The global CBD isolate market is experiencing robust growth, driven by rising consumer demand for non-psychoactive, THC-free cannabidiol products across wellness, pharmaceutical, and nutraceutical sectors. According to Grand View Research, the global CBD market was valued at USD 5.9 billion in 2022 and is expected to expand at a compound annual growth rate (CAGR) of 21.1% from 2023 to 2030. This surge is fueled by increasing legalization, expanding clinical research, and growing awareness of CBD’s therapeutic potential. As demand for high-purity, bulk CBD isolate climbs, manufacturers with scalable extraction capabilities and stringent quality certifications are emerging as key players. The following list highlights the top five bulk CBD isolate manufacturers positioned to meet this accelerating global demand with consistent, compliant, and high-quality supply.
Top 5 Bulk Cbd Isolate Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Laurelcrest
Domain Est. 2014
Website: laurelcrest.com
Key Highlights: Laurelcrest is a premier bulk CBD manufacturer in McMinnville Oregon, USA. Wholesale cannabinoid ingredients, terpenes, and finished goods….
#2 KND Labs
Domain Est. 2018
Website: kndlabs.com
Key Highlights: Get premium CBD extracts from cGMP-certified KND Labs. Your trusted CBD supplier and partner for custom blends and white label services. Order now!…
#3 GenCanna
Domain Est. 2014
Website: gencanna.com
Key Highlights: We built GenCanna to provide a scalable and sustainable supply of premium hemp derived, best-in-class cannabinoids, and non-hemp products….
#4 Red Mesa Science & Refining
Domain Est. 2019
Website: redmesascience.com
Key Highlights: ISO 9001:2015 and cGMP, Red Mesa is a globally recognized hemp processor of hemp-derived bulk CBD CBG CBN raw ingredients for sale at wholesale prices….
#5 USDA certified 100% organic CBD and CBG bulk/wholesale
Domain Est. 2019
Website: kyheritagehemp.com
Key Highlights: We are the premier provider of USDA certified 100% organic CBD and CBG extracts, distillates, and isolates….
Expert Sourcing Insights for Bulk Cbd Isolate
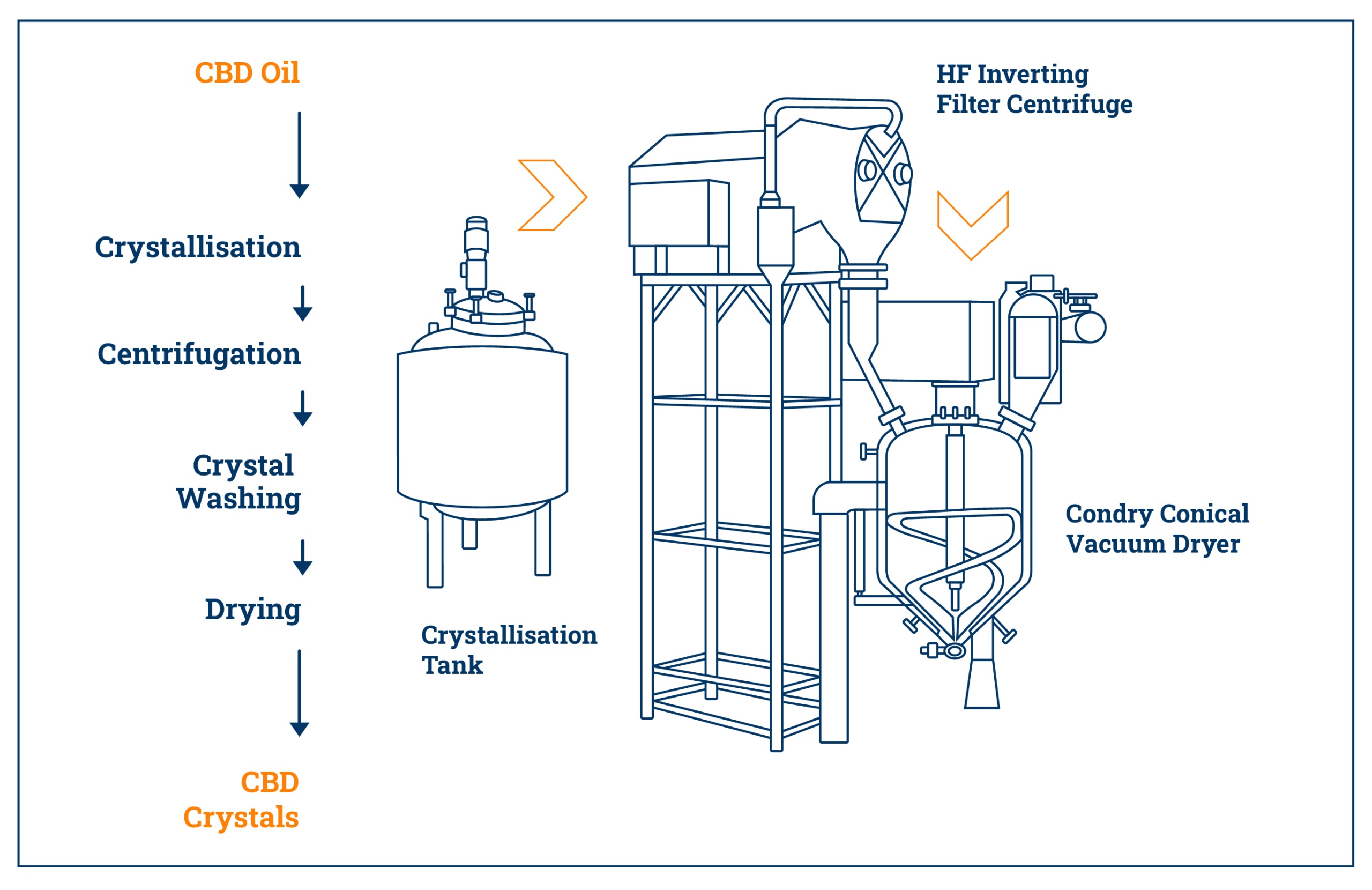
2026 Market Trends for Bulk CBD Isolate
As the global demand for cannabidiol (CBD) continues to evolve, the market for bulk CBD isolate is poised for significant transformation by 2026. Characterized by its purity and versatility, CBD isolate—containing 99%+ pure CBD with no THC or other cannabinoids—remains a preferred ingredient for manufacturers across industries. This analysis explores key market trends expected to shape the bulk CBD isolate landscape in 2026.
Increasing Demand Across Consumer Goods Sectors
By 2026, the integration of bulk CBD isolate into mainstream consumer products is expected to accelerate. Wellness brands, cosmetic companies, and functional food and beverage manufacturers are increasingly formulating products with CBD isolate due to its odorless, flavorless, and non-psychoactive properties. The isolate’s compatibility with a wide range of product types—from topical creams to gummies and energy drinks—positions it as a strategic ingredient for scalable production.
The global wellness market, projected to exceed $7 trillion by 2026, will drive much of this demand. Consumers continue to prioritize natural, plant-based solutions for stress relief, sleep support, and inflammation management—all areas where CBD isolate is gaining credibility through anecdotal and emerging clinical evidence.
Regulatory Clarity and Standardization
One of the most influential factors shaping the 2026 bulk CBD isolate market is the anticipated improvement in regulatory frameworks—particularly in the United States and European Union. By 2026, it is expected that clearer guidelines around CBD sourcing, labeling, and permissible THC thresholds will be established. This regulatory clarity will reduce market uncertainty, encourage investment, and promote consistency in product quality.
Standardization of testing protocols and certificate of analysis (COA) requirements will become more widespread, favoring suppliers who adhere to Good Manufacturing Practices (GMP) and provide transparent supply chains. As a result, large-scale buyers will increasingly prioritize traceable, lab-verified bulk CBD isolate from compliant producers.
Technological Advancements in Extraction and Purification
Innovation in extraction technologies will play a crucial role in reducing production costs and improving purity by 2026. Advanced methods such as supercritical CO2 extraction and chromatographic purification are expected to become more efficient and scalable. These improvements will lower the barrier to entry for new manufacturers and increase global supply capacity.
Additionally, automation and AI-driven quality control systems will enhance batch consistency and reduce contamination risks, further solidifying bulk CBD isolate as a reliable ingredient for pharmaceutical and nutraceutical applications.
Expansion of Global Supply Chains
The geographic footprint of bulk CBD isolate production is expected to diversify by 2026. While the U.S. remains a leading supplier due to the 2018 Farm Bill, countries such as Canada, Colombia, and several EU nations are expanding their hemp cultivation and processing capabilities. This globalization of supply will improve price competitiveness and reduce dependency on single-source regions.
Emerging markets in Southeast Asia and Africa are also beginning to explore hemp cultivation for CBD export, potentially offering lower-cost alternatives. However, adherence to international regulatory standards will be critical for these regions to gain market share.
Price Stabilization and Market Consolidation
After years of price volatility due to oversupply and inconsistent quality, the bulk CBD isolate market is expected to stabilize by 2026. Increased consolidation among producers—through mergers, acquisitions, and partnerships—will lead to fewer but stronger suppliers capable of meeting high-volume, high-compliance demands.
This consolidation, combined with improved regulation and production efficiency, is projected to result in more predictable pricing. Buyers can expect moderate cost reductions, with premium pricing reserved for pharmaceutical-grade or organically certified isolates.
Rising Interest in Pharmaceutical and Clinical Applications
By 2026, research into the therapeutic potential of CBD isolate will continue to grow, particularly in areas such as epilepsy, anxiety disorders, and chronic pain. While full-spectrum CBD has traditionally been favored for the “entourage effect,” isolate remains valuable in clinical settings where precise dosing and THC-free formulations are required.
Pharmaceutical companies are expected to increase their procurement of bulk CBD isolate for drug development and clinical trials, further legitimizing the compound and expanding its applications beyond wellness into evidence-based medicine.
Sustainability and Ethical Sourcing
Sustainability will emerge as a key differentiator in the bulk CBD isolate market. Consumers and B2B buyers alike are placing greater emphasis on environmentally friendly farming practices, minimal processing waste, and ethical labor standards. Suppliers that adopt regenerative agriculture, carbon-neutral processing, and third-party sustainability certifications will gain a competitive edge.
By 2026, eco-labeling and transparency in sourcing are likely to become standard expectations, influencing purchasing decisions across the supply chain.
Conclusion
The 2026 outlook for bulk CBD isolate is one of maturation, driven by regulatory progress, technological innovation, and expanding applications. As the market moves beyond the early adopter phase, stakeholders who prioritize quality, compliance, and sustainability will lead the industry. With growing acceptance and integration into global health and wellness ecosystems, bulk CBD isolate is set to remain a cornerstone ingredient in the cannabinoid economy.

Common Pitfalls Sourcing Bulk CBD Isolate (Quality, IP)
Sourcing bulk CBD isolate can be a cost-effective way to develop CBD-infused products, but it comes with significant risks—particularly concerning quality and intellectual property (IP). Avoiding these common pitfalls is crucial for regulatory compliance, product safety, and brand protection.
Poor or Inconsistent Product Quality
One of the biggest challenges in sourcing bulk CBD isolate is ensuring consistent, high-quality material. Many suppliers offer products that vary widely in purity, potency, and safety.
- Inadequate Third-Party Testing: Reputable suppliers provide up-to-date Certificates of Analysis (CoAs) from independent labs. Be wary of suppliers who offer only in-house testing or outdated CoAs, as this increases the risk of receiving contaminated or mislabeled products.
- Residual Solvents and Contaminants: Low-quality extraction processes can leave behind harmful residues such as butane, ethanol, or heavy metals. Always verify that the isolate is tested for residual solvents, pesticides, and microbial contamination.
- Inaccurate Potency Claims: Some isolates are diluted or spiked, leading to inconsistent CBD content. Without reliable CoAs, you risk formulating products with incorrect dosages, which can undermine efficacy and compliance.
- Degraded or Unstable Isolate: Poor storage or handling can lead to degradation of CBD into other compounds (like CBN), reducing potency and altering effects. Ensure the supplier follows strict storage and shipping protocols.
Lack of Supply Chain Transparency and Traceability
Opaque supply chains increase the risk of sourcing isolate derived from non-compliant or unethical sources.
- Unclear Hemp Origin: CBD derived from hemp must contain less than 0.3% THC on a dry weight basis to comply with U.S. federal law (per the 2018 Farm Bill). Sourcing from suppliers without traceable farming practices risks receiving non-compliant material.
- No Farm-to-Isolate Documentation: Reliable suppliers offer full traceability—from seed to final product. Without it, you cannot verify claims about organic cultivation, pesticide-free farming, or legal compliance.
Intellectual Property Risks
Using bulk CBD isolate in product development can expose companies to IP infringement, especially in the U.S., where patent landscapes are complex.
- Infringement of Existing Patents: Some CBD-related formulations, delivery methods, or extraction processes are patented. Simply using generic CBD isolate in a novel product may still infringe on method-of-use or composition patents held by competitors.
- Lack of Freedom-to-Operate (FTO) Analysis: Companies often overlook conducting an FTO search before launching products. This can lead to costly litigation or forced product reformulation.
- Supplier IP Claims: Some suppliers may claim ownership over specific formulations or processes tied to their isolate. Ensure your supply agreement clearly defines IP rights and doesn’t restrict your ability to innovate or scale.
Unreliable Supply and Scalability Issues
Many bulk suppliers cannot consistently meet volume demands, especially for long-term contracts.
- Batch-to-Batch Variability: Even with quality testing, some suppliers lack the infrastructure to maintain consistency across large batches, leading to formulation challenges.
- Supply Chain Disruptions: Dependence on a single supplier with limited production capacity can jeopardize product timelines. Always consider dual sourcing or verifying the supplier’s production scalability.
Regulatory and Compliance Gaps
CBD regulations vary by region and are subject to change. Sourcing isolate without compliance foresight can result in product recalls or legal penalties.
- Non-GRAS or Unapproved Ingredients: In certain markets (like the U.S. FDA-regulated food/beverage space), CBD is not generally recognized as safe (GRAS) for human consumption. Using isolate in ingestible products could invite regulatory scrutiny.
- Labeling and Marketing Restrictions: Even with compliant isolate, improper claims (e.g., unsubstantiated health benefits) can trigger enforcement actions. Ensure your sourcing partner supports compliant labeling practices.
Conclusion
To mitigate these pitfalls, conduct thorough due diligence on potential suppliers—demand comprehensive CoAs, verify supply chain transparency, assess scalability, and consult legal counsel on IP and regulatory compliance. Choosing the right bulk CBD isolate partner is not just about price, but about ensuring long-term product integrity and market viability.

Logistics & Compliance Guide for Bulk CBD Isolate
Overview of Bulk CBD Isolate
CBD isolate is a highly purified form of cannabidiol (CBD), typically appearing as a white crystalline powder containing 99%+ pure CBD. When procured or distributed in bulk (usually 1 kg or more), it is commonly used for manufacturing tinctures, edibles, topicals, and other CBD-infused products. Due to its potency and regulatory scrutiny, proper logistics and compliance are critical.
Legal Compliance Requirements
Bulk CBD isolate is derived from hemp, and under the U.S. 2018 Farm Bill, it is federally legal if derived from hemp containing no more than 0.3% delta-9 THC on a dry weight basis. However, compliance extends beyond federal law:
– State Laws: Some states impose additional restrictions on CBD sales, usage, or manufacturing. Always verify state-specific regulations where the product will be stored, distributed, or sold.
– FDA Regulations: The FDA does not currently approve CBD as a dietary supplement or food additive, and making unapproved health claims is prohibited.
– Import/Export Controls: International shipments require compliance with both U.S. and destination country regulations. Many countries classify CBD as a controlled substance.
– DEA & Controlled Substances Act: While hemp-derived CBD is not scheduled, ensure THC levels remain below the legal threshold to avoid classification as marijuana.
Sourcing & Supplier Verification
To ensure compliance and product integrity:
– Confirm suppliers are licensed hemp producers compliant with the U.S. Farm Bill.
– Request up-to-date Certificates of Analysis (CoAs) from third-party labs verifying cannabinoid profile, potency, and absence of contaminants (e.g., heavy metals, pesticides, residual solvents).
– Verify that the isolate is derived from hemp grown in accordance with state-authorized agricultural programs (e.g., USDA Hemp Program).
Packaging & Labeling Standards
Proper packaging and labeling prevent legal issues and ensure safety:
– Use tamper-evident, airtight, light-resistant containers to preserve stability.
– Label bulk containers with:
– Product name (e.g., “99% Pure CBD Isolate”)
– Net weight
– Batch number
– Manufacturer name and contact information
– THC content (<0.3%)
– “Hemp-Derived CBD” disclaimer
– Storage instructions
– Avoid any therapeutic claims on packaging to comply with FDA guidelines.
Storage & Handling Protocols
CBD isolate is sensitive to heat, light, and moisture:
– Store in a cool, dry, dark environment (15–25°C or 59–77°F recommended).
– Maintain low humidity to prevent clumping or degradation.
– Use dedicated, secure storage areas with restricted access to prevent contamination or theft.
– Implement a first-in, first-out (FIFO) inventory system to manage shelf life (typically 12–24 months when properly stored).
Transportation & Logistics
- Use temperature-controlled shipping when possible, especially for long-distance or international transport.
- Partner with carriers experienced in handling regulated botanical products.
- Maintain a clear chain of custody with documentation (e.g., bills of lading, CoAs, manifests) for traceability.
- For cross-border shipments, ensure all customs documentation includes accurate HS codes (e.g., 1302.19 for plant extracts) and complies with import regulations.
Recordkeeping & Traceability
Maintain detailed records for compliance audits and recalls:
– Batch-specific CoAs and supplier invoices.
– Inventory logs tracking receipt, storage, and distribution.
– Shipping and delivery records.
– Internal testing results, if applicable.
Records should be retained for a minimum of two years, or as required by state law.
Quality Assurance & Testing
- Test incoming bulk isolate upon receipt to verify CoA accuracy.
- Conduct periodic stability testing to ensure product integrity over time.
- Use ISO 17025-accredited laboratories for all compliance testing.
Worker Safety & Training
- Train staff on safe handling procedures, including use of PPE (gloves, masks) to prevent contamination.
- Educate employees on regulatory requirements and company compliance policies.
- Establish protocols for spill response and proper disposal.
Conclusion
Successfully managing bulk CBD isolate requires strict adherence to legal, logistical, and quality standards. Proactive compliance, transparent sourcing, and meticulous documentation are essential to minimizing risk and ensuring smooth operations in the evolving CBD marketplace. Always consult legal counsel or regulatory experts to stay current with changing laws.
In conclusion, sourcing bulk CBD isolate requires careful consideration of multiple factors to ensure quality, compliance, and long-term value. It is essential to partner with reputable suppliers who provide transparent sourcing practices, up-to-date Certificates of Analysis (CoAs), and adherence to industry standards such as Good Manufacturing Practices (GMP). Verifying the purity of the CBD isolate—typically 99%+ pure cannabidiol—is crucial for formulating safe and effective products. Additionally, understanding federal and state regulations, particularly around THC content and hemp sourcing under the 2018 Farm Bill, helps mitigate legal risks. Price should not be the sole deciding factor; reliability, scalability, and customer support also play vital roles in sustaining a successful supply chain. By conducting thorough due diligence and building strong relationships with trusted vendors, businesses can secure high-quality bulk CBD isolate that supports product integrity and consumer trust in the competitive wellness market.







