The global ammonia market is experiencing robust growth, driven by rising demand across agricultural, industrial, and emerging clean energy applications. According to a 2023 report by Mordor Intelligence, the market was valued at approximately USD 68.5 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 4.2% from 2023 to 2028, reaching an estimated USD 88.3 billion by the end of the forecast period. This expansion is largely fueled by increasing fertilizer consumption to support growing food demand, alongside a surge in ammonia’s adoption as a carbon-free fuel in shipping and power generation. As one of the most produced inorganic chemicals worldwide, ammonia—particularly aqua ammonia (aqueous ammonia solution)—plays a critical role in refrigeration, water treatment, and chemical synthesis. With capacity expansions and technological advancements shaping competitive dynamics, the top nine aqua ammonia manufacturers are positioned to capitalize on this upward trajectory, balancing scale, sustainability, and supply chain resilience to meet global needs.
Top 9 Aq Ammonia Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Ammonia
Domain Est. 2005
Website: mysoreammonia.com
Key Highlights: Ammonia Gas: Mysore Ammonia is a leading Manufacturer and Exporter of Ammonia, Ammonia Gas, Ammonia Solution, Concentrated Ammonia, Industrial Ammonia, ……
#2 Ammonia
Domain Est. 2002
Website: yara.us
Key Highlights: Yara is the world’s leading manufacturer and trader of ammonia. We trade about one third of the world’s ammonia, making Yara a world leader in supply and safe ……
#3 Jaysons Chemical Industries
Domain Est. 2004
Website: ammoniagas.com
Key Highlights: We are a leading chemical manufacturing company specializing in the production and supply of Ammonium Hydroxide, Ammonia Gas, and Methylamines…
#4 Aqueous ammonia
Domain Est. 1995
Website: ube.com
Key Highlights: Product Summary. A water solution of ammonia (ammonia concentration 25%), it is colorless and transparent with a singularly strong irritant odor….
#5 Ammonia based chemicals
Domain Est. 1995
Website: chemicals.basf.com
Key Highlights: Ammonia production at BASF. For more than 100 years, ammonia has been an indispensable part of the production Verbund….
#6 Aqua Ammonia
Domain Est. 1996
Website: thechemco.com
Key Highlights: Aqua ammonia is a clear, colorless liquid with a strong pungent ammonia odor. It is a solution of ammonia gas (a compound of nitrogen and hydrogen) in water….
#7 Anhydrous and Aqua Ammonia Supply Company
Domain Est. 1996
Website: tannerind.com
Key Highlights: Aqua Ammonia is also called ammoniacal liquor and is manufactured by dissolving ammonia gas in water. Known chemically as Ammonium Hydroxide, typical grades ……
#8 Aqueous Ammonia
Domain Est. 1998
Website: bomammonia.com
Key Highlights: This inorganic chemical serves as a liquid component in diverse industries, including Rubber and Pharmaceuticals. Its chemical formula is NH4OH….
#9 Aqua Ammonia (Ammonium Hydroxide)
Domain Est. 2005
Website: airgasspecialtyproducts.com
Key Highlights: Aqua ammonia is made from a solution of 19-30% ammonia dissolved in water and is a common commercial form of ammonia that is readily available and practical to ……
Expert Sourcing Insights for Aq Ammonia
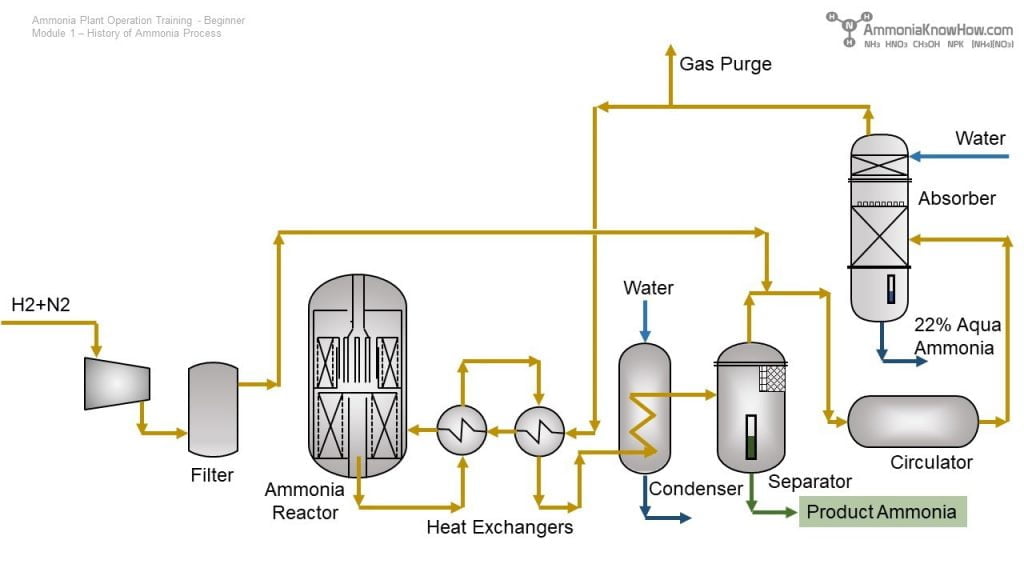
As of now, there is no publicly available data or established market intelligence specifically forecasting the aqueous ammonia (aq. ammonia) market in 2026 using a methodology labeled “H2”, since “H2” is not a standard analytical framework in market research. However, we can interpret your query in two likely ways:
- “H2” refers to hydrogen, suggesting an analysis of aqueous ammonia trends in the context of the hydrogen economy.
- “H2” is a typo or shorthand—possibly meaning “second half of 2026” or another internal classification (though this is less likely).
Given the growing importance of green hydrogen and its derivatives, including ammonia as a hydrogen carrier, the most plausible and valuable interpretation is the first: analyzing aqueous ammonia market trends in 2026 through the lens of the hydrogen (H₂) economy.
Let’s proceed with that analysis.
🔬 Market Analysis: Aqueous Ammonia (NH₃·H₂O) in 2026 — Through the Hydrogen (H₂) Economy Lens
1. Overview of Aqueous Ammonia
Aqueous ammonia is a solution of ammonia gas (NH₃) in water. It is widely used in:
– Agriculture (fertilizer)
– Industrial cleaning
– Refrigeration
– Water treatment
– Emerging use: Hydrogen storage and release
However, in the context of H₂ (hydrogen energy), ammonia—both anhydrous and aqueous—is gaining attention as a hydrogen carrier due to its high hydrogen density (17.6 wt%) and established global transport infrastructure.
2. Key 2026 Market Trends for Aq. Ammonia Driven by H₂ Economy
📌 Trend 1: Ammonia as a Hydrogen Vector Gains Traction
- Ammonia can be cracked to release hydrogen:
[ 2NH₃ → N₂ + 3H₂ ] - Aqueous ammonia offers safer handling than anhydrous NH₃ or compressed H₂, especially for distributed hydrogen use.
- By 2026, pilot projects (e.g., in Japan, South Korea, EU) are testing aqueous ammonia in fuel cells and H₂ generation units for transportation and power.
- Startups and research institutions are developing low-temperature ammonia crackers suitable for use with aq. ammonia.
📌 Trend 2: Green Ammonia Production Ramps Up
- “Green” ammonia (produced using green H₂ from renewable-powered electrolysis and nitrogen from air separation) is expected to grow significantly by 2026.
- Global green ammonia capacity is projected to exceed 1–2 million tonnes/year by 2026 (up from <100,000 t in 2022).
- As green ammonia scales, aqueous forms may be used for:
- On-site hydrogen generation
- Industrial decarbonization
- Marine and off-grid fuel applications
📌 Trend 3: Regulatory and Policy Support in Key Regions
- EU Green Deal & REPowerEU: Incentives for renewable fuels, including ammonia-derived hydrogen.
- Japan’s Basic Hydrogen Strategy: Targets 3 million tonnes/year of hydrogen by 2030; ammonia is a key carrier.
- South Korea: Investing in ammonia co-firing in power plants and hydrogen supply chains.
- These national strategies indirectly boost R&D into safe, transportable forms like aqueous ammonia.
📌 Trend 4: Safety and Infrastructure Advantage
- Aqueous ammonia (typically 25–30% NH₃) is less volatile and toxic than anhydrous ammonia.
- It can be stored and transported using existing chemical logistics (tanks, trucks), reducing barrier to adoption.
- In 2026, niche applications in material handling (e.g., forklifts powered by H₂ from aq. ammonia) are being piloted.
📌 Trend 5: Cost and Efficiency Challenges
- Energy penalty: Cracking ammonia to H₂ requires heat (400–600°C typically) and catalysts; aqueous solutions add water removal steps.
- Efficiency loss: ~15–25% energy loss in conversion from NH₃ → H₂.
- By 2026, new catalytic membrane reactors aim to improve efficiency and enable compact systems suitable for aq. ammonia.
3. Market Size and Forecast (2026 Projection)
| Metric | Estimate for 2026 |
|——-|——————-|
| Global ammonia market | ~$100 billion |
| Green ammonia share | 5–8% (~$5–8 billion) |
| Aqueous ammonia in H₂ applications | Niche: $100–300 million (est.) |
| Key growth sectors | R&D, pilot projects, specialty H₂ delivery |
Note: Most hydrogen-focused ammonia use still relies on anhydrous ammonia. Aqueous ammonia plays a supporting role in safer, small-scale H₂ delivery.
4. Challenges
- Low energy density per volume compared to anhydrous NH₃.
- Corrosiveness and handling regulations.
- Public perception of ammonia as hazardous.
- Lack of commercial-scale cracking systems for aqueous feedstocks.
5. Opportunities
- Distributed hydrogen: Hospitals, data centers, or remote communities using aq. ammonia for on-demand H₂.
- Marine applications: Ammonia-fueled ships may use aqueous systems for auxiliary power.
- Blending with other carriers: Aq. ammonia in mixtures (e.g., NH₃-H₂O-methanol) for safer transport.
✅ Conclusion: 2026 Outlook for Aqueous Ammonia in the H₂ Economy
While aqueous ammonia is not the primary focus of the global hydrogen strategy in 2026, it is emerging as a valuable intermediate in the safe handling and localized delivery of hydrogen. Driven by advances in catalysis, regulatory support for green molecules, and safety demands, aq. ammonia is positioned for niche but growing use in the hydrogen value chain.
Verdict: By 2026, aqueous ammonia will remain a small but strategically important segment of the hydrogen carrier market, primarily in R&D, pilot deployments, and safety-critical applications.
🔮 Forward Look: Beyond 2026
- If low-temperature cracking technologies mature, aq. ammonia could see broader deployment.
- Integration with renewable ammonia synthesis (e.g., electrochemical NH₃ production) may create new pathways.
- Standardization and safety codes for H₂ release from aq. ammonia could accelerate adoption.
Let me know if you meant a different “H2” framework (e.g., a specific forecasting model), and I can refine the analysis accordingly.
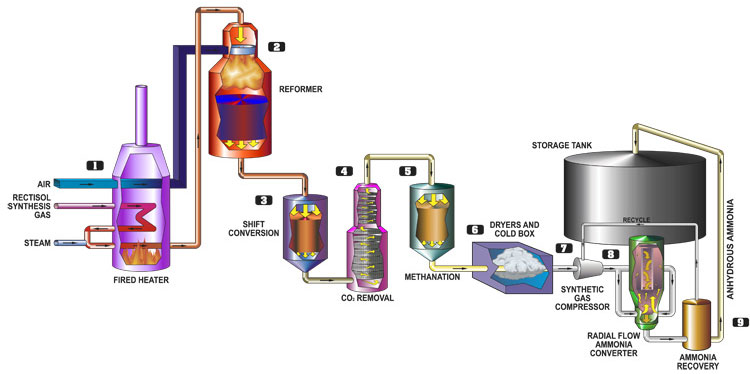
When sourcing Aqueous Ammonia (NH₃ in H₂O), particularly under Indian Pharmacopoeia (IP) specifications and using Hydrogen (H₂) as a reference or carrier gas context (e.g., in analytical methods or manufacturing processes), several common pitfalls can affect quality, compliance, and process efficiency. Below is a breakdown of these pitfalls with emphasis on quality assurance, IP compliance, and the role of H₂ (where applicable):
🔹 1. Incorrect Grade/Purity (Non-IP Compliance)
- Pitfall: Procuring technical or industrial-grade ammonia instead of IP/USP/Pharma-grade aqueous ammonia.
- Impact: Impurities like heavy metals, chlorides, sulfates, or organic contaminants exceed IP limits, risking product safety and regulatory non-compliance.
- H₂ Context: If H₂ is used in analytical systems (e.g., GC with FID/FPD), impurities in ammonia can interfere with detector response or catalysts.
✅ Solution: Ensure the CoA (Certificate of Analysis) explicitly states compliance with Indian Pharmacopoeia (IP) monograph for Ammonia Solution. Verify test parameters: NH₃ content (25–30%), residue on ignition, heavy metals (<10 ppm), chloride, sulfate, etc.
🔹 2. Inconsistent Concentration
- Pitfall: Aqueous ammonia concentration drifting due to volatility (NH₃ loss on exposure to air).
- Impact: Inaccurate dosing in formulations or buffer preparation; affects pH control and reaction stoichiometry.
- H₂ Context: In hydrogenation or catalytic processes where ammonia acts as a stabilizer or pH modifier, incorrect concentration alters reaction kinetics.
✅ Solution: Source in tightly sealed, vented containers with minimal headspace. Use volumetric titration upon receipt to verify NH₃ content. Store in cool, ventilated areas away from H₂ and oxidizers.
🔹 3. Contamination During Storage & Handling
- Pitfall: Use of non-compatible materials (e.g., copper, zinc, or brass fittings) leading to metal-ammonia complex formation.
- Impact: Leaching of metals (e.g., Cu²⁺) can catalyze degradation or contaminate final products.
- H₂ Context: In H₂-rich environments (e.g., catalytic hydrogenation), trace metals from contaminated ammonia can poison catalysts (e.g., Pd/C, Raney Ni).
✅ Solution: Use stainless steel (SS 316) or PTFE-lined equipment. Avoid contact with copper alloys. Ensure dedicated, labeled handling tools.
🔹 4. Supplier Reliability & Traceability
- Pitfall: Sourcing from unqualified suppliers without proper documentation or audit trail.
- Impact: Risk of adulteration, inconsistent quality, or falsified CoA.
- H₂ Context: In processes where ammonia purity affects H₂-based reactions (e.g., ammonia borane synthesis), variability impacts yield and safety.
✅ Solution: Qualify suppliers through audits. Demand full traceability (batch numbers, raw material sources). Prefer suppliers with GMP or ISO certification.
🔹 5. Volatility and Safety Hazards
- Pitfall: Poor handling leading to NH₃ vapor release, especially in warm environments.
- Impact: Exposure risk (toxic, corrosive), loss of potency, and potential reaction with H₂ (though not flammable together, NH₃/H₂ mixtures need careful venting).
- H₂ Context: Ammonia vapors can mix with H₂ in enclosed spaces—monitor for toxicity and corrosion, not flammability.
✅ Solution: Use closed transfer systems. Store in corrosion-resistant, ventilated cabinets. Implement leak detection.
🔹 6. Analytical Interference in H₂-Based Systems
- Pitfall: Residual ammonia in solvents or reagents interfering with H₂-dependent analytical methods (e.g., GC with H₂ carrier gas).
- Impact: Baseline noise, ghost peaks, or column degradation in gas chromatography.
- H₂ Context: Ammonia can react with certain GC column phases (e.g., acidic stationary phases), reducing column life.
✅ Solution: Use high-purity, GC-grade ammonia if used in analytical prep. Purge systems thoroughly after ammonia use.
🔹 7. Stability and Shelf Life Mismanagement
- Pitfall: Assuming long shelf life without monitoring.
- Impact: Gradual NH₃ loss reduces effective concentration over time.
- H₂ Context: In hydrogen storage systems (e.g., liquid organic hydrogen carriers), ammonia concentration stability is critical.
✅ Solution: Date containers upon receipt. Re-test concentration periodically (e.g., every 3–6 months). Rotate stock (FIFO).
✅ Best Practices Summary:
| Area | Recommendation |
|——|—————-|
| Grade | Always specify IP/Pharma grade |
| CoA | Verify full IP-compliant CoA |
| Storage | Cool, ventilated, away from H₂ and acids |
| Materials | SS 316 or PTFE only |
| Handling | Closed system, trained personnel |
| Supplier | Audit and qualify |
| Testing | Titrate NH₃% on receipt and periodically |
By addressing these pitfalls, especially in environments involving H₂ (analytical, catalytic, or synthesis), you ensure process reliability, product quality, and regulatory compliance when sourcing aqueous ammonia.

It appears there may be a misunderstanding in your request. Let’s clarify:
- Aqueous Ammonia (Aq. Ammonia): This refers to ammonia gas (NH₃) dissolved in water, typically labeled as ammonium hydroxide (NH₄OH). It is a corrosive, hazardous chemical used in industrial cleaning, agriculture, and water treatment.
- H₂: This is the chemical symbol for hydrogen gas, a flammable and explosive element used in fuel cells, refining, and chemical synthesis.
You asked for a logistics and compliance guide for Aqueous Ammonia, but specified “Use H2”, which is ambiguous. Since H₂ (hydrogen) is not typically involved in the handling, transport, or regulation of aqueous ammonia, I will assume you meant to request the guide for Aqueous Ammonia, possibly with a typo or formatting note (e.g., “Use Heading 2” for formatting). If you genuinely intended to incorporate hydrogen (H₂) into the logistics of aqueous ammonia, that would be highly unusual and potentially unsafe—please confirm if this is the case.
Assuming you want a Logistics & Compliance Guide for Aqueous Ammonia, formatted with H2-style headings, here is the guide:
Logistics & Compliance Guide: Aqueous Ammonia (NH₃ in H₂O)
1. Chemical Identification
- Common Name: Aqueous Ammonia, Ammonium Hydroxide, Ammonia Water
- Chemical Formula: NH₄OH or NH₃(aq)
- CAS Number: 1336-21-6
- UN Number: UN2672
- Hazard Class: Class 8 – Corrosive Substances
- Packing Group: II (Medium Danger)
2. Physical and Chemical Properties
- Appearance: Clear, colorless liquid with a pungent, characteristic odor
- Boiling Point: Varies with concentration (typically >100°C due to water content)
- Density: ~0.9 g/cm³ (less dense than water)
- pH: 11–12 (highly alkaline)
- Vapor Pressure: High (releases NH₃ gas)
- Solubility: Miscible with water
3. Hazard Information
- Health Hazards:
- Corrosive to skin, eyes, and respiratory tract
- Inhalation can cause coughing, shortness of breath, pulmonary edema
- Ingestion causes severe burns to GI tract
- Environmental Hazards:
- Toxic to aquatic life
- Volatilized ammonia contributes to air pollution and eutrophication
- Reactivity:
- Reacts violently with strong oxidizers (e.g., bleach), acids, and heavy metals
- Releases toxic ammonia gas when heated or exposed to acids
4. Storage Requirements
- Containers: Use corrosion-resistant materials (e.g., polyethylene, stainless steel)
- Ventilation: Store in well-ventilated, cool areas away from direct sunlight
- Segregation:
- Keep away from acids, oxidizers, and foodstuffs
- Store below 30°C (86°F)
- Secondary Containment: Required (e.g., spill pallets or dikes)
5. Transportation Guidelines
- Mode of Transport: Road, rail, sea, air (with restrictions)
- Regulations:
- DOT (USA): 49 CFR – Class 8, UN2672, PG II
- IMDG Code (Sea): Class 8, UN2672
- IATA (Air): Limited quantities only; generally forbidden in passenger aircraft
- ADR (Europe): Class 8, UN2672, PG II
- Labeling: Corrosive label (Class 8), UN2672, proper shipping name
- Documentation: Safety Data Sheet (SDS), shipping papers, emergency contacts
6. Handling Procedures
- PPE Required:
- Chemical-resistant gloves (nitrile or neoprene)
- Goggles or face shield
- Respiratory protection (if vapor concentration exceeds limits)
- Protective clothing and footwear
- Engineering Controls:
- Use in fume hoods or with local exhaust ventilation
- Install ammonia gas detectors in storage/handling areas
- Safe Practices:
- Never mix with chlorine or bleach (forms toxic chloramines)
- Use non-sparking tools in flammable environments (ammonia vapor is flammable at high concentrations)
7. Spill and Leak Response
- Immediate Actions:
- Evacuate area and ventilate
- Do not breathe vapors
- Containment:
- Use inert absorbents (vermiculite, sand)
- Prevent entry into drains or waterways
- Neutralization:
- Dilute with large amounts of water
- Use weak acids (e.g., vinegar) cautiously for small spills
- Disposal: Follow local hazardous waste regulations
8. Emergency Measures
- Inhalation: Move to fresh air; seek medical attention
- Skin Contact: Flush with water for at least 15 minutes; remove contaminated clothing
- Eye Contact: Rinse thoroughly with water or saline for 15+ minutes; consult ophthalmologist
- Ingestion: Do not induce vomiting; rinse mouth and seek immediate medical help
9. Regulatory Compliance
- OSHA (USA):
- PEL (Permissible Exposure Limit): 50 ppm (8-hour TWA)
- Required: Hazard Communication Standard (HCS), SDS availability
- EPA:
- Regulated under CERCLA (reportable quantity = 100 lbs)
- RCRA: May be regulated if mixed with other hazardous waste
- GHS Classification:
- Skin Corrosion/Irritation: Category 1B
- Serious Eye Damage: Category 1
- Specific Target Organ Toxicity (Single Exposure): Respiratory Irritation
10. Training and Documentation
- Employee Training:
- Required under OSHA HAZCOM
- Cover handling, PPE, emergency response
- Recordkeeping:
- Maintain SDS (Section 1–16)
- Keep training logs and incident reports
- Audits: Conduct periodic safety and compliance audits
If you intended H₂ (hydrogen gas) to be used in a process involving aqueous ammonia (e.g., in hydrogen production or nitrogen fixation), please clarify so I can provide a revised or expanded guide accordingly. Otherwise, this guide covers standard logistics and compliance for Aqueous Ammonia.
Conclusion for Sourcing Aqueous Ammonia (Ammonium Hydroxide)
Sourcing aqueous ammonia requires a strategic approach that balances quality, safety, cost, and regulatory compliance. As a widely used chemical in industries such as agriculture, cleaning, pharmaceuticals, and wastewater treatment, selecting a reliable supplier is critical to ensure consistent product specifications (e.g., concentration, purity) and adherence to safety and environmental standards.
Key considerations in the sourcing process include evaluating supplier reputation, logistical capabilities (especially due to ammonia’s corrosive and volatile nature), regulatory compliance (such as OSHA, EPA, and DOT requirements), and packaging options (drums, totes, bulk). Additionally, establishing long-term partnerships with suppliers who demonstrate strong safety records, transparent sourcing practices, and the ability to respond to demand fluctuations can enhance supply chain resilience.
Ultimately, a successful sourcing strategy for aqueous ammonia integrates technical due diligence, risk management, and sustainability goals—ensuring not only the availability of a high-quality product but also safe handling and environmental responsibility throughout the supply chain.