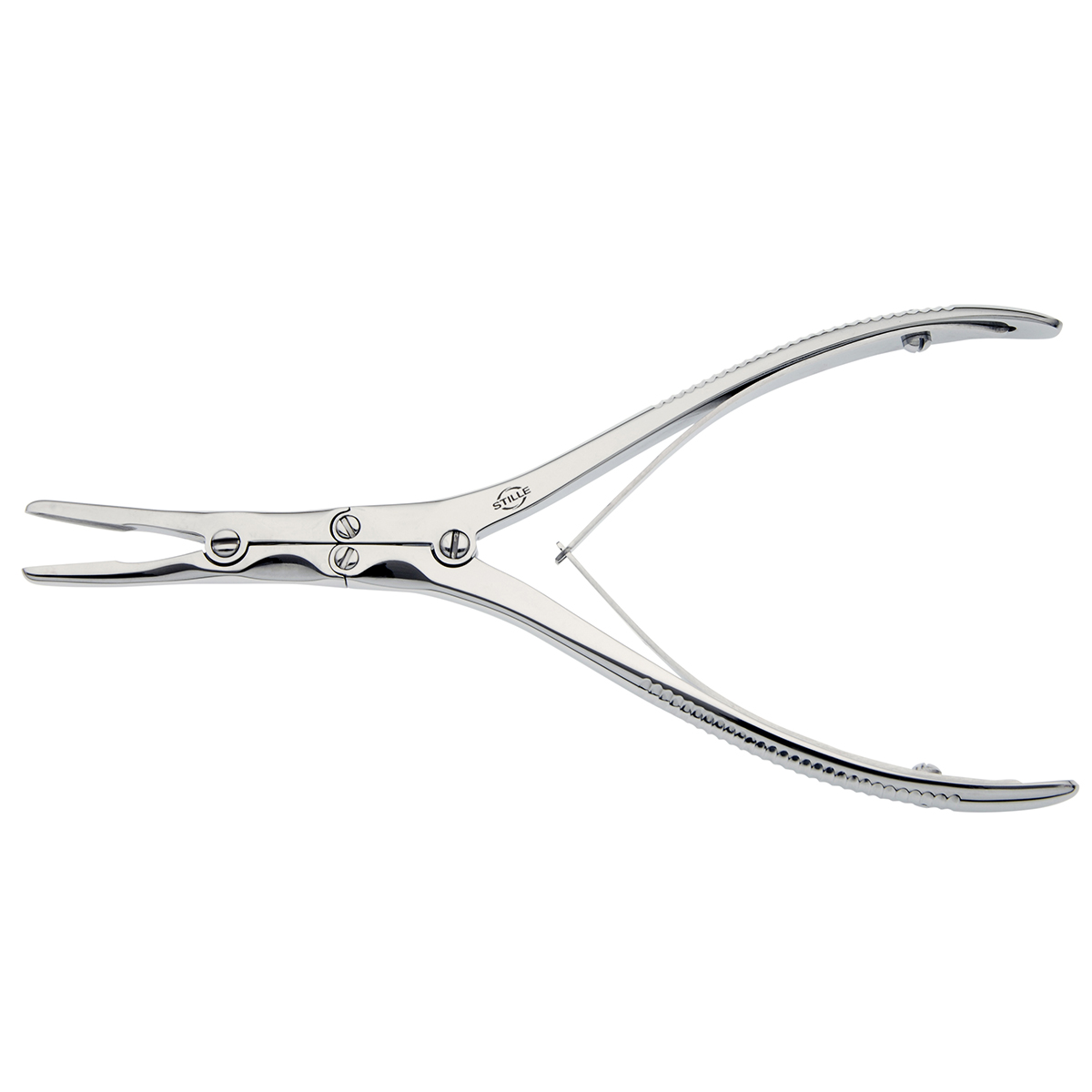
The global surgical instruments market, driven by rising orthopedic and neurosurgical procedures, is witnessing sustained growth, with the segment for specialized tools like bone rongeurs expanding in tandem. According to Grand View Research, the global surgical instruments market size was valued at USD 19.3 billion in 2022 and is expected to grow at a compound annual growth rate (CAGR) of 7.8% from 2023 to 2030. This growth is fueled by increasing demand for minimally invasive surgeries, an aging population predisposed to spinal and joint disorders, and technological advancements in precision surgical tools. Bone rongeurs—critical for cutting, shaping, and removing bone in neurosurgery, orthopedics, and spinal procedures—have become indispensable in modern operating rooms. As demand intensifies, manufacturers are investing in ergonomic design, enhanced durability, and single-use variants to meet stringent clinical requirements. In this evolving landscape, a select group of manufacturers has emerged as leaders, combining innovation, regulatory compliance, and global distribution to capture significant market share. Here’s a data-driven look at the top seven bone rongeur manufacturers shaping the future of surgical instrumentation.
Top 7 Bone Rongeur Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
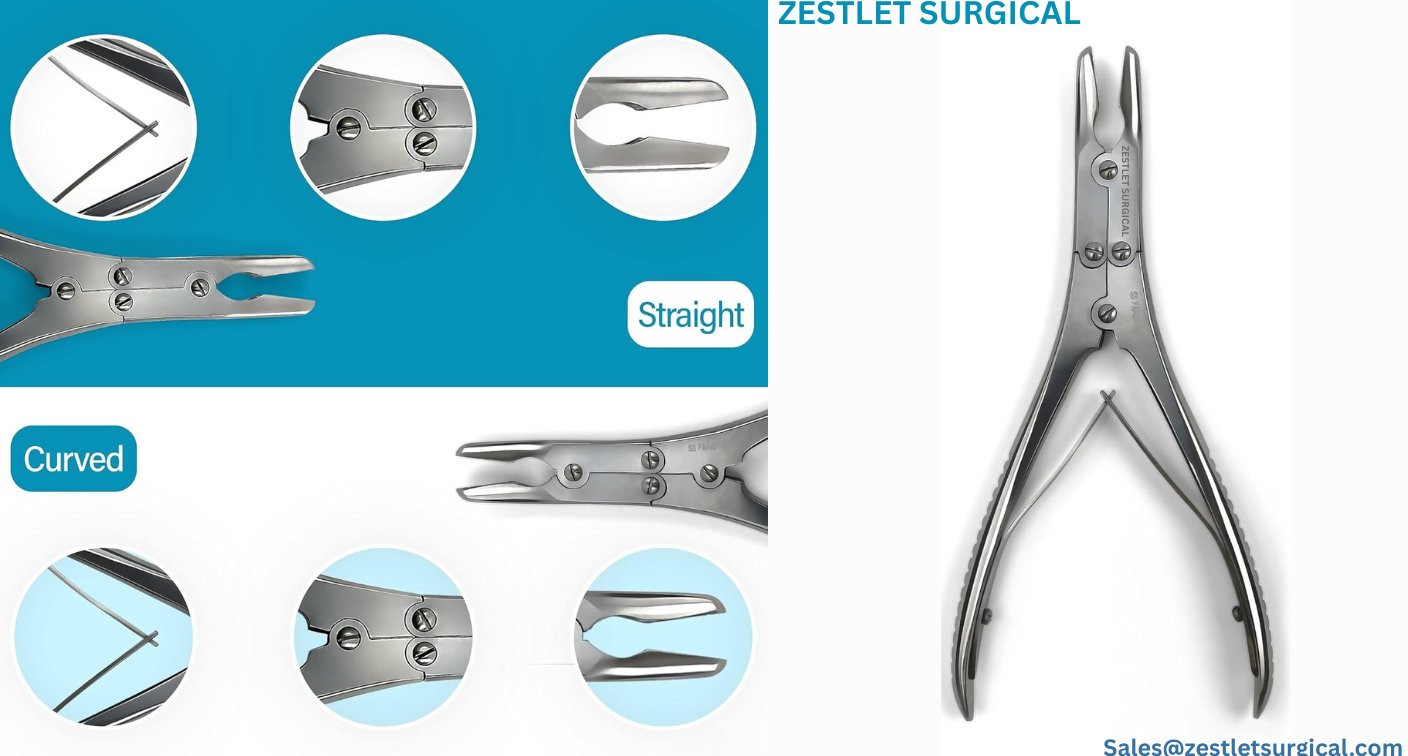
#1 Bone Rongeur – Small
Domain Est. 2004
Website: germedusa.com
Key Highlights: Free deliveryWe offer Bone Rongeur – Small used to manipulate bones in orthopedic procedures. It features jaws and a plier design handle. Available in various sizes….
#2 Bone Cutters
Domain Est. 1995
#3 BONE RONGEURS
Domain Est. 1996
Website: dental-instruments.bbraun.com
Key Highlights: Rongeurs ; Length (Inch) · 7″ 7 7/8″ 6″ 9″ ; Length · 180.00 mm 200.00 mm 150.00 mm 230.00 mm ; Author · CASPAR LOVE-GRUENWALD SPURLING CUSHING…
#4 Rongeurs Archives
Domain Est. 1997
Website: nordent.com
Key Highlights: Rongeurs. Used for contouring and removing alveolar bone during extraction procedures. Our rongeurs are hardened and tempered to maintain sharpness and have ……
#5 Bone Instruments
Domain Est. 1997
Website: roboz.com
Key Highlights: We offer a wide variety of bone instruments including rongeurs, bone cutting forceps, periosteal elevators, chisels, saws, trephines, and much more….
#6 Bone Rongeurs
Domain Est. 1998
Website: atitan.com
Key Highlights: $20 delivery Free 30-day returnsBlumenthal, Friedman, Lempert, and Kerrison Rongeurs are made in Germany from 100% stainless steel. Used to cut bone, Rongeurs should never be used …
#7 Durable Bone Rongeurs for Orthopedic Surgical Procedures
Domain Est. 2001
Website: gsource.com
Key Highlights: $18 delivery 90-day returnsgSource bone rongeurs are crafted from high-quality German surgical stainless steel and have hollowed, cup-like, sharp working ends similar to a curette….
Expert Sourcing Insights for Bone Rongeur
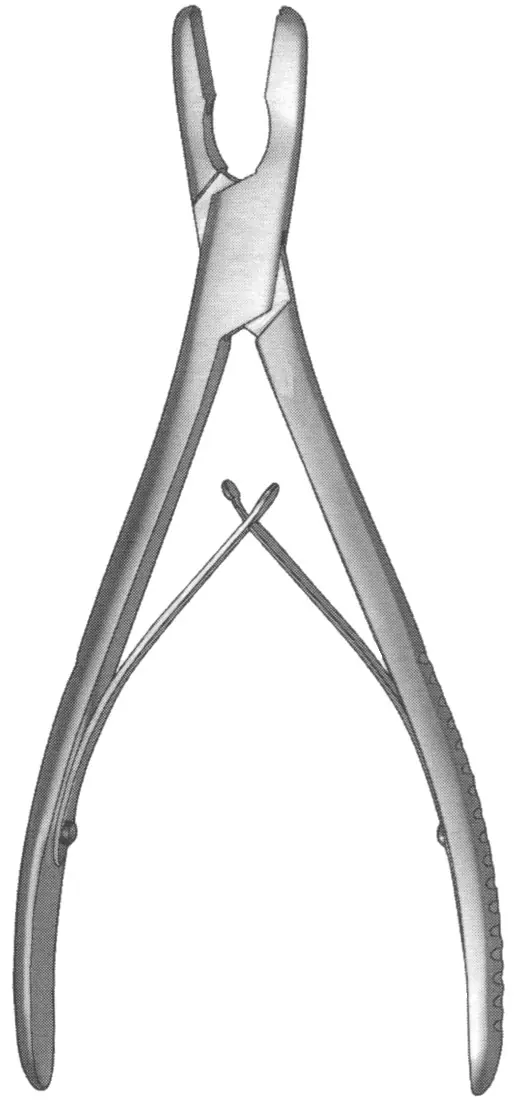
2026 Market Trends for Bone Rongeur
The global bone rongeur market is poised for significant evolution by 2026, driven by advancements in surgical technologies, rising prevalence of orthopedic and neurosurgical conditions, and increasing demand for minimally invasive procedures. Bone rongeurs—precision surgical instruments used primarily in orthopedic, spinal, and cranial surgeries to remove or shape bone—are witnessing enhanced innovation and expanded clinical applications. This analysis explores key market trends expected to shape the bone rongeur landscape in 2026.
Rising Incidence of Musculoskeletal Disorders
A primary driver of the bone rongeur market is the growing global burden of musculoskeletal disorders, including osteoarthritis, spinal deformities, and traumatic fractures. Aging populations, particularly in North America, Europe, and parts of Asia-Pacific, are contributing to higher rates of joint degeneration and spinal pathologies. By 2026, the demand for joint replacement and spinal fusion surgeries—common procedures requiring bone rongeurs—is projected to rise significantly, directly increasing instrument utilization.
Shift Toward Minimally Invasive Surgery (MIS)
Minimally invasive surgical techniques continue to gain traction across orthopedic and neurosurgical fields. These procedures require specialized, high-precision instruments, including ergonomically designed and miniaturized bone rongeurs. By 2026, manufacturers are expected to focus on developing MIS-compatible rongeurs with enhanced articulation, improved visibility, and reduced tissue trauma. This trend is supported by patient preference for faster recovery and shorter hospital stays, pushing hospitals and surgical centers to upgrade instrument inventories.
Technological Innovation and Product Differentiation
Innovation is a key competitive differentiator in the bone rongeur market. Companies are investing in advanced materials (such as high-grade stainless steel and titanium alloys), improved mechanical leverage systems, and disposable or single-use rongeurs to reduce infection risks. Additionally, integration with navigation systems and robotic-assisted surgery platforms is emerging. By 2026, smart rongeurs equipped with force feedback or compatibility with augmented reality (AR)-guided surgery may begin entering niche clinical settings, especially in neurosurgery and complex spinal interventions.
Expansion in Emerging Markets
Emerging economies in Asia-Pacific, Latin America, and Africa are experiencing rapid growth in healthcare infrastructure and surgical capabilities. Increased government spending, rising medical tourism, and expanding insurance coverage are improving access to advanced surgical care. As a result, the demand for surgical instruments like bone rongeurs is expected to grow robustly in these regions by 2026. Local manufacturing and partnerships with global players may further accelerate market penetration.
Focus on Infection Control and Reusable Instrument Challenges
With increasing scrutiny on hospital-acquired infections (HAIs), the reprocessing of reusable surgical instruments like traditional rongeurs is under review. Challenges related to cleaning complex geometries and ensuring sterility are prompting interest in single-use or pre-sterilized disposable bone rongeurs. By 2026, this segment is anticipated to grow, especially in ambulatory surgical centers (ASCs) and low-resource settings where sterilization infrastructure may be limited.
Competitive Landscape and Strategic Collaborations
The bone rongeur market is moderately consolidated, with key players including Aesculap (B. Braun), Medtronic, Stryker, DePuy Synthes (Johnson & Johnson), and KLS Martin. These companies are expected to strengthen their portfolios through product launches, mergers, and collaborations with surgical robotics firms. By 2026, strategic alliances aimed at integrating rongeurs into comprehensive surgical ecosystems will likely define market leadership.
Regulatory and Reimbursement Dynamics
Regulatory approvals and reimbursement policies will continue to influence market adoption. In regions like the U.S. and EU, stringent FDA and CE marking requirements ensure high instrument quality, while favorable reimbursement for orthopedic procedures supports device utilization. However, pricing pressures and value-based care models may push manufacturers to demonstrate cost-effectiveness and clinical outcomes linked to their instruments.
Conclusion
By 2026, the bone rongeur market will be shaped by demographic shifts, technological innovation, and evolving surgical practices. Growth will be fueled by increasing surgical volumes, the rise of MIS, and expansion in emerging markets. Success for manufacturers will depend on their ability to innovate, ensure compliance, and adapt to changing clinical and economic demands in global healthcare systems.
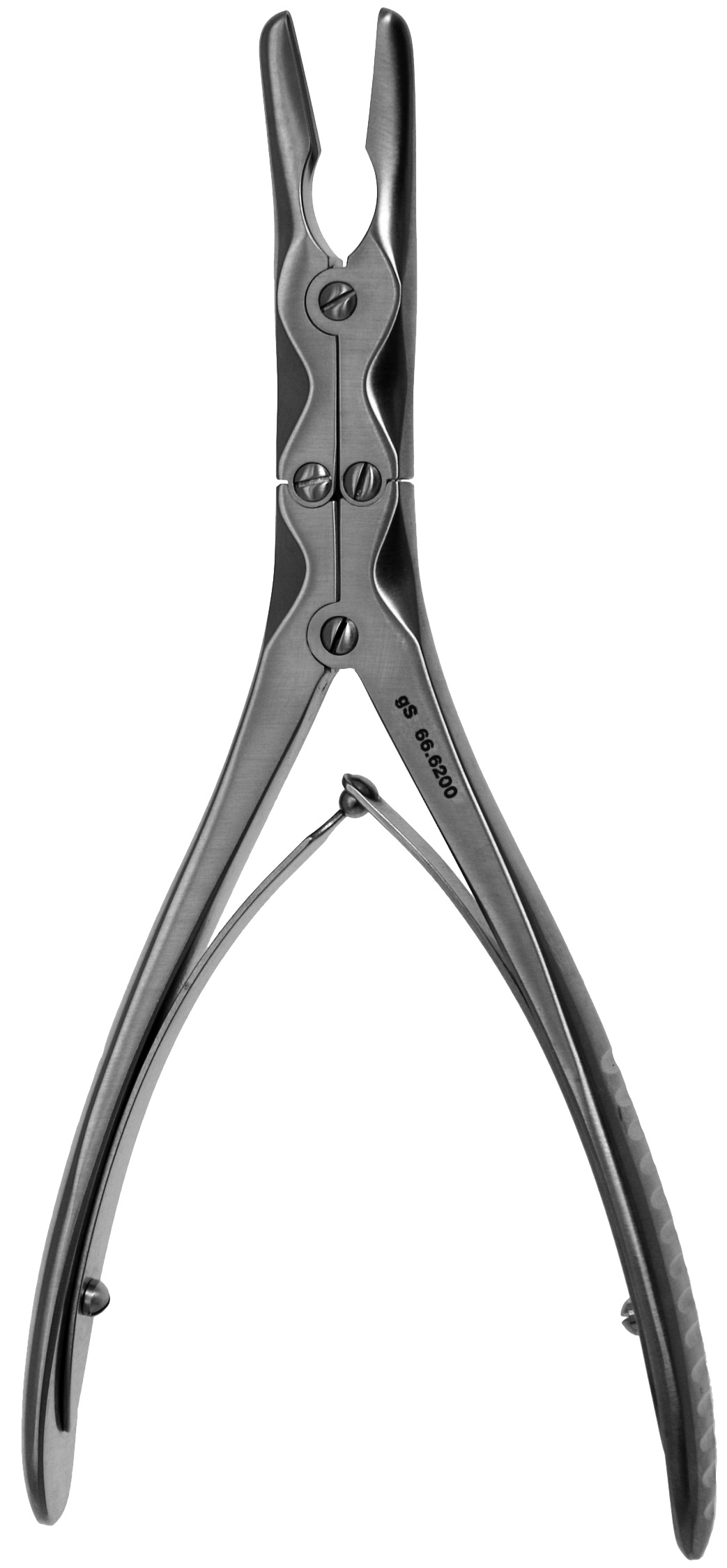
Common Pitfalls When Sourcing Bone Rongeurs (Quality and Intellectual Property)
Sourcing high-quality bone rongeurs—essential surgical instruments used in orthopedic and neurosurgical procedures—requires careful due diligence. Overlooking key factors can lead to compromised surgical outcomes, regulatory issues, and legal risks. Below are critical pitfalls related to quality and intellectual property (IP) that procurement teams and medical device companies should avoid.
Inadequate Quality Assurance and Manufacturing Standards
One of the most significant risks in sourcing bone rongeurs is partnering with manufacturers that do not adhere to stringent quality control standards. Low-cost suppliers, especially in regions with lax regulatory oversight, may produce instruments that fail under surgical stress due to poor material selection or substandard heat treatment.
- Use of Inferior Stainless Steel: Some manufacturers use non-medical-grade stainless steel that corrodes or dulls quickly, increasing the risk of surgical complications.
- Inconsistent Heat Treatment: Improper hardening reduces cutting edge durability, leading to frequent instrument replacement and potential intraoperative failure.
- Lack of ISO 13485 Certification: Reputable suppliers should be certified to ISO 13485, the international standard for medical device quality management systems. Sourcing from non-certified vendors increases the risk of non-compliant products.
Poor Ergonomic Design and Functional Deficiencies
Bone rongeurs must offer precise control and consistent performance. Offshore or generic suppliers may replicate the appearance of premium instruments without replicating their functionality.
- Misaligned Jaws or Loose Hinges: Poor assembly can result in imprecise cutting, tissue damage, or instrument jamming during use.
- Suboptimal Spring Mechanism: Weak or fatigued springs reduce usability and surgeon comfort, especially during prolonged procedures.
- Lack of Prototyping and Surgeon Feedback: High-quality rongeurs are typically developed with input from surgeons. Suppliers that skip this step often produce ergonomically flawed tools.
Intellectual Property (IP) Infringement Risks
Sourcing from third-party manufacturers without proper IP clearance can expose your organization to legal challenges, especially when replicating branded instruments like those from Aesculap, KLS Martin, or Stryker.
- Counterfeit or “Look-Alike” Instruments: Some suppliers offer rongeurs that closely mimic patented designs. Using these can result in cease-and-desist orders, product recalls, or costly litigation.
- Lack of Design Freedom: Without thorough IP due diligence (e.g., freedom-to-operate analysis), your sourced product may infringe on active utility or design patents.
- Unverified Supplier Claims: Suppliers may falsely claim their designs are IP-safe. Always conduct independent patent landscaping or consult IP counsel before large-scale procurement.
Inconsistent Regulatory Compliance
Bone rongeurs are regulated medical devices in most markets (e.g., FDA in the U.S., CE marking in Europe). Sourcing instruments without proper documentation can delay market entry or result in non-compliance penalties.
- Missing Technical Documentation: Lack of device master records, biocompatibility reports, or sterilization validation can prevent regulatory approval.
- Unregistered Manufacturing Facilities: Ensure the supplier’s facility is registered with relevant authorities (e.g., FDA Establishment Registration).
- Labeling and UDI Non-Compliance: Improper labeling or absence of Unique Device Identification (UDI) can lead to distribution bans.
Supply Chain and Traceability Gaps
Reliable sourcing requires full traceability—from raw materials to finished devices. Poor traceability increases recall risks and complicates quality investigations.
- Undocumented Material Sourcing: Inability to trace stainless steel batches makes it difficult to respond to corrosion or contamination issues.
- Lack of Serialisation: High-risk surgical instruments should be individually serialized for tracking; many low-tier suppliers do not provide this.
Mitigation Strategies
To avoid these pitfalls:
– Conduct on-site audits of supplier facilities.
– Require full compliance documentation (ISO 13485, FDA registration, CE certificates).
– Perform independent testing of samples for material quality and performance.
– Engage IP counsel to review designs before finalizing sourcing agreements.
– Establish long-term contracts with clear quality and IP indemnification clauses.
By proactively addressing quality and intellectual property concerns, organizations can ensure the safe, legal, and effective sourcing of bone rongeurs.
Logistics & Compliance Guide for Bone Rongeur
Overview
A Bone Rongeur is a surgical instrument used primarily in orthopedic, neurosurgical, and podiatric procedures to cut and shape bone. Due to its classification as a medical device, shipping, handling, and regulatory compliance require strict adherence to international and local standards. This guide outlines key logistics and compliance considerations for Bone Rongeurs throughout the supply chain.
Regulatory Classification
Bone Rongeurs are typically classified as Class I or Class II medical devices under regulatory frameworks such as the U.S. FDA 510(k) pathway or the EU Medical Device Regulation (MDR). Classification affects labeling, registration, and post-market surveillance requirements. Confirm the specific classification based on design, intended use, and jurisdiction.
Regulatory Approvals and Certifications
Ensure the Bone Rongeur has the necessary regulatory approvals prior to distribution:
– U.S. FDA Clearance: Verify 510(k) clearance or registration with the FDA.
– EU CE Marking: Confirm compliance with EU MDR (Regulation (EU) 2017/745) and affix CE marking.
– Other Markets: Obtain certifications such as Health Canada license, TGA (Australia), or NMPA (China) as applicable.
– Maintain valid ISO 13485 certification for quality management systems.
Labeling Requirements
Labels must comply with regional regulations and include:
– Device name and model number
– Manufacturer name and address
– CE mark (if applicable) or FDA establishment number
– UDI (Unique Device Identifier) in compliance with FDA and EU MDR
– Sterility status (sterile or non-sterile)
– Single-use or reusable designation
– Expiry date (if applicable)
– IFU (Instructions for Use) reference
Packaging and Sterilization
- Use sterile barrier systems compliant with ISO 11607 for sterile devices.
- Confirm validated sterilization method (e.g., ethylene oxide, gamma radiation).
- Packaging must protect against contamination, moisture, and physical damage during transit.
- Include humidity and temperature indicators if required.
Shipping and Handling
- Ship in temperature-controlled environments if sterilization or material integrity is sensitive.
- Use tamper-evident packaging.
- Label packages as “Medical Device” and include handling instructions (e.g., “Fragile,” “Do Not Freeze”).
- Maintain chain of custody documentation for traceability.
Import and Export Compliance
- Prepare accurate Harmonized System (HS) code (typically 9018.90 for surgical instruments).
- Provide commercial invoice, packing list, and certificate of origin.
- Include FDA prior notice for U.S. imports and EU Importer Declaration under MDR.
- Comply with customs requirements in destination country; consider engaging a licensed customs broker.
Storage Conditions
- Store in a clean, dry, temperature-controlled environment (typically 15–30°C, 35–70% RH).
- Avoid direct sunlight and chemical exposure.
- Segregate sterile and non-sterile devices.
- Follow first-in, first-out (FIFO) inventory practices.
Post-Market Surveillance and Traceability
- Implement a robust UDI system for tracking devices through distribution and use.
- Monitor and report adverse events per FDA MedWatch or EU Vigilance reporting requirements.
- Maintain distribution records for the device’s lifecycle (minimum 5–10 years depending on jurisdiction).
Reusable Device Considerations
If the Bone Rongeur is reusable:
– Provide validated reprocessing instructions (cleaning, disinfection, sterilization).
– Ensure compliance with AAMI ST79 and other relevant standards.
– Train end-users on proper handling and maintenance.
Disposal and Environmental Compliance
- Follow biohazard or medical waste regulations for contaminated devices.
- Comply with local, national, and international environmental directives (e.g., WEEE, RoHS).
- Offer take-back or recycling programs if applicable.
Conclusion
Proper logistics and regulatory compliance are essential to ensure the safe and legal distribution of Bone Rongeurs. Stakeholders must remain vigilant about evolving regulations, maintain accurate documentation, and partner with certified suppliers and distributors to uphold quality and patient safety.
Conclusion for Sourcing Bone Rongeur:
In conclusion, sourcing a high-quality bone rongeur requires careful evaluation of several key factors, including material composition (preferably surgical-grade stainless steel), precision engineering for durability and sharpness, compliance with international medical device standards (such as ISO 13485 and FDA regulations), and reliable sterilization capabilities. It is essential to partner with reputable suppliers or manufacturers—whether domestic or international—who maintain consistent quality control and offer comprehensive documentation and traceability. Additionally, considerations such as ergonomic design, intended surgical application (e.g., neurosurgery, orthopedics, or ENT), and long-term cost-efficiency through instrument longevity and reusability should guide the procurement decision. Ultimately, investing in a dependable bone rongeur from a trusted source enhances surgical outcomes, ensures patient safety, and supports the efficiency of medical procedures.