The global fermentation equipment market is experiencing robust growth, driven by rising demand in the food and beverage, pharmaceutical, and biofuel industries. According to a report by Mordor Intelligence, the fermentation equipment market was valued at USD 11.63 billion in 2023 and is projected to reach USD 17.27 billion by 2029, growing at a CAGR of 6.7% during the forecast period. This expansion is fueled by increasing investments in biopharmaceuticals, a surge in probiotic consumption, and advancements in industrial biotechnology. As demand for large-scale fermentation escalates, the need for reliable, high-capacity bulk fermentation containers has become critical. Leading manufacturers are responding with scalable, hygienic, and technologically advanced solutions to meet stringent industry standards. In this evolving landscape, identifying key suppliers capable of delivering consistent quality and performance is essential for producers aiming to optimize yield, maintain sterility, and scale operations efficiently. The following list highlights the top eight bulk fermentation container manufacturers shaping the industry with proven track records, innovative engineering, and global supply capabilities.
Top 8 Bulk Fermentation Container Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Air Products:
Domain Est. 1995
Website: airproducts.com
Key Highlights: Air Products provide essential industrial gases, related equipment and applications expertise to customers in dozens of industries. Find out more….
#2 Stainless Steel Storage & Processing Vessels Nationwide
Domain Est. 1996
Website: dciinc.com
Key Highlights: At DCI, Inc., we serve the cosmetics and personal care industries with bulk storage vessels and equipment for mixing, blending, and batching applications….
#3 Plastic Packaging Manufacturer & Product Innovation
Domain Est. 2000
Website: packagingsolutions.amcor.com
Key Highlights: Amcor, leaders in plastic packaging manufacturing, process & sustainable product innovation & design. Learn more about our capabilities….
#4 Paul Mueller Company
Domain Est. 1995
Website: paulmueller.com
Key Highlights: Paul Mueller Company specializes in the design and manufacturing of stainless steel processing equipment and systems for a wide range of industries….
#5 Klosterman Baking Company
Domain Est. 1998
Website: klostermanbakery.com
Key Highlights: Klosterman Bakery, a trusted nationwide wholesale bakery delivering artisan bread, buns, and donuts to restaurants, grocers & institutions across the U.S ……
#6 Snyder Industries: Poly Tanks
Domain Est. 1998
Website: snydernet.com
Key Highlights: Snyder Industries manufactures plastic & steel tanks, IBC totes, bins, containers, pallets & more including custom products….
#7 Cultures For Health
Domain Est. 2008
Website: culturesforhealth.com
Key Highlights: Cultures for Health offers more than just high-quality cultures – we’re your expert guide in the home fermentation journey….
#8 Ss Brewtech
Domain Est. 2013
Website: ssbrewtech.com
Key Highlights: 4–7 day delivery Free 30-day returnsSs Brewtech engineers high end stainless steel equipment for brewing beer, kombucha, and much more at home or professionally….
Expert Sourcing Insights for Bulk Fermentation Container
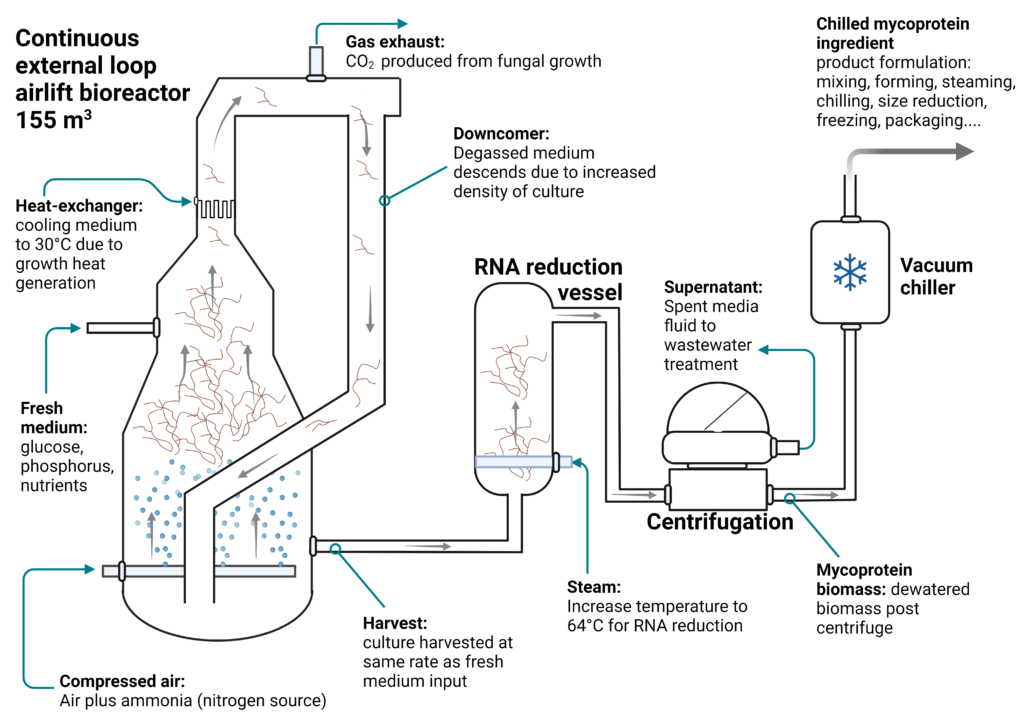
2026 Market Trends for Bulk Fermentation Containers
The global market for bulk fermentation containers is poised for significant transformation by 2026, driven by advancements in biotechnology, sustainability imperatives, and evolving production demands. Key trends shaping this landscape include:
H2: Dominance of Single-Use Bioreactors and Disposables
A pivotal trend propelling the market is the accelerating shift from traditional stainless steel systems to single-use bioreactors (SUBs) and disposable fermentation containers. This transition is fueled by compelling advantages: dramatically reduced cleaning and sterilization costs, minimized risk of cross-contamination, faster turnaround between batches, and greater flexibility in multi-product facilities. By 2026, single-use technologies are expected to capture a dominant share, particularly in biopharmaceutical manufacturing (e.g., monoclonal antibodies, vaccines) and high-value nutraceuticals, where batch integrity and speed-to-market are critical. Innovations in film materials, sensor integration, and larger-scale disposable bags (up to 2000L+) will further solidify this trend.
H2: Integration of Advanced Process Monitoring and Automation
The demand for enhanced process control, consistency, and data integrity is driving the integration of sophisticated sensors and automation into bulk fermentation containers. By 2026, intelligent bioreactors equipped with real-time monitoring for parameters like pH, dissolved oxygen (DO), glucose, and viable cell density will become standard. This shift towards Process Analytical Technology (PAT) and Quality by Design (QbD) frameworks enables predictive maintenance, adaptive control strategies, and data-driven optimization. Cloud-based platforms and AI-powered analytics will allow remote monitoring and centralized management of fermentation processes across global facilities, improving efficiency and reducing failures.
H2: Focus on Sustainability and Circular Economy
Growing environmental regulations and corporate sustainability goals are pushing the industry towards more eco-friendly solutions. While single-use systems offer operational benefits, their plastic waste footprint is a major concern. In response, key trends by 2026 will include: development of bio-based or recyclable films for disposable bags, expansion of take-back and recycling programs by major suppliers, and increased adoption of hybrid approaches combining reusable components with disposable liners. Additionally, energy-efficient designs for stainless steel systems and water-saving cleaning technologies will gain traction, reflecting a broader industry commitment to reducing the environmental impact of fermentation operations.
H2: Expansion into Novel Applications and Diversified Sectors
Beyond traditional pharmaceuticals and industrial enzymes, bulk fermentation containers are finding new applications in rapidly growing sectors. The cultivated meat and alternative protein industry will drive demand for large-scale, high-performance bioreactors capable of supporting animal cell growth. Similarly, the bio-based chemicals and sustainable materials market (e.g., bioplastics, biofuels) will require cost-effective, scalable fermentation solutions. This diversification is pushing innovation in container design for specific cell types (e.g., mycelium, algae) and process conditions, leading to more specialized and application-optimized systems by 2026.
H2: Regional Market Growth and Localization
Geopolitical factors and supply chain resilience are influencing regional investment patterns. While North America and Europe remain strong markets due to advanced biotech hubs, significant growth is expected in the Asia-Pacific region—particularly China, India, and South Korea—driven by government support for biomanufacturing, rising domestic demand for biologics, and expanding CMO/CDMO capacity. This regional shift will encourage localized production of fermentation containers and components, reducing lead times and logistical risks. By 2026, a more balanced global manufacturing footprint will characterize the market.

Common Pitfalls When Sourcing Bulk Fermentation Containers (Quality and Intellectual Property)
Sourcing bulk fermentation containers—especially for food, beverage, or biopharmaceutical applications—requires careful consideration beyond just cost and capacity. Overlooking quality and intellectual property (IP) aspects can lead to operational failures, regulatory non-compliance, and legal exposure. Below are key pitfalls to avoid:
Quality-Related Pitfalls
1. Compromising on Material Specifications
Choosing substandard materials (e.g., non-food-grade stainless steel such as 304 instead of 316L) can result in corrosion, contamination, and shortened equipment life. Inadequate surface finishes (e.g., Ra > 0.8 µm) may harbor microbial growth, compromising product safety and process hygiene.
2. Inadequate Design for Cleanability and Sanitation
Containers with poor weld quality, dead legs, or inaccessible areas hinder effective CIP (Clean-in-Place) and SIP (Sterilize-in-Place) procedures. This increases biofilm risk and regulatory scrutiny, especially in GMP environments.
3. Overlooking Pressure and Temperature Ratings
Fermentation processes may involve overpressure from CO₂ or require steam sterilization. Sourcing containers without proper ASME or PED certification for the required operating conditions can lead to safety hazards and system failure.
4. Ignoring Third-Party Certification and Documentation
Failure to obtain material test reports (MTRs), pressure vessel certifications, or FDA/EC 1935/2004 compliance documentation can delay validation and regulatory submissions. Lack of traceability increases audit risk.
5. Poor Manufacturing and Welding Practices
Low-cost suppliers may use inexperienced welders, leading to porosity, cracks, or inconsistent passivation. This compromises structural integrity and sterility—especially problematic in aseptic fermentations.
Intellectual Property-Related Pitfalls
1. Infringing on Patented Designs or Technologies
Some bulk fermentation systems incorporate proprietary features (e.g., specialized agitators, gas sparging systems, or sensor integrations). Sourcing a container that replicates patented designs—even unknowingly—can result in infringement lawsuits and costly recalls.
2. Lack of IP Clarity in Custom Designs
When working with suppliers on custom container designs, failure to define IP ownership in contracts can result in disputes. Suppliers may claim rights to design improvements or reuse your specifications for competitors.
3. Insufficient Protection of Process-Specific Features
Unique configurations (e.g., baffling, port placement, or internal coatings) tailored to your fermentation process may not be adequately protected. Competitors could reverse-engineer or source similar containers if these features are not patented or kept as trade secrets.
4. Supplier Use of Your Specifications for Marketing
Uncontrolled disclosure of technical drawings or performance data to suppliers may allow them to market similar products to your competitors. Always use non-disclosure agreements (NDAs) and limit technical disclosure to what’s essential.
5. Dependency on Proprietary Interfaces or Software
Some modern fermentation containers include integrated control systems or IoT sensors. Sourcing such systems without clear IP licensing terms may restrict your ability to modify, service, or integrate equipment, creating vendor lock-in.
Mitigation Strategies
- Conduct thorough supplier audits and request full technical documentation.
- Require compliance with relevant standards (ASME BPE, FDA, EHEDG).
- Engage legal counsel to review contracts for IP ownership and liability clauses.
- File provisional patents or maintain key design elements as trade secrets.
- Use NDAs and restrict access to sensitive design information.
Avoiding these pitfalls ensures not only reliable, compliant fermentation operations but also protects your competitive advantage and reduces long-term risk.

Logistics & Compliance Guide for Bulk Fermentation Containers
Overview and Purpose
Bulk fermentation containers are specialized vessels used in the biopharmaceutical, food, and beverage industries to cultivate microorganisms or cells at large scale. Proper logistics and compliance management are essential to maintain product integrity, ensure regulatory adherence, and prevent contamination or spoilage. This guide outlines best practices for handling, transporting, storing, and complying with regulations when using bulk fermentation containers.
Container Specifications and Handling
Bulk fermentation containers are typically made from single-use bioprocess bags, stainless steel, or glass-lined steel, depending on the application. They range from 100L to over 20,000L in capacity. Key handling considerations include:
– Material Compatibility: Ensure the container material is compatible with the culture medium and process conditions (e.g., pH, temperature, shear stress).
– Sterility Assurance: Single-use systems must be pre-sterilized (gamma-irradiated or ETO) and remain sealed until use. Reusable containers must undergo validated cleaning and sterilization (SIP – Steam-in-Place).
– Handling Protocols: Use proper lifting equipment (e.g., forklifts with cradles, overhead hoists) to prevent damage. Avoid dragging or dropping containers.
Transportation Requirements
Transporting bulk fermentation containers involves strict controls to maintain sterility and prevent physical damage:
– Packaging: Use rigid outer containers with cushioning to protect flexible bioprocess bags. For reusable vessels, secure with locking mechanisms and protective wraps.
– Temperature Control: If the container holds live cultures or temperature-sensitive media, use refrigerated or climate-controlled transport. Monitor with data loggers.
– Orientation and Labeling: Clearly label containers with “This Side Up,” “Fragile,” and biohazard symbols if applicable. Maintain upright orientation to prevent leaks.
– Shipping Documentation: Include a Certificate of Conformance (CoC), shipping manifest, and any required permits (e.g., for hazardous biological materials).
Storage Conditions
Proper storage ensures container readiness and prevents degradation:
– Environment: Store in a clean, dry, temperature-controlled area (typically 15–25°C). Avoid direct sunlight and high humidity.
– Sterility Maintenance: Keep single-use containers in original sealed packaging. Reusable vessels should be stored clean, dry, and capped to prevent contamination.
– Shelf Life: Adhere to manufacturer-specified shelf life for single-use systems (usually 2–3 years). Reusable containers have no expiration but require periodic inspection and requalification.
Regulatory Compliance
Bulk fermentation processes and containers are subject to multiple regulatory frameworks:
– FDA 21 CFR Part 211 (cGMP): Requires validation of equipment and processes, including container cleaning and sterilization for pharmaceutical applications.
– EMA Guidelines: Similar to FDA, EMA mandates quality risk management and documentation for biopharmaceutical manufacturing in the EU.
– USP <797> and <825>: Apply if sterile compounding or radiopharmaceuticals are involved; dictate container handling and environmental controls.
– REACH and RoHS: For containers containing plastics or electronic components, ensure compliance with chemical safety and hazardous substance restrictions.
– Customs and International Trade: For cross-border shipments, comply with import/export regulations, including phytosanitary certificates for biological materials (e.g., under IPPC ISPM 15 for wooden pallets).
Validation and Documentation
Robust documentation supports compliance and traceability:
– IQ/OQ/PQ (Installation, Operational, and Performance Qualification): Required for reusable fermentation vessels to prove fitness for use.
– Batch Records: Maintain logs for each fermentation run, including container ID, sterilization date, and usage history.
– Traceability: Assign unique identifiers to single-use containers and track from receipt to disposal.
– Audit Readiness: Maintain records for at least 5 years (or per local regulation) for regulatory inspections.
Safety and Environmental Considerations
- Biohazard Waste Disposal: Decontaminate and dispose of used single-use containers according to local biohazard regulations (e.g., autoclaving before landfill).
- Chemical Safety: Handle cleaning agents (e.g., NaOH, HNO₃) used in reusable vessel sanitation with appropriate PPE and spill controls.
- Spill Response Plan: Have protocols in place for leaks or ruptures, including containment, decontamination, and reporting.
Conclusion
Effective logistics and compliance management of bulk fermentation containers are vital for operational success and regulatory approval. By adhering to standardized handling, transportation, storage, and documentation practices—and staying current with evolving regulations—organizations can ensure product safety, quality, and continuity in large-scale fermentation operations.
In conclusion, sourcing a bulk fermentation container requires careful consideration of several critical factors including material quality (preferably food-grade stainless steel), capacity, design features (such as airlocks, spigots, and seal integrity), and compliance with hygiene and safety standards. It is essential to evaluate suppliers based on their reliability, certifications, and ability to meet specific operational needs—whether for commercial food production, brewing, or large-scale fermentation projects. Additionally, balancing cost-effectiveness with long-term durability and performance ensures a smart investment. By prioritizing functionality, scalability, and ease of maintenance, businesses can select a fermentation solution that supports efficiency, product consistency, and growth. Proper sourcing not only enhances process reliability but also contributes to the overall success and sustainability of fermentation-based operations.