and ensure the device’s certification explicitly covers its intended therapeutic application (e.g., DVT prevention, lymphatic drainage). Using uncertified devices in medical settings may violate regulations and void insurance coverage.
By proactively addressing these quality and IP-related pitfalls, buyers can ensure the safe, legal, and effective deployment of calf pressure machines in their operations.

Logistics & Compliance Guide for Calf Pressure Machine
This guide outlines the essential logistics considerations and compliance requirements for the safe, efficient, and legal handling, transportation, and operation of a Calf Pressure Machine. Adherence to these guidelines ensures product integrity, regulatory compliance, and user safety.
Product Handling and Storage
Handle the Calf Pressure Machine with care to prevent damage to sensitive components such as pressure sensors, control units, and inflatable cuffs. Always transport the device in its original packaging or a protective case when possible. Store the machine in a clean, dry, temperature-controlled environment (typically 10°C to 40°C and less than 80% non-condensing humidity) away from direct sunlight, dust, and corrosive substances. Avoid stacking heavy items on top of the unit or its accessories.
Transportation Requirements
When shipping the Calf Pressure Machine domestically or internationally, use a reputable courier experienced in medical device logistics. Ensure the device is securely packed with shock-absorbing materials to withstand vibration and impact. Clearly label the package as “Fragile” and “This Side Up.” For air transport, comply with IATA regulations—lithium batteries (if applicable) must meet IATA Dangerous Goods Regulations (DGR), including proper packaging, labeling, and documentation. Provide the Harmonized System (HS) code for customs clearance (e.g., 9018.90 for other electrotherapeutic apparatus).
Regulatory Compliance
The Calf Pressure Machine must comply with relevant medical device regulations in the target market. In the United States, it is typically classified under FDA Class II (e.g., 21 CFR 890.5925 for compression devices) and requires 510(k) clearance. In the European Union, it must meet the Medical Device Regulation (MDR) 2017/745 and carry the CE mark. Ensure the device has a valid Declaration of Conformity, includes UDI (Unique Device Identification), and is manufactured under a certified Quality Management System (e.g., ISO 13485).
Labeling and Documentation
All units must feature permanent labeling including the device name, model number, serial number, manufacturer details, power specifications, and regulatory markings (e.g., CE, FDA). Include a user manual in the local language(s) of the destination market, containing instructions for use, contraindications, warnings, maintenance procedures, and troubleshooting. The manual must comply with IEC 60601-1 (general safety) and IEC 60601-2-34 (particular requirements for compression therapy devices).
Import and Export Controls
Verify export licensing requirements based on destination country and device classification. Some countries may require import permits, registration with local health authorities (e.g., ANVISA in Brazil, TGA in Australia), or conformity assessment by national bodies. Maintain accurate records of all shipping documents, invoices, certificates of origin, and regulatory approvals for audit purposes.
Post-Market Surveillance and Reporting
Implement a system for monitoring device performance and adverse events post-distribution. Report any incidents involving malfunction, injury, or death to relevant authorities (e.g., FDA MAUDE database, EUDAMED in the EU) according to local timelines. Maintain records of complaints, field safety notices, and corrective actions for a minimum of 10 years post-device withdrawal.
Environmental and Disposal Compliance
Dispose of the Calf Pressure Machine and its components in accordance with local e-waste and medical device disposal regulations. Batteries must be recycled through certified programs. Follow WEEE (Waste Electrical and Electronic Equipment) directives in the EU and equivalent regulations elsewhere to minimize environmental impact.
Conclusion for Sourcing a Calf Pressure Machine:
After thorough research and evaluation of available options, sourcing a calf pressure machine proves to be a valuable investment for enhancing circulation, reducing muscle soreness, and supporting recovery—particularly beneficial for athletes, individuals with sedentary lifestyles, or those managing circulatory conditions. Key factors considered in the sourcing process include machine efficacy, design features (such as adjustable pressure settings, portability, and ease of use), brand reputation, cost, and customer reviews. Opting for a medically certified and clinically supported device ensures safety and reliability. Ultimately, selecting a high-quality calf pressure machine from a reputable supplier will provide long-term health benefits, improve user comfort, and deliver a strong return on investment through enhanced well-being and recovery performance.