The global whey protein market is experiencing robust expansion, driven by rising consumer demand for high-protein dietary supplements across fitness, sports nutrition, and medical nutrition applications. According to a 2023 report by Mordor Intelligence, the global protein supplements market was valued at USD 25.1 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 9.2% through 2028, with whey isolate—a premium, low-lactose, high-bioavailability form of whey protein—emerging as one of the fastest-growing segments. This surge is further supported by Grand View Research, which highlights that increasing awareness of health and wellness, coupled with the proliferation of active lifestyles, has intensified demand for pure, efficiently absorbed protein sources. As a result, bulk whey isolate manufacturing has become a highly competitive landscape, dominated by companies with advanced filtration technologies, strong quality assurance, and scalable production. In this environment, identifying the top manufacturers is essential for brands, supplement formulators, and distributors seeking reliable, high-quality supply chains. Below, we present the top 9 bulk whey isolate protein manufacturers globally, selected based on production capacity, certifications, purity standards, market reach, and third-party testing transparency.
Top 9 Bulk Whey Isolate Protein Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Whey Protein Isolates
Domain Est. 1996
Website: nzmp.com
Key Highlights: Our NZMP™ protein range of whey protein isolates are all made from fresh New Zealand milk. In fact, we’re the sole New Zealand manufacturer of WPI, and pioneers ……
#2 WHEY WHOLESALE
Domain Est. 2008
Website: mullinswhey.com
Key Highlights: WHEY PROTEIN ISOLATE Manufactured from sweet dairy whey using cold microfiltration and ultrafiltration membrane technology. The product is then gently spray ……
#3 Bulk™
Domain Est. 1996
Website: bulk.com
Key Highlights: Bulk™ is the leading supplier of bodybuilding supplements & sports nutrition – covering Protein, Creatine, Vitamins, Fat Loss & more! Formerly Bulk Powders….
#4 Whey Protein Isolate
Domain Est. 1996
Website: bulkfoods.com
Key Highlights: In stock $5 deliveryAll natural Instantized Whey Protein Isolate powder containing 90 percent protein without fillers or flavors added. (1/4 cup contains 20 grams protein)…
#5 ISO100® Hydrolyzed Protein Powder
Domain Est. 1997
Website: dymatize.com
Key Highlights: One of the highest-quality protein powders in the game, it’s filtered to remove excess lactose, carbs, fat, and sugar for maximum mixability and gains….
#6 Whey Protein Isolate 90% Powder
Domain Est. 2011
#7 Wisconsin Whey Protein
Domain Est. 2013
Website: wisconsinwhey.com
Key Highlights: Your preferred supplier of whey protein and lactose ingredients serving the infant, medical, health and wellness industries. Explore Our Products….
#8 Driven Nutrition
Domain Est. 2013
Website: drivennutrition.net
Key Highlights: Free delivery over $99 30-day returns…
#9 Whey Protein Isolate (BULK)
Domain Est. 2018
Website: bulkprotein.com
Key Highlights: This Instant (Easy to mix) Whey Protein Isolate is a product of processing by ultrafiltration the liquid whey from Mozzarella and Cheddar cheese manufacturing….
Expert Sourcing Insights for Bulk Whey Isolate Protein

H2: 2026 Market Trends for Bulk Whey Isolate Protein
The global market for bulk whey protein isolate (WPI) is poised for continued expansion and transformation in 2026, driven by evolving consumer demands, technological advancements, and shifting global dynamics. Key trends shaping the landscape include:
1. Heightened Demand for Ultra-Pure, Functional Ingredients: Consumers and manufacturers alike are demanding higher purity levels (>90% protein). This drives innovation in filtration technologies (like advanced microfiltration and chromatography) to produce WPI with minimal lactose (<0.5%), fat, and ash, catering to strict dietary needs (keto, paleo, low-FODMAP) and premium product formulations. Expect increased focus on bioactivity and specific functional properties (e.g., enhanced solubility, emulsification, gelling) beyond just protein content.
2. Sustainability & Transparency as Core Value Drivers: Environmental impact will be a major differentiator. Key trends include:
* Traceability: Demand for full supply chain transparency, from pasture to powder, with blockchain and digital platforms gaining traction.
* Carbon Footprint Reduction: Manufacturers will invest in renewable energy, optimized logistics, and low-impact packaging (recyclable, compostable) to meet ESG goals and consumer expectations.
* Regenerative Agriculture: Sourcing milk from farms practicing regenerative methods will become a premium selling point.
3. Diversification of End-Markets & Applications: While sports nutrition remains dominant, growth will accelerate in:
* Medical & Clinical Nutrition: WPI’s high digestibility and bioavailability make it ideal for medical foods, enteral formulas, and healthy aging/sarcopenia management products.
* Weight Management & Meal Replacements: Low-calorie, high-satiety WPI formulations will be crucial in functional foods and beverages.
* Bakery & Dairy Alternatives: Clean-label WPI is replacing traditional binders and fortifying plant-based products (yogurts, cheeses) for improved texture and protein content.
* Convenience Foods: Fortification of snacks, bars, and ready-to-eat meals with WPI for on-the-go nutrition.
4. Rise of Personalization & Niche Formulations: The “one-size-fits-all” approach is fading. Expect growth in:
* Targeted Blends: WPI combined with specific bioactive peptides (e.g., for immune support, muscle recovery, satiety) or other proteins (like collagen or plant proteins) for functional benefits.
* Specialty WPI: Hydrolyzed WPI for faster absorption (clinical/sports recovery), glycomacropeptide (GMP)-enriched WPI for specific health claims, and native WPI for cold-process applications.
5. Geopolitical & Supply Chain Resilience: The market will adapt to ongoing challenges:
* Regional Sourcing: Increased investment in regional production (e.g., North America, Europe, Asia) to reduce dependency on single regions and mitigate trade disruptions.
* Price Volatility Management: Fluctuations in milk prices and energy costs will persist. Long-term contracts, hedging strategies, and efficiency gains will be critical.
* Regulatory Harmonization: Efforts to align global standards (e.g., Codex) will continue, facilitating trade but requiring ongoing compliance.
6. Technological Advancements in Production: Efficiency and quality will be enhanced by:
* Process Optimization: AI and IoT for predictive maintenance, yield optimization, and real-time quality control.
* Water & Energy Efficiency: Closed-loop water systems and energy recovery technologies to reduce environmental impact and costs.
* Membrane Technology: Continued refinement of ultrafiltration and diafiltration for higher yields and purer fractions.
Conclusion: The 2026 bulk WPI market will be characterized by a push towards purity, functionality, sustainability, and application diversity. Success will depend on suppliers’ ability to innovate in technology, ensure transparent and resilient supply chains, and develop tailored solutions for expanding health, wellness, and functional food sectors. The focus will shift from being a simple protein source to being a high-performance, value-added ingredient.
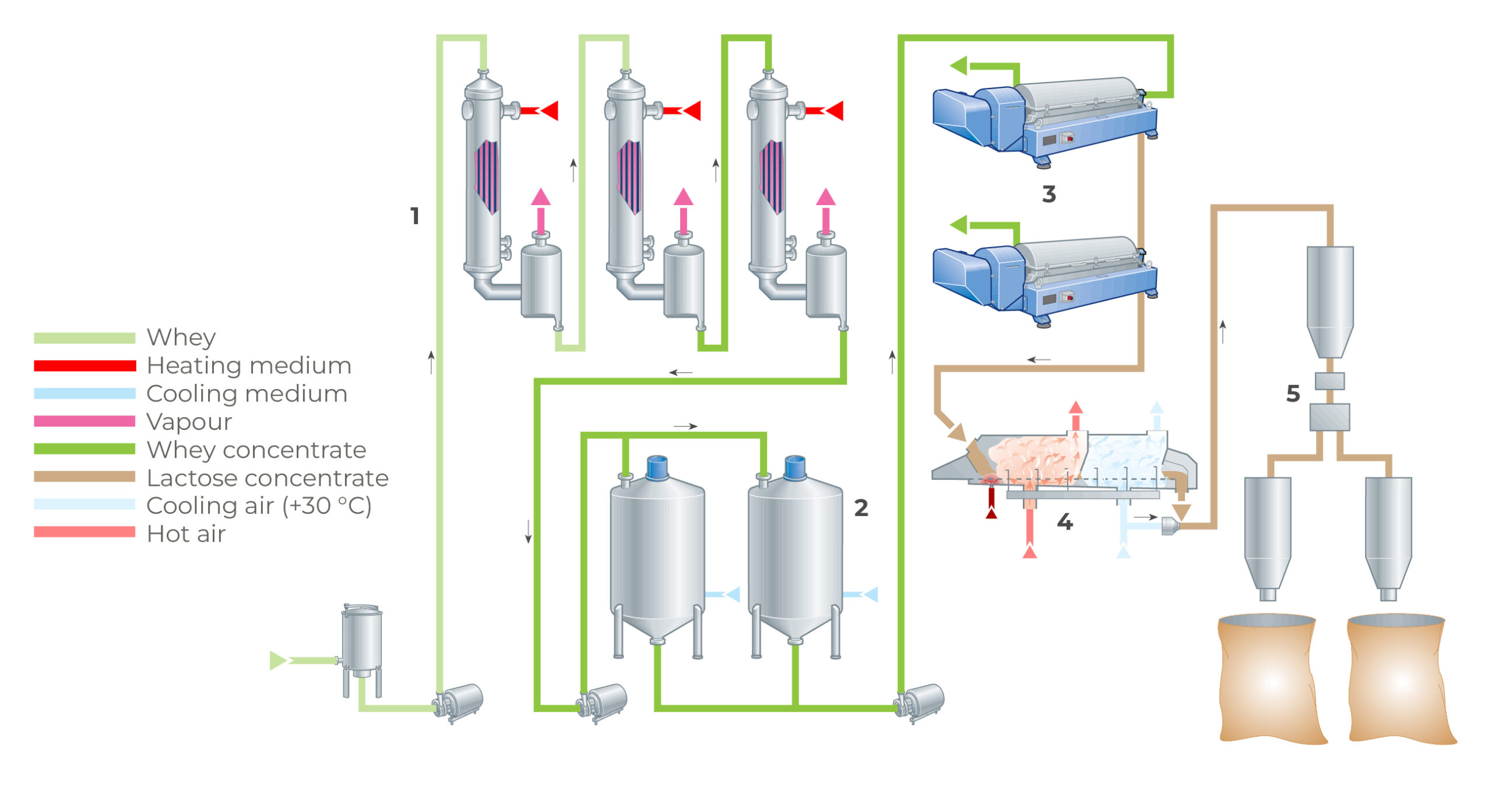
Common Pitfalls When Sourcing Bulk Whey Isolate Protein (Quality & IP)
Sourcing bulk whey protein isolate (WPI) requires careful attention to both quality and intellectual property (IP) considerations. Overlooking these aspects can lead to product failure, legal issues, and reputational damage. Below are key pitfalls to avoid:
Quality-Related Pitfalls
1. Inadequate Purity and Protein Content Verification
Not all WPI is created equal. A common mistake is assuming all isolates meet high standards. Some suppliers may provide products with protein content below 90%, often due to poor processing or adulteration. Always demand a Certificate of Analysis (CoA) with each batch and verify protein content via independent testing (e.g., Kjeldahl or Dumas method).
2. Poor Processing Methods Leading to Denatured Proteins
WPI is sensitive to heat and harsh processing. Suppliers using excessive heat during filtration or drying can denature proteins, reducing bioavailability and functional properties. Opt for suppliers using cold microfiltration or ultrafiltration techniques to preserve native protein structure.
3. Contamination Risks (Heavy Metals, Allergens, Microbial)
Bulk WPI can be contaminated with heavy metals (e.g., lead, cadmium), allergens (e.g., soy, gluten), or pathogens like Salmonella. Ensure suppliers follow Good Manufacturing Practices (GMP), conduct regular contaminant screening, and provide allergen control documentation.
4. Inconsistent Amino Acid Profile
Variability in amino acid composition affects the product’s nutritional value. This can result from inconsistent raw milk sources or processing fluctuations. Request amino acid profiles and ensure they align with your formulation requirements.
5. Misleading Labeling and Adulteration
Some suppliers blend WPI with cheaper proteins (e.g., hydrolyzed collagen, soy protein) or fillers to cut costs. Use third-party testing (e.g., mass spectrometry) to confirm ingredient authenticity and detect adulteration.
Intellectual Property (IP)-Related Pitfalls
1. Infringing on Patented Formulations or Processes
Certain WPI isolates are produced using patented technologies (e.g., specific filtration methods or bioactive peptide enrichment). Sourcing from a supplier using protected processes without licensing can expose your business to legal action. Conduct due diligence on the supplier’s manufacturing methods and IP status.
2. Lack of IP Ownership in Custom Blends
When developing proprietary blends containing WPI, ensure your agreement with the supplier clearly assigns IP rights to your company. Otherwise, the manufacturer may claim ownership of the formulation, limiting your ability to scale or switch suppliers.
3. Inadequate Protection of Trade Secrets
Sharing sensitive information (e.g., formulations, target markets) with suppliers without a robust Non-Disclosure Agreement (NDA) risks IP leakage. Always execute strong confidentiality agreements before disclosing any proprietary data.
4. Overlooking Trademark and Branding Conflicts
Using a supplier’s branded WPI (e.g., “Grass-Fed EliteWhey™”) without permission can lead to trademark infringement. Confirm whether the ingredient is generic or branded and obtain necessary rights for use in marketing.
5. Ignoring Regulatory Compliance in Claims
Health or performance claims tied to WPI (e.g., “clinically proven to increase muscle growth”) may be protected by the supplier’s IP or require regulatory substantiation. Ensure your marketing claims are legally defensible and do not violate third-party IP or regulatory standards (e.g., FDA, EFSA).
By proactively addressing these quality and IP pitfalls, businesses can secure reliable, compliant, and competitive bulk WPI supply chains.

Logistics & Compliance Guide for Bulk Whey Isolate Protein
Product Overview and Classification
Whey Protein Isolate (WPI) is a highly refined form of whey protein derived from milk during cheese production. It typically contains 90% or more protein by weight, with minimal fat and lactose. Bulk WPI is commonly traded in powder form in large quantities (e.g., 20–25 kg multi-wall paper bags with polyethylene liners, or in supersacks). It is classified under HS (Harmonized System) Code 0402.29 in most countries, which covers “whey, whether or not concentrated or containing added sugar or other sweetening matter, not for infant formula.”
Regulatory Compliance Requirements
Bulk WPI must comply with food safety and labeling regulations in both the country of origin and the destination market. Key regulatory bodies include the U.S. FDA (Food and Drug Administration), EU EFSA (European Food Safety Authority), Health Canada, FSANZ (Australia and New Zealand), and others. Producers and importers must ensure adherence to Good Manufacturing Practices (GMP), Hazard Analysis and Critical Control Points (HACCP), and relevant food additive regulations. WPI must be free from unauthorized substances, allergens (clearly labeled if milk is present), and contaminants such as heavy metals, pathogens (e.g., Salmonella, E. coli), and residual antibiotics.
Labeling and Documentation
Accurate labeling and documentation are critical for international trade. Each bulk shipment must include:
– Product name (“Whey Protein Isolate”)
– Net weight
– Batch/lot number
– Manufacturing and expiration dates
– Storage conditions (e.g., “Store in a cool, dry place”)
– Allergen statement (“Contains: Milk”)
– Manufacturer and/or distributor information
– Country of origin
Required documentation includes a Certificate of Analysis (CoA), Certificate of Origin, Commercial Invoice, Packing List, Bill of Lading/Air Waybill, and, where applicable, a Health Certificate issued by the national food safety authority. The CoA should verify protein content, moisture, fat, ash, microbiological safety, and compliance with heavy metal limits (e.g., lead, arsenic, cadmium).
Import and Export Regulations
Exporting and importing bulk WPI are subject to national and international trade regulations. Exporters must verify the destination country’s import requirements, which may include pre-shipment inspection, registration of foreign food facilities (e.g., FDA facility registration for U.S. imports), or special permits. Some countries require prior approval of the manufacturing facility or facility audits. Importers must ensure compliance with local food standards (e.g., EU Regulation (EC) No 178/2002, U.S. FSMA – Food Safety Modernization Act) and may be required to submit prior notice of imported food.
Storage and Handling Guidelines
Bulk WPI must be stored in a clean, dry, temperature-controlled environment (typically 15–25°C, relative humidity <60%) to prevent clumping, microbial growth, and degradation. It should be kept away from direct sunlight, strong odors, and incompatible materials. Handling areas must comply with food-grade hygiene standards. Use dedicated, sanitized equipment to avoid cross-contamination. FIFO (First In, First Out) inventory management is recommended to ensure product freshness and minimize spoilage.
Transportation and Shipping
WPI should be transported in dedicated, food-grade vehicles that are clean, dry, and free from contaminants. Containers or trucks must be sealed to prevent tampering and exposure to moisture or pests. Temperature control may be required in extreme climates. For international shipping, use intermodal containers with moisture barriers and desiccants if necessary. Ensure compliance with international transport regulations, such as the International Maritime Dangerous Goods (IMDG) Code (non-hazardous classification for WPI) and air transport regulations (IATA).
Quality Assurance and Testing
Suppliers should implement a robust quality management system (e.g., ISO 22000, FSSC 22000) and conduct routine batch testing. Testing parameters include:
– Protein content (via Kjeldahl or Dumas method)
– Moisture (≤5%)
– Fat (≤1%)
– Ash (≤5%)
– Lactose (≤1%)
– Microbial limits (total plate count, coliforms, Salmonella, E. coli, yeast/mold)
– Heavy metals (lead, arsenic, cadmium, mercury)
– Pesticide and antibiotic residues
Third-party laboratory testing and audit reports enhance credibility and support compliance during customs clearance.
Allergen and Contamination Control
As a milk-derived product, WPI is a known allergen. Facilities producing or handling WPI must have allergen control programs in place, including dedicated production lines or thorough cleaning protocols between runs. Prevent cross-contact with common allergens (e.g., soy, gluten, nuts). Facilities should validate cleaning effectiveness and conduct allergen swab testing. Labeling must clearly state “Contains: Milk” in accordance with local food labeling laws.
Sustainability and Ethical Sourcing
Increasingly, buyers require evidence of sustainable and ethical sourcing. This includes responsible dairy farming practices, animal welfare standards (e.g., following FARM or RSPCA guidelines), and environmental impact assessments (e.g., carbon footprint, water usage). Suppliers may need to provide certifications such as Non-GMO Project Verified, Organic (e.g., USDA Organic, EU Organic), or Fair Trade, depending on market demand.
Recalls and Traceability
A full traceability system—from raw milk to finished bulk WPI—is essential. Each batch should be traceable through a unique identifier linked to raw material sources, processing data, and distribution records. In the event of a contamination or compliance issue, a recall plan must be in place to quickly identify and retrieve affected batches. Notify relevant regulatory authorities promptly and follow jurisdiction-specific recall procedures.
Conclusion: Sourcing Bulk Whey Isolate Protein
Sourcing bulk whey isolate protein requires a strategic approach that balances quality, cost, and reliability. Whey isolate, known for its high protein content, low lactose, and minimal fat, is ideal for athletes, fitness enthusiasts, and health-conscious consumers. When procuring in bulk, it is essential to partner with reputable suppliers who adhere to stringent manufacturing standards, provide third-party testing, and offer transparent documentation such as Certificates of Analysis (COA).
Key considerations include verifying the protein’s purity (typically 90%+ protein by weight), ensuring it is free from contaminants, and confirming compliance with food safety regulations (e.g., FDA, cGMP, ISO certification). Additionally, evaluating factors like minimum order quantities (MOQ), lead times, packaging options, and scalability ensures long-term supply stability.
Ultimately, successful bulk sourcing not only reduces per-unit costs but also supports consistent product quality, brand reputation, and customer satisfaction. By conducting thorough due diligence and fostering strong supplier relationships, businesses can secure a reliable supply of high-quality whey isolate protein to meet growing market demands.