The global blister packaging market for medications is experiencing robust growth, driven by increasing demand for tamper-evident, patient-compliant, and shelf-stable pharmaceutical packaging solutions. According to Grand View Research, the global pharmaceutical packaging market size was valued at USD 104.8 billion in 2022 and is expected to expand at a compound annual growth rate (CAGR) of 8.7% from 2023 to 2030. Blister packaging, in particular, dominates the segment due to its effectiveness in protecting dosage forms, enhancing product shelf life, and supporting unit-dose compliance—critical factors in both prescription and over-the-counter medication distribution. Mordor Intelligence further projects that the global blister packaging market will grow at a CAGR of over 5.5% during the forecast period of 2023–2028, bolstered by advancements in materials, rising geriatric populations, and expanding pharmaceutical production in emerging markets. As demand intensifies, a select group of manufacturers have emerged as leaders, combining innovation, scalability, and regulatory compliance to meet the evolving needs of the pharmaceutical industry. Here are the top 10 blister packaging medication manufacturers shaping the future of drug delivery and safety.
Top 10 Blister Packaging Medication Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Klöckner Pentaplast
Domain Est. 1999
Website: kpfilms.com
Key Highlights: Global manufacturer of sustainable and recyclable plastic films and high-barrier protective packaging for Food, Pharmaceutical, Health and Durable products….
#2 Oral Drug Blister Packaging
Domain Est. 2000
Website: amcor.com
Key Highlights: At Amcor we specialise in oral drug packaging. Our experts work with pharma customers to develop safe and blister strip and sachet solutions for oral drugs.Missing: medication man…
#3 Pharmaworks: Blister Machines & Cartoners
Domain Est. 2004
Website: pharmaworks.com
Key Highlights: Pharmaworks is the USA’s leading pharmaceutical blister machine manufacturer. Innovation, flexibility, and dependability are at the core of all our blister ……
#4 Medication Blister Pack Manufacturer & Supplier
Domain Est. 2009
Website: pharmapackagingsolutions.com
Key Highlights: Tjoapack US is a US based blister packaging supplier and manufacturer serving clients from design to delivery….
#5 Central Pharmacy Blister Packager
Domain Est. 1994
Website: omnicell.com
Key Highlights: The Central Pharmacy Blister Packager is a flexible solution that supports virtually all oral solid sizes and types. It delivers a throughput of up to 100 ……
#6 Contract Packaging Solutions
Domain Est. 1999
Website: sonicpackaging.com
Key Highlights: Sonic Packaging delivers custom packaging solutions and contract packaging services including blisters, pouches, bottles, and complex applicator systems….
#7 Sepha: Non-destructive Leak Testing
Domain Est. 1999
Website: sepha.com
Key Highlights: Our range of products enable the pharmaceutical and medical device industries to accurately detect leaks in packaging, recover high-value products from rejected ……
#8 Primary Blister Packaging
Domain Est. 2000
Website: sharpservices.com
Key Highlights: Sharp has been a leader in blister packaging services for pharmaceutical products for over 65 years. We have the flexibility to meet your drug requirements….
#9 Pharmaceutical Packaging Blister Films
Domain Est. 2007
Website: perlenpackaging.com
Key Highlights: We are the only systems provider of packaging films with a clearly defined pharmaceutical profile. Our processes, facilities, products and environment are ……
#10 Specialized Multi
Domain Est. 2019
Website: joneshealthcaregroup.com
Key Highlights: Our blister packaging services for medications, including tablets, capsules, hard gels, and soft gels are equipped with a two-lane Uhlmann blister line….
Expert Sourcing Insights for Blister Packaging Medication
2026 Market Trends for Blister Packaging in Medication
The blister packaging market for medication is poised for significant transformation by 2026, influenced by technological advancements, regulatory shifts, sustainability demands, and evolving consumer expectations. As pharmaceutical manufacturers prioritize product safety, patient compliance, and environmental responsibility, blister packaging continues to adapt. Below are the key trends expected to shape the market in 2026.
Rising Demand for Patient-Centric Packaging
By 2026, patient-centric design will be a dominant force in blister packaging. With an aging global population and increasing prevalence of chronic diseases, there is growing demand for packaging that enhances medication adherence. Features such as easy-open tabs, braille labeling, color-coded strips, and integrated reminders (e.g., QR codes linking to dosage instructions) are becoming standard. Smart blister packs with embedded sensors or NFC tags that track usage and send alerts via mobile apps are expected to see wider adoption, especially in markets with advanced digital health infrastructure.
Growth in Sustainable and Eco-Friendly Materials
Environmental concerns are driving a shift toward sustainable blister packaging. By 2026, regulatory pressure and consumer demand will push pharmaceutical companies to replace traditional PVC/PE/PVDC blister materials with recyclable or biodegradable alternatives. Materials like mono-material thermoformable films (e.g., PP or PET), paper-based blister solutions, and compostable polymers are gaining traction. Major packaging suppliers are investing in R&D to create barrier properties equivalent to conventional materials without the environmental cost. Regulatory bodies in the EU and North America are expected to enforce stricter packaging sustainability mandates, accelerating this trend.
Advancements in Child-Resistant and Senior-Friendly Designs
Balancing child resistance with senior accessibility remains a critical challenge. By 2026, innovative designs that meet both safety and usability requirements will dominate the market. Push-through-and-turn or peel-push mechanisms that are easy for arthritic hands yet secure against children are being widely adopted. Regulatory standards such as ISO 8317 and ASTM D3475 will continue to influence packaging innovations, with manufacturers increasingly integrating dual functionality into single-unit blister cards.
Expansion in Emerging Markets
The blister packaging market will see robust growth in emerging economies, particularly in Asia-Pacific, Latin America, and Africa. Rising healthcare access, government initiatives for generic drug distribution, and increased local pharmaceutical production are fueling demand. Blister packaging is favored for its ability to maintain drug integrity in regions with variable climate conditions. By 2026, multinational packaging companies are expected to establish regional manufacturing hubs to meet localized needs and reduce logistics costs.
Technological Integration and Industry 4.0
The pharmaceutical packaging industry is embracing digital transformation. By 2026, smart manufacturing technologies—such as AI-driven quality control, real-time serialization, and blockchain for traceability—will be integrated into blister packaging lines. Serialization and anti-counterfeiting features (e.g., unique identifiers, tamper-evident seals) will become mandatory in more markets, driven by regulations like the EU Falsified Medicines Directive (FMD) and FDA DSCSA. These technologies enhance supply chain transparency and consumer trust.
Consolidation and Strategic Partnerships
The blister packaging sector is expected to see increased consolidation as pharmaceutical firms seek end-to-end packaging solutions. Strategic partnerships between pharma companies, packaging manufacturers, and tech providers will accelerate innovation. Outsourcing to contract packaging organizations (CPOs) with specialized capabilities in sustainable and smart packaging is likely to rise, allowing pharma companies to focus on core R&D while ensuring compliance and scalability.
Conclusion
By 2026, the medication blister packaging market will be defined by innovation, sustainability, and patient empowerment. As regulatory landscapes evolve and environmental accountability grows, manufacturers who invest in smart, eco-conscious, and user-friendly solutions will lead the market. The convergence of digital technology and packaging design will not only improve medication safety and compliance but also position blister packaging as a strategic component of pharmaceutical delivery systems.

Common Pitfalls in Sourcing Blister Packaging for Medication: Quality and Intellectual Property Concerns
Quality-Related Pitfalls
Inadequate Material Specifications
One of the most common quality pitfalls is sourcing blister packaging materials without clearly defined specifications. Using substandard thermoform films, aluminum foils, or adhesives can compromise the integrity of the packaging, leading to reduced shelf life, moisture ingress, or contamination. It is essential to ensure materials meet regulatory standards such as USP <661> or Ph. Eur. 3.1.
Poor Sealing Performance
Inconsistent or weak sealing between the lidding foil and the thermoformed base can result in leaks, tampering, or exposure to environmental factors. Suppliers may cut corners on sealing parameters or use incompatible material combinations. Proper validation of the sealing process (e.g., peel strength, burst testing) is critical to ensure product safety.
Lack of Regulatory Compliance
Sourcing from manufacturers not compliant with cGMP (current Good Manufacturing Practices) or lacking certifications such as ISO 15378 (for primary packaging materials) increases the risk of non-compliant products. Regulatory bodies like the FDA or EMA may reject drug submissions if packaging components do not meet required standards.
Insufficient Testing and Qualification
Many sourcing decisions are made without adequate performance testing under real-world conditions (e.g., stability studies, child-resistance testing, or transit simulation). Skipping these steps can lead to field failures, recalls, or patient safety issues.
Intellectual Property (IP)-Related Pitfalls
Infringement of Patented Packaging Designs
Blister pack designs, including specific configurations, child-resistant features, or anti-tampering mechanisms, may be protected by patents. Sourcing generic or copied designs without conducting a freedom-to-operate (FTO) analysis can expose companies to legal action, injunctions, or costly litigation.
Unauthorized Use of Brand-Specific Tooling
Some blister packs use custom molds or tooling designed for a specific drug product. Sourcing packaging from unauthorized vendors who replicate these tools violates intellectual property rights and may compromise product authenticity and consumer trust.
Counterfeit or Grey Market Packaging
Using third-party suppliers in regions with weak IP enforcement increases the risk of receiving counterfeit or grey market packaging materials. These may appear legitimate but lack proper quality controls and infringe on trademarks or design rights.
Lack of IP Clauses in Supplier Agreements
Failing to include robust intellectual property clauses in sourcing contracts can lead to disputes over ownership of custom designs, reverse engineering, or unauthorized resale of packaging to competitors. Clear contractual terms are essential to protect proprietary innovations.
Conclusion
To mitigate these pitfalls, pharmaceutical companies must conduct thorough due diligence, work only with qualified and audited suppliers, ensure compliance with global standards, and safeguard intellectual property through legal and technical measures. Proper risk management in sourcing blister packaging is vital to ensuring patient safety and regulatory success.

Logistics & Compliance Guide for Blister Packaging Medication
Blister packaging is a widely used method for pharmaceutical products due to its ability to ensure product integrity, provide tamper evidence, and support patient compliance. However, the logistics and compliance requirements for blister-packaged medications are stringent, given the need to maintain drug safety, efficacy, and regulatory adherence throughout the supply chain. This guide outlines key considerations for handling, storing, transporting, and ensuring regulatory compliance of blister-packaged medications.
Regulatory Framework and Standards
Blister-packaged medications must comply with international, national, and regional regulations. Key regulatory bodies include the U.S. Food and Drug Administration (FDA), European Medicines Agency (EMA), and World Health Organization (WHO). Compliance involves adherence to Good Manufacturing Practices (GMP), Good Distribution Practices (GDP), and relevant packaging standards such as:
- ISO 15378: Primary packaging materials for medicinal products – GMP requirements.
- ICH Q1A-Q1E: Stability testing of new drug substances and products.
- EU GDP Guidelines: Ensuring proper storage and transportation conditions.
- 21 CFR Part 211 (FDA): Current GMP for finished pharmaceuticals.
Manufacturers and distributors must ensure that blister packaging materials are tested and validated for compatibility, barrier properties (moisture, oxygen), and seal integrity to prevent contamination and degradation.
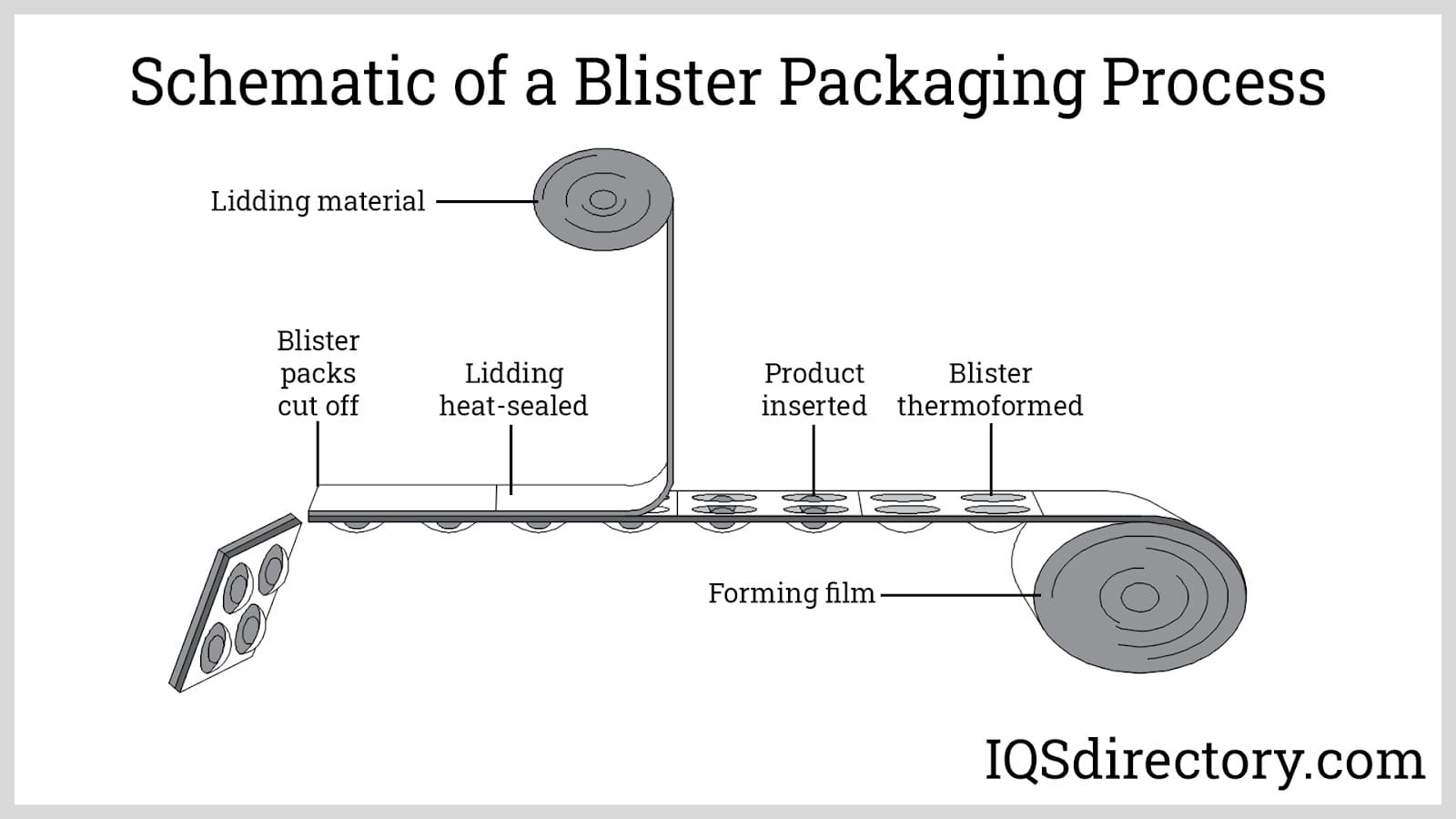
Packaging Design and Material Compliance
Blister packaging typically consists of a thermoformed cavity (usually PVC, PVDC, or aluminum) and a lidding material (often aluminum foil). Key compliance considerations include:
- Material Safety: All materials must be pharmaceutical-grade and comply with food-contact regulations (e.g., FDA 21 CFR, EU 10/2011).
- Child-Resistant and Senior-Friendly Features: Where required, packaging must meet ISTA or ISO standards for child resistance while remaining accessible to elderly users.
- Labeling Requirements: Must include product name, strength, batch number, expiration date, storage conditions, and regulatory approval markings (e.g., CE, Rx).
- Serialization and Traceability: Compliance with track-and-trace regulations such as the U.S. Drug Supply Chain Security Act (DSCSA) and EU Falsified Medicines Directive (FMD), which mandate unique identifiers (e.g., 2D barcodes) on each package.
Storage and Environmental Controls
Blister-packaged medications are sensitive to environmental conditions. Proper storage is critical to maintaining shelf life and efficacy:
- Temperature Control: Store according to labeled requirements (e.g., room temperature 15–25°C, or refrigerated 2–8°C). Continuous monitoring with calibrated devices is required.
- Humidity Management: Relative humidity should be controlled, especially for moisture-sensitive drugs. Desiccants may be included in secondary packaging.
- Light Protection: Use opaque or aluminum-based blister foils to protect light-sensitive medications.
- Stock Rotation: Implement FIFO (First In, First Out) or FEFO (First Expired, First Out) systems to prevent expiry.
Storage facilities must be qualified and regularly audited for compliance with GDP.
Transportation and Distribution
Logistics of blister-packaged medications must ensure product integrity from manufacturer to end-user:
- Cold Chain Management: For temperature-sensitive products, use validated cold chain packaging (e.g., insulated shippers with phase-change materials) and real-time temperature monitoring devices (data loggers).
- Packaging Integrity: Secondary packaging (e.g., cartons, trays) must protect blister cards from physical damage, moisture, and contamination during transit.
- Tamper-Evident Seals: Use tamper-evident shipping containers and security seals to prevent diversion and counterfeiting.
- Documentation and Traceability: Maintain shipping records, temperature logs, and batch traceability data. Electronic systems should support DSCSA verification requirements.
Transportation providers must be qualified and comply with GDP guidelines.
Quality Control and Validation
Robust quality systems are essential throughout the lifecycle of blister-packaged medications:
- Seal Integrity Testing: Perform regular testing (e.g., dye penetration, vacuum decay) to ensure blister seals are hermetic.
- Stability Studies: Conduct ongoing stability programs to validate shelf life under recommended storage conditions.
- Process Validation: Validate thermoforming, filling, and sealing processes to ensure consistency and compliance.
- Supplier Qualification: Audit and approve all packaging material suppliers per GMP requirements.
Returns, Recalls, and Waste Management
Procedures must be in place for handling returned or recalled blister-packaged medications:
- Quarantine and Investigation: Segregate returned products and investigate reasons for return (e.g., temperature excursion, damage).
- Recall Readiness: Maintain systems for rapid identification and retrieval of affected batches using serialization data.
- Environmental Disposal: Follow local and international regulations (e.g., EPA, EU Waste Framework Directive) for disposal of expired or non-compliant medications.
Training and Documentation
Personnel involved in handling blister-packaged medications must be trained in:
- GDP and GMP principles
- Handling of temperature-sensitive products
- Cold chain procedures
- Serialization and traceability systems
- Incident reporting and recall execution
All processes must be documented, and records retained for regulatory inspections.
Conclusion
Effective logistics and compliance for blister-packaged medications require a holistic approach integrating regulatory adherence, environmental control, traceability, and quality assurance. By following established standards and maintaining robust operational procedures, pharmaceutical companies can ensure the safety, efficacy, and integrity of their products throughout the supply chain.
Conclusion for Sourcing Blister Packaging for Medication:
Sourcing blister packaging for medication requires a strategic approach that balances regulatory compliance, product protection, patient safety, and cost-efficiency. Blister packaging plays a critical role in ensuring the stability, integrity, and tamper-evidence of pharmaceutical products, particularly in maintaining dosage accuracy and extending shelf life. When selecting a supplier, it is essential to evaluate factors such as material quality, manufacturing capabilities, adherence to Good Manufacturing Practices (GMP), and certifications like ISO 13485 and compliance with FDA or EMA standards.
Additionally, considerations around sustainability, patient usability (especially for elderly or pediatric populations), and automation compatibility in packaging lines should inform the decision-making process. Partnering with reliable and experienced suppliers who offer innovation, scalability, and responsive technical support can significantly enhance supply chain resilience and product quality.
In conclusion, a well-executed sourcing strategy for blister packaging not only ensures regulatory and safety compliance but also contributes to brand reputation, patient trust, and long-term operational success in the competitive pharmaceutical market.