The global calcium alginate dressing market is experiencing robust growth, driven by the increasing prevalence of chronic wounds and rising demand for advanced wound care products. According to Grand View Research, the global wound care market was valued at USD 18.5 billion in 2022 and is expected to expand at a compound annual growth rate (CAGR) of 5.6% from 2023 to 2030, with alginate dressings holding a significant share due to their superior absorbency and biocompatibility. Mordor Intelligence further projects that the calcium alginate segment will witness steady growth, fueled by an aging population, higher incidence of diabetes-related ulcers, and advancements in biomaterials. As demand intensifies, manufacturers are focusing on innovation, scalability, and clinical efficacy—making it essential to identify the leading players shaping the future of alginate-based wound care solutions.
Top 7 Calcium Alginate Ag Dressing Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Algicell® Ag Calcium Alginate Dressing with Antimicrobial Silver
Domain Est. 1998
Website: byramhealthcare.com
Key Highlights: GENTELL, INC. Algicell® Ag Calcium Alginate Dressing with Antimicrobial Silver. Silver has been used as an antimicrobial since ancient times, ……
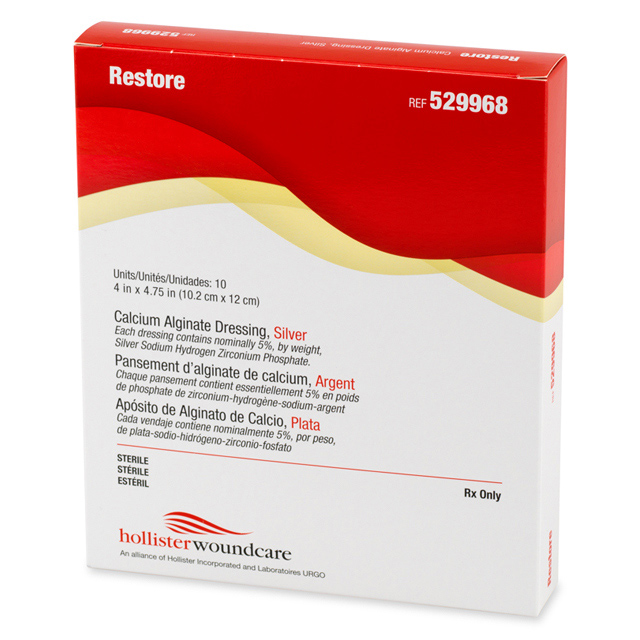
#2 Restore™ Calcium Alginate Dressing – Silver
Domain Est. 1994
Website: hollister.com
Key Highlights: A sterile, non-woven calcium alginate dressing composed of an ionic silver complex (silver sodium hydrogen zirconium phosphate), which releases silver ions….
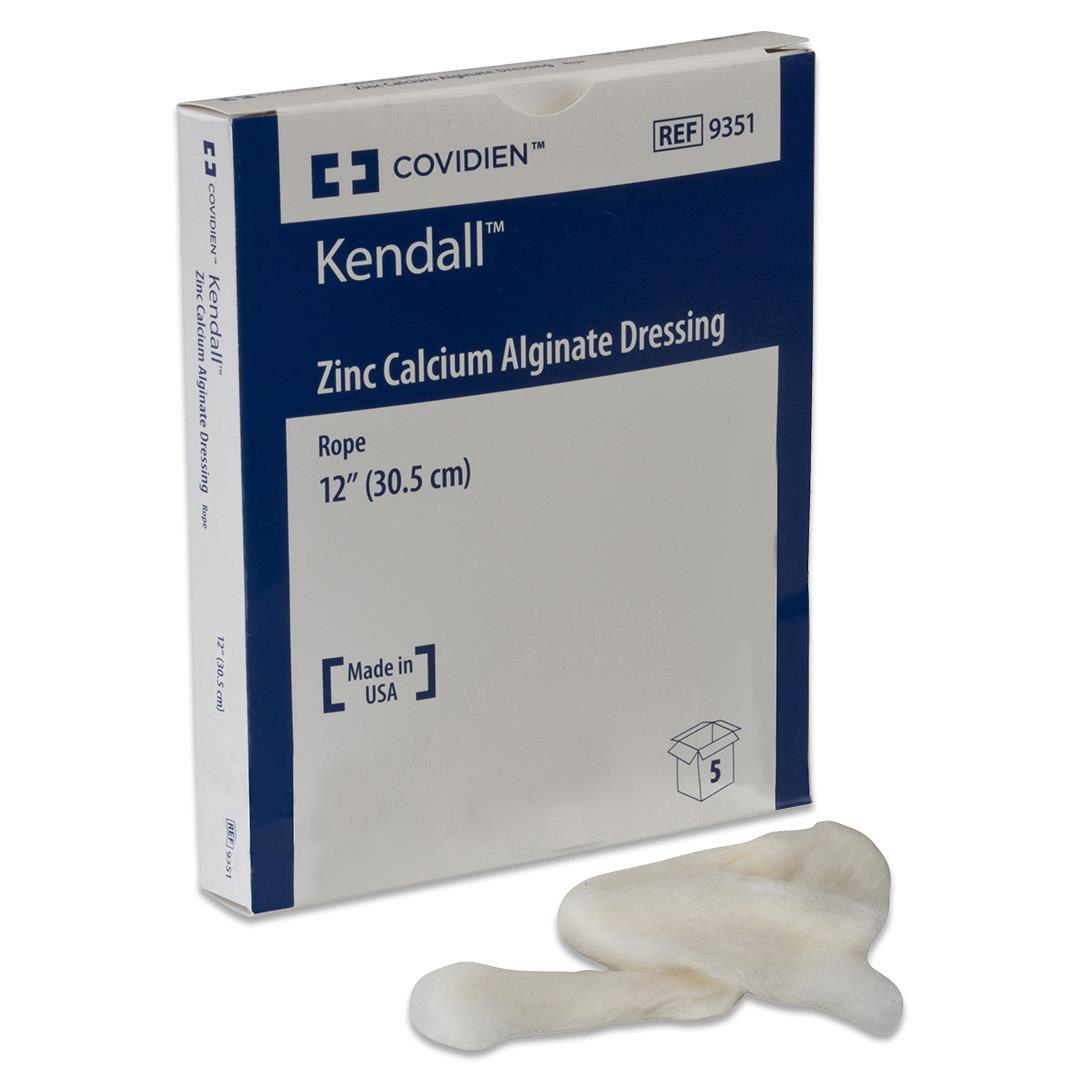
#3 Zinc Calcium Alginate Dressing
Domain Est. 1996
Website: cardinalhealth.com
Key Highlights: Alginate dressings are designed to absorb wound exudate, form a gel that keeps the wound moist and protect the wound from contamination….
#4 Calcium Alginate Dressings
Domain Est. 1998
Website: silverlon.com
Key Highlights: Calcium Alginate Dressings are an effective barrier to microbial penetration for moderate to heavy exudating partial and full thickness wounds….
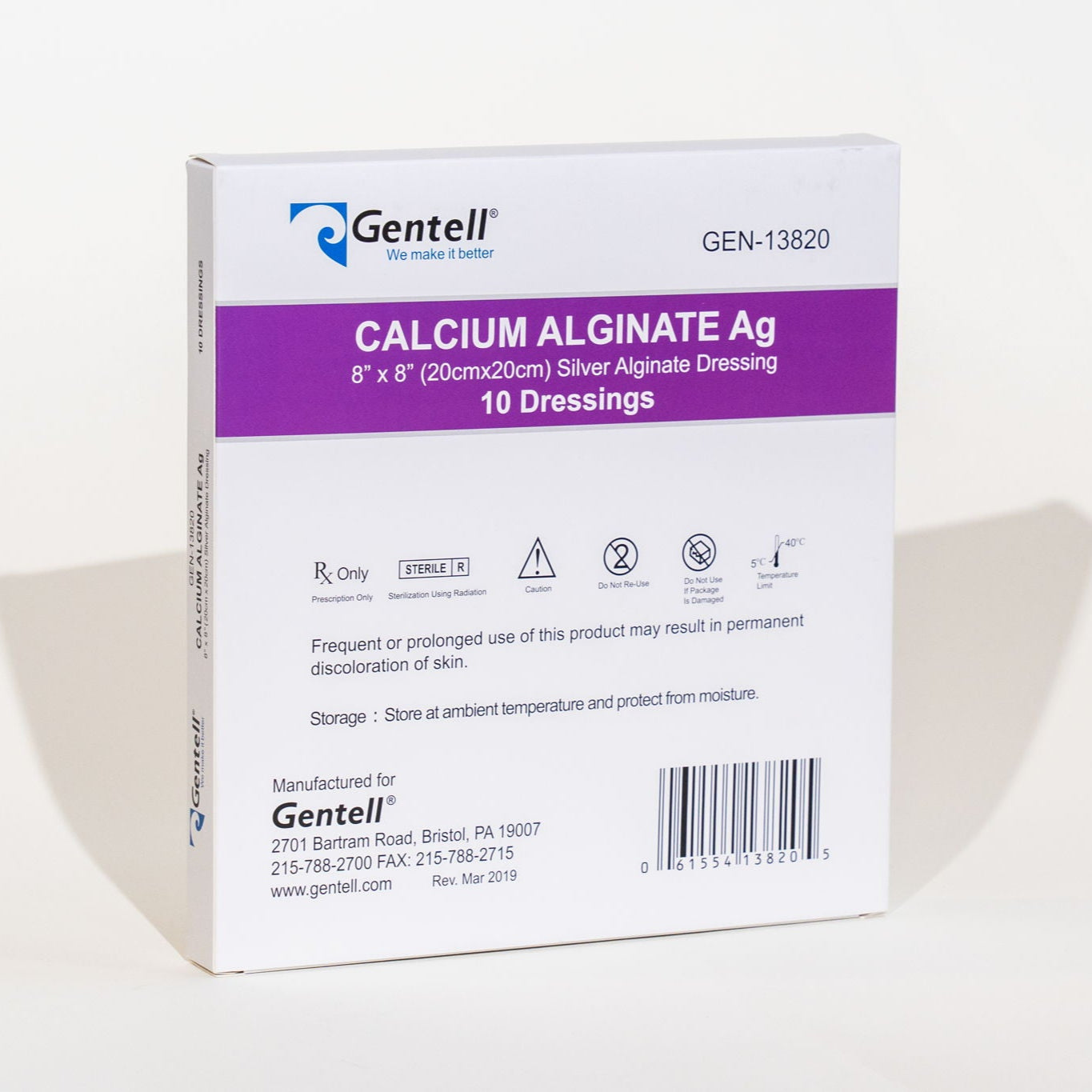
#5 Calcium Alginate Ag (Silver) Dressings
Domain Est. 2000
Website: gentell.com
Key Highlights: Gentell Calcium Alginate Ag (Silver) Dressings are a sterile, antimicrobial, comfortable, fiber-structured alginate with high absorbency….
#6 DynaGinate AG Silver Calcium Alginate Wound Dressing 4 x 5 Inches
Domain Est. 2012
#7 HALYARD* Calcium Alginate Dressing
Domain Est. 2013
Website: products.halyardhealth.com
Key Highlights: Features: · Highly absorbent and conformable · Maintains Structural Intergrity when wet · Non-woven calcium alginate · Dressing may remain in place for up to 7 days….
Expert Sourcing Insights for Calcium Alginate Ag Dressing
H2: 2026 Market Trends for Calcium Alginate AG Dressing
The global market for Calcium Alginate AG (Antimicrobial Silver) Dressings is poised for significant evolution by 2026, driven by technological advancements, rising chronic wound prevalence, and increased demand for infection-preventive wound care solutions. Below is an analysis of the key market trends expected to shape the industry in 2026:
1. Rising Prevalence of Chronic Wounds and Diabetic Ulcers
By 2026, the growing incidence of diabetes, venous insufficiency, and pressure ulcers—particularly in aging populations across North America, Europe, and parts of Asia—will continue to drive demand for advanced wound care products. Calcium Alginate AG dressings, known for their hemostatic properties, high absorbency, and sustained silver ion release, are increasingly favored in managing heavily exuding chronic wounds. This demographic shift will expand market adoption, especially in outpatient and home healthcare settings.
2. Shift Toward Antimicrobial and Infection-Control Solutions
With antibiotic resistance becoming a global health concern, healthcare providers are prioritizing infection prevention in wound management. Calcium Alginate AG dressings offer broad-spectrum antimicrobial protection due to the incorporation of silver, reducing biofilm formation and local infection risks. In 2026, regulatory emphasis on reducing hospital-acquired infections (HAIs) is expected to further boost demand for silver-based dressings in both hospital and long-term care facilities.
3. Innovation in Dressing Formulations and Delivery Systems
Manufacturers are investing in next-generation formulations that enhance biocompatibility, extend silver release kinetics, and improve patient comfort. By 2026, expect to see hybrid dressings combining calcium alginate with other biomaterials (e.g., chitosan, hydrocolloids) or advanced silver nanoparticles for improved efficacy. Additionally, user-friendly application formats—such as ribbons, sheets with adhesive borders, and pre-moistened pads—are gaining traction, particularly in home care.
4. Expansion in Emerging Markets
Asia-Pacific, Latin America, and the Middle East are anticipated to register the highest compound annual growth rates (CAGR) by 2026. Increasing healthcare expenditure, improving wound care infrastructure, and rising awareness among clinicians are accelerating market penetration in countries like India, China, Brazil, and Saudi Arabia. Local production and partnerships with global players will further reduce costs and increase accessibility.
5. Regulatory and Reimbursement Landscape
In 2026, stringent regulatory standards—particularly from the FDA and EU MDR—will continue to shape product development, with an emphasis on clinical evidence and biocompatibility. Reimbursement policies in key markets will increasingly favor cost-effective advanced dressings that reduce healing time and hospital stays, positioning Calcium Alginate AG dressings as a favorable option under value-based care models.
6. Sustainability and Biodegradability Concerns
As environmental sustainability becomes a priority, manufacturers are exploring eco-friendly production methods and biodegradable packaging. Calcium alginate, derived from brown seaweed, is inherently biodegradable—a feature that will be increasingly marketed as a sustainable advantage over synthetic alternatives.
Conclusion
By 2026, the Calcium Alginate AG Dressing market will be characterized by innovation, geographic expansion, and a strong focus on infection control and patient outcomes. With the compound effects of aging populations, chronic disease burden, and healthcare system modernization, the market is expected to grow at a CAGR of approximately 7–9% during the 2021–2026 forecast period, reaching an estimated market value of USD 700–800 million. Strategic investments in R&D, regulatory compliance, and emerging market outreach will be crucial for stakeholders aiming to capture sustained growth.

Common Pitfalls Sourcing Calcium Alginate Ag Dressing (Quality, IP)
Sourcing Calcium Alginate Silver (Ag) dressings, a critical advanced wound care product combining calcium alginate’s absorbency with silver’s antimicrobial properties, involves navigating significant quality and intellectual property (IP) risks. Avoiding these common pitfalls is essential for ensuring patient safety, product efficacy, regulatory compliance, and protecting your business.
Uncertified or Inadequate Quality Standards
Relying on suppliers without verifiable certifications is a primary risk. Calcium Alginate Ag dressings are Class IIa/III medical devices requiring strict adherence to ISO 13485 (Quality Management) and relevant ISO standards (e.g., ISO 10993 for biocompatibility, ISO 11737 for sterility). Sourcing from manufacturers lacking these certifications, or providing only self-declared conformity, significantly increases the risk of substandard products. Always demand valid, current certificates from accredited third-party bodies and verify them directly with the certifying organization. Do not accept “equivalent” standards without rigorous validation.
Inconsistent or Unverified Silver Content and Release Profile
The therapeutic efficacy hinges on consistent, controlled silver ion release. A major pitfall is accepting supplier claims about silver concentration (e.g., ppm) without independent verification through batch-specific Certificates of Analysis (CoA) from accredited labs. More critically, the release profile – how effectively silver ions are delivered over time in a wound environment – is often poorly characterized or misrepresented. Insist on access to validated test data (e.g., according to ASTM E2149 or similar) demonstrating sustained antimicrobial efficacy, not just total silver content. Inconsistent release can lead to treatment failure or potential cytotoxicity.
Questionable Raw Material Sourcing and Traceability
The quality of the final dressing is directly tied to the source and quality of the alginate and silver compound. A common pitfall is inadequate traceability. Suppliers may use alginates from unverified sources with variable purity (e.g., high endotoxin levels) or inconsistent G/M ratios, impacting gel formation and absorption. Similarly, the form of silver (e.g., silver alginate, nanocrystalline silver, silver sulfadiazine) and its source matter greatly. Demand full traceability documentation (CoO – Certificate of Origin) for key raw materials and audit supplier quality control processes for incoming materials.
Lack of Robust Biocompatibility and Sterility Data
Assuming sterility and biocompatibility based on supplier statements is dangerous. Pitfalls include:
* Inadequate Sterility Assurance: Verifying the sterilization method (typically E-beam or gamma) and the associated SAL (Sterility Assurance Level, typically 10⁻⁶) is crucial. Request validation reports and batch sterility test results (e.g., direct inoculation or membrane filtration).
* Insufficient Biocompatibility Testing: Ensure the complete finished product (not just components) has undergone comprehensive testing per ISO 10993 (e.g., Cytotoxicity, Sensitization, Irritation, Systemic Toxicity, Genotoxicity). Relying on component data or outdated studies is insufficient and non-compliant.
Intellectual Property Infringement Risks
This is a critical, often overlooked pitfall. Calcium Alginate Ag technology is heavily patented. Sourcing from manufacturers without legitimate IP rights can expose you to severe legal and financial consequences:
* Infringing Patents: Many proprietary formulations, manufacturing processes (e.g., specific methods for incorporating silver), and dressing structures (e.g., fiber vs. non-woven) are protected. Using a “generic” product that copies a patented design infringes on the innovator’s rights.
* Lack of Freedom-to-Operate (FTO): Failing to conduct an FTO analysis before sourcing can lead to injunctions, product seizures, costly litigation, and reputational damage. Always require your supplier to warrant that their product does not infringe valid third-party IP and, ideally, provide evidence of their own patents or licensing agreements.
Inadequate Documentation and Regulatory Support
Poor or incomplete documentation hinders regulatory submissions and quality management. Pitfalls include:
* Missing or Generic Technical Files: Requiring access to a comprehensive Device Master Record (DMR) or Technical File, including design specifications, manufacturing processes, validation reports, and risk analysis (ISO 14971).
* Lack of Regulatory Dossier Support: For market entry (e.g., 510(k), CE Marking, NMPA), you need full regulatory support from the supplier, including access to their Quality Management System documentation and cooperation with audits.
Poor Supplier Reliability and Scalability
Assessing a supplier’s operational stability is vital. Pitfalls involve:
* Unproven Manufacturing Capacity: Ensure the supplier can reliably meet your volume demands consistently without compromising quality. Request production capacity data and audit manufacturing facilities.
* Supply Chain Vulnerability: Evaluate the robustness of their own supply chain for critical raw materials (alginate, silver) to avoid disruptions. Single-source dependencies without contingency plans are a significant risk.
* Inadequate Change Control: Suppliers must have strict procedures for managing changes to materials, processes, or design, with clear communication to customers. Lack of transparency here can lead to unexpected product changes.
Insufficient Clinical Evidence and Claims
Beware of suppliers making exaggerated or unsubstantiated clinical claims. While robust clinical data may be limited for some products, ensure any claims made are supported by in vitro efficacy data, relevant biocompatibility testing, and, ideally, published clinical studies. Marketing materials should not overstate benefits beyond the available evidence.
By proactively addressing these common pitfalls through rigorous supplier qualification, demanding comprehensive and verifiable documentation, conducting thorough due diligence on IP, and prioritizing quality and compliance at every step, organizations can successfully source safe, effective, and legally compliant Calcium Alginate Ag dressings.

Logistics & Compliance Guide for Calcium Alginate Ag Dressing
Product Overview
Calcium Alginate Ag Dressing is a sterile, absorbent wound care product infused with silver (Ag) to provide antimicrobial properties. It is designed for the management of moderate to heavily exuding wounds, including infected or at-risk-of-infection chronic and acute wounds such as leg ulcers, pressure ulcers, surgical wounds, and traumatic injuries. The dressing forms a gel upon contact with wound exudate, promoting a moist wound healing environment and facilitating non-traumatic removal.
Regulatory Classification
United States (FDA)
- Regulatory Pathway: Class II Medical Device
- 510(k) Cleared: Yes (Typically under product code NGS / k073221)
- Premarket Notification: Required unless previously cleared
- Establishment Registration: Required for manufacturers, distributors, and importers
- Labeling Requirements: Must comply with 21 CFR Part 801; includes UDI (Unique Device Identification)
European Union (EU)
- Classification: Class IIb Medical Device under Regulation (EU) 2017/745 (MDR)
- CE Marking: Required; demonstrated conformity with MDR
- Notified Body Involvement: Mandatory for Class IIb devices
- Technical Documentation: Must include risk management, clinical evaluation, and post-market surveillance (PMS)
- EU Representative: Required for non-EU manufacturers
- Post-Market Surveillance (PMS): Ongoing monitoring and reporting of adverse events
United Kingdom (UK)
- UKCA Marking: Required for placement on the UK market (England, Wales, Scotland)
- MHRA Registration: Mandatory for medical devices
- UK Responsible Person: Required for non-UK manufacturers
Canada (Health Canada)
- Classification: Class II Medical Device
- Medical Device License (MDL): Required for import and sale
- Quality Management System: Must comply with ISO 13485
- Labeling: Must include proper device identifier and manufacturer information
Australia (TGA)
- Classification: Class IIb
- ARTG Entry: Must be included in the Australian Register of Therapeutic Goods
- Sponsor Responsibility: Local sponsor required for non-Australian manufacturers
Labeling and Packaging Requirements
- Sterility Indication: Clearly state “Sterile” and method of sterilization (typically ethylene oxide or gamma irradiation)
- Single-Use Only: Marked as “For single use only”
- Expiry Date: Clearly printed on primary and secondary packaging
- Storage Conditions: Typically store in a cool, dry place away from direct sunlight; recommended range: 15°C to 30°C
- Contents: Include quantity, size, and sterile status
- UDI Compliance: Must include UDI in the form of a barcode (human- and machine-readable)
- Language Requirements: Local language(s) required in target markets (e.g., French in Canada, German in Germany)
Import and Export Compliance
Export Documentation
- Commercial Invoice
- Packing List
- Certificate of Manufacture and Conformity (CoC)
- Certificate of Free Sale (if required)
- Sterilization Certificate
- U.S. FDA Export Certificate (for U.S.-based exporters)
- MDR Declaration of Conformity (for EU exports)
Import Requirements
- Customs Classification: HS Code typically under 3005.90 (Wound dressings, medicated)
- Import License: May be required in certain countries (e.g., India, China, Saudi Arabia)
- Product Registration: Required in most regulated markets prior to import
- Customs Clearance: Ensure proper tariff classification and duty payment
Transportation and Storage
Shipping Conditions
- Mode of Transport: Suitable for air, sea, and ground freight
- Cold Chain: Not required; ambient transport acceptable within 15–30°C
- Humidity Control: Avoid high humidity to prevent packaging compromise
- Sterility Maintenance: Packaging must remain sealed and undamaged
Shelf Life
- Typically 3 years from date of manufacture
- Monitor batch-specific expiry dates during distribution
Quality and Safety Standards
- ISO 13485: Quality Management System for medical devices – mandatory for manufacturing and distribution
- ISO 10993: Biocompatibility testing (cytotoxicity, sensitization, irritation)
- EN 13795: Requirements for sterility in surgical dressings
- Antimicrobial Efficacy: Testing per ISO 20743 or JIS L 1902 for silver ion release and microbial reduction
Post-Market Surveillance and Vigilance
- Adverse Event Reporting: Required in all major markets (FDA MAUDE, EUDAMED, Health Canada MedEffect)
- Field Safety Notices (FSNs): Issue promptly for product corrections or withdrawals
- Periodic Safety Update Reports (PSURs): Required under EU MDR for Class IIb devices
- Complaint Handling: Maintain a robust system compliant with ISO 13485
Environmental and Disposal Considerations
- Disposal: Considered clinical waste; dispose of according to local biomedical waste regulations
- Silver Content: Note presence of silver; follow environmental guidelines for disposal in healthcare settings
- Packaging: Use recyclable materials where possible; comply with local packaging waste directives (e.g., EU Packaging Waste Directive)
Summary Checklist for Compliance
| Requirement | Status |
|————————————|——–|
| Valid CE Mark (EU) | ☐ |
| FDA 510(k) Clearance (US) | ☐ |
| UKCA Mark (UK) | ☐ |
| TGA ARTG Listing (AU) | ☐ |
| Health Canada MDL | ☐ |
| ISO 13485 Certification | ☐ |
| UDI Compliance | ☐ |
| Sterile, Single-Use Labeling | ☐ |
| Local Language Labeling | ☐ |
| Valid Shelf Life (≥12 months) | ☐ |
| Post-Market Surveillance System | ☐ |
This guide serves as a general reference for logistics and compliance considerations for Calcium Alginate Ag Dressings. Always consult local regulatory authorities and engage qualified regulatory consultants to ensure full compliance in each target market.
Conclusion:
Sourcing calcium alginate AG (antimicrobial silver) dressings requires a strategic approach that balances clinical effectiveness, regulatory compliance, cost-efficiency, and supply chain reliability. These advanced wound care products offer significant benefits in managing infected or high-risk chronic wounds due to their hemostatic properties, exudate absorption, and sustained silver release. When selecting a supplier, it is essential to evaluate product quality certifications (such as ISO and FDA approvals), consistency in manufacturing standards, and the availability of clinical evidence supporting efficacy. Additionally, considerations such as pricing, minimum order quantities, delivery timelines, and technical support play a crucial role in ensuring uninterrupted patient care. Establishing relationships with reputable manufacturers or distributors—preferably those with a proven track record in medical-grade alginate production—will contribute to reliable access to high-performing wound care solutions. Ultimately, a well-informed sourcing strategy for calcium alginate AG dressings enhances patient outcomes while supporting efficient and sustainable healthcare delivery.