The global infant formula market is experiencing robust growth, driven by rising demand for safe, nutritionally balanced feeding alternatives and increasing awareness of early-life nutrition. According to Grand View Research, the market was valued at USD 72.5 billion in 2023 and is projected to expand at a compound annual growth rate (CAGR) of 6.8% from 2024 to 2030. This surge is fueled by expanding middle-class populations, urbanization, and heightened regulatory standards—particularly in Asia-Pacific and emerging markets. As demand escalates, bulk formula manufacturers play a pivotal role in meeting supply needs for private-label brands, pharmaceutical companies, and retail chains. The following list highlights the top 10 bulk formula manufacturers leveraging scale, innovation, and compliance to lead this evolving landscape.
Top 10 Bulk Formula Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Formula Corp
Domain Est. 1996 | Founded: 1983
Website: formulacorp.com
Key Highlights: Formula Corp. is a manufacturer of custom blended chemicals used in personal care, sanitary maintenance, and industrial cleaning markets since 1983….
#2 Enfamil Newborn, Infant & Toddler Nutrition
Domain Est. 1996
Website: enfamil.com
Key Highlights: Discover tailored formulas to help meet your child’s nutritional needs. Pediatrician recommended for newborns, infants and toddlers. Find expert support….
#3 Similac Formula and Nutrition Products
Domain Est. 2005
Website: abbottnutrition.com
Key Highlights: Similac has a comprehensive portfolio including formulas for routine feeding, specialized nutrition, and innovations like formulas with 5 HMOs….
#4 Infant Formula
Domain Est. 2009
Website: lamedicalwholesale.com
Key Highlights: 7-day returnsLearn more about Infant Formula and place an order to purchase from LA Medical Wholesale….
#5 Novalac Infant & Toddler Formula
Domain Est. 2010
Website: novalac.me
Key Highlights: Pioneer in formulas for infants with acid reflux, colics, gas, constipation, allergy & more, Novalac’s goal is to improve solutions for babies from birth up ……
#6 Formuland: European Baby Formula
Domain Est. 2014
Website: formuland.com
Key Highlights: We offer a wide selection of high-quality European formula brands, including HiPP, Holle, Lebenswert, Kabrita, and so much more….
#7 Happy Family Organics
Domain Est. 2016
Website: happyfamilyorganics.com
Key Highlights: Mom-founded and parent-operated, Happy Family Organics provides helpful resources and premium organic products for babies, tots, kids, and mamas….
#8 Organic Life Start® // Shop Organic Formulas
Domain Est. 2021
Website: organiclifestart.com
Key Highlights: Shop for the finest European organic, non GMO formulas, from trusted brands like HiPP, Holle, and Lebenswert. From grass-fed Swiss and German cows, ……
#9 Clean Infant Milk Formula
Domain Est. 2021
Website: aussiebubs.com
Key Highlights: Free delivery over $100 30-day returnsExplore Bubs clean infant formula made from goat and cow milk—gentle, nutritious, and designed to support healthy development from day one….
#10 Organic’s Best
Domain Est. 2019
Website: organicsbestshop.com
Key Highlights: Shop European organic baby formula and baby food from top brands like HiPP, Holle &more. Unbeatable bulk pricing, express delivery, authentic products!…
Expert Sourcing Insights for Bulk Formula

H2: Projected Market Trends for Bulk Formula in 2026
Looking ahead to 2026, the bulk formula market—encompassing private-label or unbranded nutritional supplements, protein powders, meal replacements, and specialty blends sold in large quantities—is poised for significant evolution shaped by shifting consumer demands, technological advancements, and macroeconomic factors. Key trends expected to define the landscape include:
1. Heightened Demand for Personalization and Customization
By 2026, consumers will increasingly seek tailored nutrition solutions. Bulk formula suppliers will respond by offering modular or customizable blends (e.g., mix-and-match protein bases, added adaptogens, or gut health boosters). Advances in AI-powered nutrition platforms will enable retailers and brands to create region- or demographic-specific formulas, driving demand for flexible bulk manufacturing with scalable small-batch capabilities.
2. Clean Label and Transparency as Non-Negotiables
Consumers will prioritize ingredient transparency, traceability, and sustainability. Bulk formula producers will need to provide full supply chain documentation, non-GMO, vegan, and allergen-free certifications, and clean labels free of artificial additives. Blockchain-enabled ingredient tracking is expected to gain traction, enhancing trust and differentiating premium suppliers.
3. Rise of Functional and Cognitive Support Formulas
Beyond basic protein and vitamins, demand will surge for formulas targeting mental clarity, stress resilience, immune support, and metabolic health. Ingredients like lion’s mane mushroom, ashwagandha, L-theanine, and postbiotics will become common in bulk nootropic and adaptogenic blends. Cognitive wellness, especially among aging populations and high-performance demographics, will be a major growth vector.
4. Sustainability and Eco-Conscious Packaging
Environmental impact will heavily influence purchasing decisions. Bulk formula brands and manufacturers will shift toward biodegradable, compostable, or reusable packaging solutions. Carbon-neutral production and regenerative agricultural sourcing will become key selling points, with third-party certifications (e.g., B Corp, Climate Neutral) adding competitive advantage.
5. Expansion of Direct-to-Consumer (DTC) and E-commerce Channels
E-commerce will remain a dominant sales channel, with DTC brands leveraging social media, influencer marketing, and subscription models. Bulk formula suppliers will increasingly offer white-label solutions optimized for digital storefronts, including portioned sachets, sample kits, and digital content (e.g., usage guides, QR-linked educational material).
6. Regulatory Scrutiny and Quality Standardization
Regulatory bodies in the U.S., EU, and Asia are expected to tighten oversight on supplement claims, contamination, and labeling. Leading bulk manufacturers will invest in advanced third-party testing, GMP (Good Manufacturing Practice) compliance, and real-time quality monitoring to meet higher standards and reduce liability risks.
7. Geopolitical and Supply Chain Resilience
Ongoing global instability will push companies to diversify ingredient sourcing and regionalize production. Nearshoring and dual-sourcing strategies will reduce dependency on single regions (e.g., Asia for botanicals), with increased investment in local fermentation and plant-based protein facilities in North America and Europe.
8. Integration with Digital Health Ecosystems
Bulk formula will increasingly integrate with wearables, health apps, and telehealth platforms. Personalized supplement regimens based on biometric data (e.g., sleep, activity, glucose levels) will drive demand for interoperable, data-informed bulk formulations, especially in the sports nutrition and longevity markets.
Conclusion
By 2026, the bulk formula market will be defined by innovation, accountability, and personalization. Success will depend on agility in formulation, commitment to sustainability, and the ability to deliver scientifically backed, transparent products through digitally enabled supply chains. Manufacturers who proactively align with these trends will capture significant market share in an increasingly competitive and conscientious landscape.

Common Pitfalls Sourcing Bulk Formula (Quality, IP)
Sourcing bulk infant formula or adult nutritional formula presents significant challenges, particularly concerning product quality and intellectual property (IP). Failing to address these issues can lead to regulatory violations, reputational damage, and legal liabilities. Below are key pitfalls to avoid:
Quality Control and Consistency
One of the most critical risks when sourcing bulk formula is ensuring consistent product quality. Nutritional formulas are highly sensitive to variations in raw materials, manufacturing processes, and storage conditions.
- Inconsistent Raw Material Sourcing: Suppliers may source ingredients from multiple vendors without proper vetting, leading to variability in nutrient profiles, allergen presence, or contamination risks (e.g., pathogens like Cronobacter).
- Lack of cGMP Compliance: Not all manufacturers adhere to current Good Manufacturing Practices (cGMPs), especially in regions with lax regulatory oversight. This increases the risk of contamination, improper labeling, and batch inconsistencies.
- Inadequate Testing Protocols: Some suppliers perform minimal or no third-party testing for microbial contaminants, heavy metals, nutritional content, or adulterants. Relying solely on supplier-provided certificates of analysis (CoA) without verification is a major oversight.
- Poor Supply Chain Transparency: Opaque supply chains make it difficult to trace ingredients back to origin, complicating recalls and increasing vulnerability to fraud or contamination events.
Intellectual Property Infringement
Bulk formula often incorporates proprietary blends, patented formulations, or trademarked processes. Sourcing without proper IP due diligence can lead to legal disputes and market exclusion.
- Unauthorized Use of Patented Formulations: Many advanced formulas (e.g., hydrolyzed proteins, specific probiotic strains, or lipid blends) are protected by patents. Sourcing a “similar” formula from a third party may still infringe on composition, method of use, or manufacturing process patents.
- Copying Branded Product Profiles: Attempting to replicate a competitor’s marketed formula—even if ingredients are legally sourced—can violate trade dress or unfair competition laws if the product is too similar in labeling, claims, or appearance.
- Lack of Licensing Agreements: Some ingredients (e.g., specific DHA/ARA blends or patented prebiotics) require licensing for use in formula. Sourcing bulk product containing these without proper authorization exposes the buyer to infringement claims.
- Weak Contractual Protections: Supplier agreements that fail to include IP warranties, indemnification clauses, or clear ownership of formulations leave buyers vulnerable if the product is later challenged for IP violations.
Regulatory and Labeling Risks
Bulk formula must comply with complex regulations (e.g., FDA, EU Commission, or local health authorities), and non-compliance often stems from sourcing decisions.
- Non-Compliant Nutrient Profiles: Formulas must meet strict compositional standards. Suppliers may adjust formulations to reduce costs, risking non-compliance with mandatory nutrient levels.
- Misleading or Inaccurate Labeling: Bulk suppliers may provide incorrect or incomplete labeling data, especially for allergens, nutrition facts, or ingredient sourcing—leading to recalls or regulatory action.
- Unapproved Health Claims: Some suppliers include unauthorized structure/function or disease claims on bulk product documentation, which can implicate the buyer upon resale or repackaging.
Mitigation Strategies
To avoid these pitfalls:
– Conduct thorough audits of supplier facilities and quality systems.
– Require independent, batch-specific testing from accredited labs.
– Verify all ingredients and formulations for patent and trademark conflicts.
– Secure clear contractual terms on quality, IP ownership, and liability.
– Work with legal and regulatory experts familiar with food and supplement laws in target markets.
Sourcing bulk formula demands rigorous due diligence—compromising on quality or IP can result in severe financial and reputational consequences.
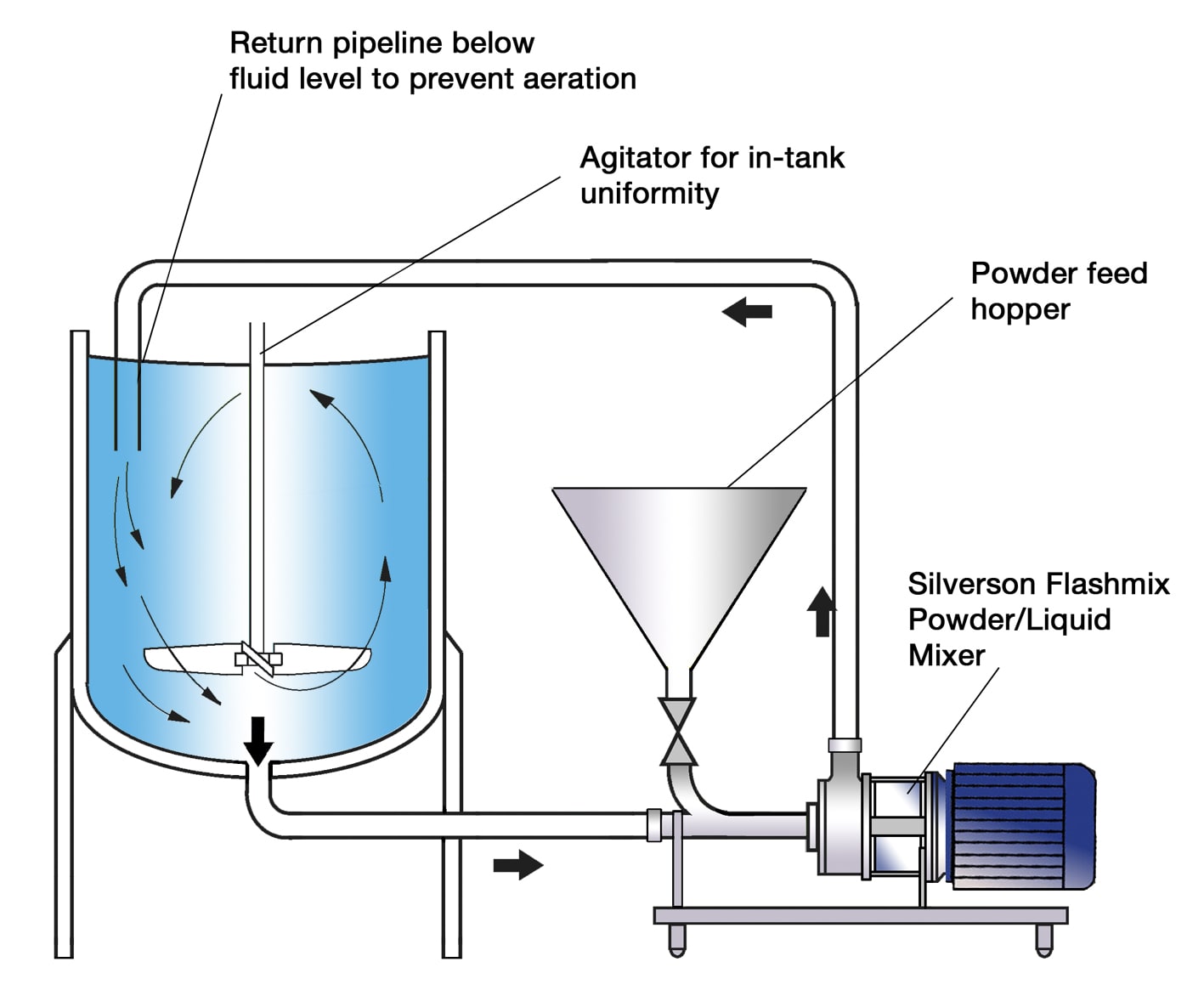
Logistics & Compliance Guide for Bulk Formula
Overview
This guide outlines the key logistics and compliance considerations when handling, storing, transporting, and documenting bulk infant formula. Due to its sensitive nature and regulatory scrutiny, strict adherence to food safety, labeling, and transportation standards is required to ensure product integrity and legal compliance.
Regulatory Compliance
Bulk formula is subject to stringent regulations from agencies such as the U.S. Food and Drug Administration (FDA), the European Food Safety Authority (EFSA), and Codex Alimentarius. Key compliance areas include:
– FDA 21 CFR Part 106 & Part 107: Governs manufacturing, labeling, and quality control for infant formula.
– FSMA (Food Safety Modernization Act): Requires risk-based preventive controls and traceability throughout the supply chain.
– Labeling Requirements: Bulk containers must include product name, ingredient list, nutritional information, lot number, use-by date, storage instructions, and manufacturer details.
– Registration: Facilities involved in manufacturing, packing, or holding infant formula must be registered with the FDA.
Manufacturing & Quality Control
- Facilities must implement Current Good Manufacturing Practices (cGMPs).
- Each batch must undergo microbiological and nutritional testing before release.
- Maintain detailed batch records for traceability (minimum 3 years).
- Conduct regular audits to verify compliance with internal and external standards.
Packaging & Container Requirements
- Use food-grade, tamper-evident containers made of materials approved for direct food contact.
- Ensure bulk containers (e.g., totes, drums, tanks) are clean, sanitized, and dedicated to infant formula to prevent cross-contamination.
- Clearly label all containers with batch-specific information for full traceability.
Storage Conditions
- Store in a dry, temperature-controlled environment (typically 15–25°C / 59–77°F).
- Protect from direct sunlight, moisture, and contaminants.
- Implement FIFO (First In, First Out) inventory management.
- Monitor and record storage conditions regularly.
Transportation & Distribution
- Use dedicated or thoroughly sanitized vehicles to prevent cross-contact.
- Maintain cold chain if required by formulation (verify per product specification).
- Secure loads to prevent container damage during transit.
- Require carriers to comply with FSMA’s Sanitary Transportation Rule (21 CFR Part 1, Subpart O).
- Provide shipping documentation including bill of lading, certificate of analysis (CoA), and handling instructions.
Import/Export Compliance
- Ensure compliance with destination country regulations (e.g., EU Commission Directive 2006/141/EC).
- Prepare accurate Harmonized System (HS) codes, commercial invoices, and certificates of origin.
- Obtain necessary permits and pre-notify customs authorities where required.
- Verify that foreign manufacturers are listed with the FDA under interim final rule for importation.
Recordkeeping & Traceability
- Maintain comprehensive records for:
- Supplier verification
- Batch production and testing
- Distribution logs (full forward and backward traceability)
- Recall plans and mock recalls
- Records must be accessible within 4 hours of request during FDA inspections.
Recall Preparedness
- Develop and regularly test a written recall plan.
- Assign a recall coordinator and response team.
- Ensure systems are in place to quickly identify affected lots and notify distributors/customers.
- Coordinate with FDA within 24 hours of initiating a recall.
Training & Personnel
- Train all personnel involved in handling bulk formula on:
- GMPs and hygiene practices
- Allergen control
- FSMA requirements
- Emergency response procedures
- Maintain training records and conduct annual refreshers.
Conclusion
Proper logistics and compliance management for bulk formula is critical to ensuring the safety and quality of a nutritionally sensitive product. Adhering to regulatory standards, maintaining rigorous documentation, and implementing robust quality systems will minimize risks and support smooth operations across the supply chain.
Conclusion for Sourcing Bulk Formula
Sourcing bulk formula requires careful consideration of quality, safety, regulatory compliance, supplier reliability, and cost-efficiency. As demand for infant and specialty nutritional formulas continues to grow, establishing partnerships with reputable manufacturers who adhere to stringent production standards—such as FDA, EU, or GMP certifications—is essential. Conducting thorough due diligence, including facility audits, ingredient traceability checks, and product testing, helps mitigate risks associated with contamination or non-compliance.
Additionally, logistics, storage requirements, and import/export regulations must be factored in, especially when sourcing internationally. Building long-term relationships with suppliers who offer transparency, scalability, and consistent product quality ensures supply chain resilience and brand integrity.
In summary, successful bulk formula sourcing hinges on balancing affordability with uncompromising quality and regulatory adherence. A strategic, well-informed approach not only safeguards consumer health but also supports sustainable business growth in a highly sensitive and competitive market.