The global laboratory equipment market, which includes essential tools like Bunsen burners, is experiencing steady growth driven by rising investments in research and development across pharmaceutical, biotechnology, and academic sectors. According to Grand View Research, the global laboratory equipment market was valued at USD 68.5 billion in 2022 and is expected to expand at a compound annual growth rate (CAGR) of 5.8% from 2023 to 2030. A key segment within this market is gas-based laboratory burners, where demand is increasingly shaped by safety, precision, and energy source preferences—particularly between alcohol and butane-fueled Bunsen burners.
While traditional butane-powered models remain dominant due to their higher flame temperature and portability, alcohol-based burners are gaining traction in educational and eco-conscious environments for their cleaner combustion and reduced regulatory burden. This shift has catalyzed innovation among manufacturers, with established players and emerging brands alike refining their offerings to meet evolving safety standards and application-specific needs. Based on production output, market presence, safety certifications, and product reviews, the following nine manufacturers represent the leading innovators in alcohol and butane Bunsen burner technology—each contributing uniquely to a niche that balances tradition, performance, and sustainability.
Top 9 Alcohol Vs Butane Bunsen Burner Manufacturers 2026
(Ranked by Factory Capability & Trust Score)
#1 Lab Bunsen Burners And Lighters
Domain Est. 1995
Website: thomassci.com
Key Highlights: The safe alternative for traditional Bunsen burner or alcohol! Flame without a match Innovative Security Technology Easy-to-use Low cost and environmentally ……
#2 Bunsen Burners
Domain Est. 1994
Website: coleparmer.com
Key Highlights: 3-day delivery 30-day returnsAs a lab burner, the Bunsen burner operates with artificial, natural, or liquid propane (LP) gas; it features a grid top or straight tube design….
#3 Burners and Lighters
Domain Est. 1995
Website: fishersci.com
Key Highlights: Browse a full range of Burners and Lighters products from leading suppliers. Shop now at Fisher Scientific for all of your scientific needs….
#4 Flame 100 – WLD-TEC GmbH
Domain Est. 1997
Website: wld-tec.com
Key Highlights: Flame 100 – sets new standards in the Bunsen burner class! The safe alternative for all traditional Bunsen or alcohol burners! Item Number: 2.100.000. 1; 2; 3….
#5 Bunsen Burners & Butane Canisters
Domain Est. 1999
Website: praxisdienst.com
Key Highlights: Free 14-day returnsA vital tool for any lab, we stock a selection of high performance Bunsen burners and compatible butane canisters. Shop now!…
#6 Burners and Lighters
Domain Est. 2000
Website: fishersci.ca
Key Highlights: Browse a full range of Burners and Lighters products from leading suppliers. Shop now at Fisher Scientific for all of your scientific needs….
#7 Bunsen Burner vs Alternatives
Domain Est. 2013
Website: supertekglassware.com
Key Highlights: Rating 5.0 (5) Apr 16, 2025 · The Bunsen burner remains a classic for general lab work. However, if you are in a school lab, the alcohol burner may be better due to its safety an…
#8 Lab Burner
Domain Est. 2014
#9 Liquid Propane or Butane Gas
Domain Est. 2018
Website: congeriem.com
Key Highlights: In stock Rating 4.8 (2,275) Eisco Labs Bunsen Burner. Suitable for LPG (Liquid Propane Gas) or Butane Gas. Made of high quality brass and mounted on a steel powder coated base….
Expert Sourcing Insights for Alcohol Vs Butane Bunsen Burner
H2: Comparative Market Trend Analysis of Alcohol and Butane Bunsen Burners (2026 Outlook)
As we approach 2026, the market for laboratory and educational Bunsen burners is witnessing a shift in demand dynamics between traditional fuel types—specifically alcohol (ethanol or methanol) and butane. This analysis evaluates key market trends influencing the adoption, regulation, cost, and technological evolution of alcohol versus butane Bunsen burners.
-
Safety and Regulatory Environment
-
Butane Burners: Butane is highly flammable and pressurized, requiring strict safety protocols. In 2026, increasing safety regulations in schools and laboratories—especially in the EU and North America—are making institutions cautious about storing and using compressed gas. Regulatory bodies such as OSHA (U.S.) and REACH (EU) continue to scrutinize volatile organic compounds (VOCs), indirectly affecting butane use.
-
Alcohol Burners: Liquid alcohol burners are perceived as safer due to lower pressure and reduced explosion risk. In response to stricter safety standards, many schools are transitioning to alcohol-based systems, particularly in primary and secondary education. Alcohol (especially denatured ethanol) emits fewer hazardous byproducts, aligning with growing institutional emphasis on indoor air quality.
Market Implication: By 2026, alcohol burners are gaining favor in education markets due to safety compliance, while butane remains dominant in advanced labs where flame control is critical.
-
Environmental and Sustainability Trends
-
Butane: As a fossil fuel derivative, butane contributes to carbon emissions. With global net-zero targets influencing procurement policies, institutions are re-evaluating reliance on hydrocarbon-based fuels. The carbon footprint of butane production and transport is under greater scrutiny.
-
Alcohol: Ethanol, especially when derived from renewable biomass, is viewed as a greener alternative. Bioethanol-powered burners are increasingly marketed as sustainable lab tools. In regions promoting circular economy principles (e.g., Scandinavia, Canada), demand for alcohol burners is rising.
Market Implication: Sustainability mandates are tilting procurement decisions toward alcohol burners, particularly in public educational systems and environmentally certified laboratories.
-
Performance and Application Needs
-
Butane Burners: Offer higher flame temperatures (up to 1,970°C) and precise adjustability, making them ideal for advanced chemistry, microbiology (sterilization), and metallurgical applications. Research labs and universities with specialized needs continue to prefer butane for its performance.
-
Alcohol Burners: Typically reach 900–1,000°C, sufficient for basic sterilization and heating tasks in classrooms. However, the lower heat output limits their use in high-temperature experiments. Innovations in wick and chamber design are improving efficiency, but a performance gap remains.
Market Implication: Butane maintains dominance in high-performance laboratory settings, while alcohol burners are becoming the standard for K–12 and introductory college labs.
-
Cost and Accessibility
-
Butane: Requires pressurized canisters and specialized regulators. Refills can be costly and less accessible in remote or developing regions. However, refill infrastructure is well-established in urban areas.
-
Alcohol: Fuel is cheaper, easier to store, and widely available. Denatured ethanol is non-pressurized, reducing shipping and storage costs. In low-resource settings (e.g., rural schools in Africa or Southeast Asia), alcohol burners offer a cost-effective and logistically simpler solution.
Market Implication: In emerging markets, alcohol burners are expanding rapidly due to affordability and ease of supply, whereas butane remains cost-competitive in developed economies with robust distribution networks.
-
Technological Innovation and Market Adoption
-
Butane burner designs are evolving with safety-enhanced valves and auto-shutoff features, helping retain market share. Smart integration (e.g., IoT-enabled gas monitoring) is emerging in research-grade equipment.
-
Alcohol burners are seeing design improvements—such as spill-proof reservoirs, adjustable air intake, and longer burn times—making them more reliable and user-friendly. Some manufacturers now offer hybrid models with dual-fuel capability.
Market Implication: Innovation is blurring the lines between the two, but distinct use cases persist. The hybrid model may represent a niche growth area by 2026.
Conclusion (H2 Summary):
By 2026, the Bunsen burner market is increasingly segmented by application and regulatory context. Alcohol burners are gaining ground in educational and sustainability-focused institutions due to safety, lower cost, and environmental benefits. Conversely, butane burners remain entrenched in professional and research laboratories where high flame intensity and precision are non-negotiable. Market trends suggest a divergent but coexisting trajectory: alcohol for accessibility and safety, butane for performance and control. Manufacturers adapting to this duality—offering fuel-specific or hybrid solutions—are best positioned for growth.
When comparing Alcohol vs. Butane Bunsen Burners—especially with a focus on quality, intellectual property (IP), and the potential use of hydrogen (H₂) as an alternative fuel—it’s important to understand the common pitfalls in sourcing and implementation. Below is a structured analysis of the challenges and considerations:
1. Sourcing Pitfalls: Alcohol vs. Butane Bunsen Burners
A. Quality Concerns
| Factor | Alcohol Burner Pitfalls | Butane Burner Pitfalls |
|——-|————————|————————|
| Flame Stability & Control | – Lower flame temperature (~400–600°C), inconsistent with evaporation rates.
– Harder to regulate flame precisely. | – Higher temperature (~800–1400°C), but can be too intense for delicate work.
– Risk of flashback or flame lift if poorly designed. |
| Fuel Purity & Consistency | – Denatured alcohol may contain impurities affecting combustion.
– Evaporation during storage reduces efficiency. | – Butane quality varies; impurities (e.g., propane, mercaptans) affect burn.
– Pressure fluctuations in canisters affect flame. |
| Material Compatibility | – Alcohol can degrade rubber seals or certain plastics over time. | – Butane requires robust O-rings and seals (e.g., nitrile); cheap versions fail. |
| Durability | – Simpler design, but wicks degrade and require frequent replacement. | – More complex valves and regulators prone to wear; low-cost units fail early. |
B. Intellectual Property (IP) Risks
| Issue | Alcohol Burners | Butane Burners |
|——|——————|—————-|
| Design Infringement | – Basic designs are often generic, low IP risk. | – Many butane burners mimic patented lab equipment (e.g., Thermo Fisher, Bunsen-Pro).
– Risk of copying valve mechanisms or safety features. |
| Counterfeit Products | – Less common due to simplicity. | – High risk; many Chinese-sourced butane burners copy branded designs without licenses. |
| Compliance & Certification | – Few regulated standards for alcohol units. | – Butane devices may require CE, UL, or gas safety certifications.
– Sourcing uncertified units risks legal liability. |
Key IP Tip: When sourcing butane burners, verify if the supplier holds legitimate design patents or licenses. Avoid units that closely mimic branded lab equipment unless authorized.
2. Introducing Hydrogen (H₂) – A Safer, Cleaner Alternative?
While alcohol and butane are common, hydrogen (H₂) is an emerging option for clean lab combustion. However, sourcing H₂-compatible systems introduces new challenges:
Pitfalls with H₂ Use:
| Challenge | Explanation |
|———|————-|
| Combustion Safety | – H₂ has a wide flammability range (4–75% in air) and low ignition energy.
– Risk of explosion if leaks occur. |
| Material Compatibility | – H₂ embrittlement can crack metals (e.g., steel) over time.
– Requires H₂-rated seals (e.g., PTFE, Viton). |
| Storage & Delivery | – H₂ gas requires high-pressure cylinders or electrolytic generators.
– Sourcing reliable, lab-scale H₂ systems is expensive. |
| Regulatory Compliance | – Strict handling, storage, and ventilation regulations (e.g., NFPA 2, OSHA).
– Labs may need special permits. |
| Cost & Infrastructure | – H₂ systems are not drop-in replacements; require retrofitting.
– High upfront cost compared to alcohol/butane. |
Quality & IP in H₂ Burners:
- Few off-the-shelf H₂ Bunsen burners exist; most are custom or research-grade.
- Risk of sourcing untested 3D-printed or DIY designs with poor safety controls.
- IP may involve patented catalytic combustion or flame-arrestor tech—verify freedom to operate.
3. Recommendations When Sourcing
- Prioritize Certified Suppliers
- For butane: Choose suppliers with CE, UL, or ISO 9001 certification.
-
For H₂: Use only lab equipment from reputable scientific vendors (e.g., Fisher Scientific, WPI).
-
Avoid Counterfeits
- Check for branding, packaging quality, and technical documentation.
-
Request test reports for gas compatibility and pressure safety.
-
Evaluate Total Cost of Ownership
- Alcohol: Cheap upfront, but frequent refills and wick replacements.
- Butane: Higher efficiency, but ongoing cartridge costs.
-
H₂: High setup cost, but clean, zero-carbon if green H₂ is used.
-
Consider Hybrid or Future-Proof Designs
- Some modern burners support multiple fuels (e.g., butane or H₂ with nozzle swaps).
-
Look for modular, IP-protected designs that allow safe fuel switching.
-
Conduct a Risk Assessment
- Especially for H₂: Perform a lab safety review and staff training before deployment.
Conclusion
| Fuel Type | Best For | Key Pitfalls | H₂ Viability |
|———|———|————-|————-|
| Alcohol | Teaching labs, low-temp apps | Low heat, wick maintenance, evaporation | Low – inefficient, not ideal |
| Butane | General lab use | IP risk, gas variability, safety | Medium – possible with retrofit |
| Hydrogen (H₂) | Green labs, precision work | Safety, cost, infrastructure | High – future-proof, clean, but complex |
✅ Best Practice: Use butane burners from certified suppliers with clear IP status, and explore H₂ only with proper safety protocols and institutional support. Avoid alcohol for high-precision work, and never compromise on gas system quality.
H2: Logistics & Compliance Guide: Alcohol vs. Butane Bunsen Burners
This guide provides a comparative overview of the logistics and regulatory compliance considerations for using alcohol-based and butane-powered Bunsen burners in laboratory, educational, and industrial settings. Understanding these differences is essential for safe handling, storage, transportation, and adherence to local, national, and international regulations.
H2: 1. Chemical Properties & Classification
Alcohol (e.g., Ethanol or Methanol):
– Classification: Flammable liquid (Class 3 under UN GHS).
– Flash Point: ~13°C (ethanol), ~11°C (methanol) — highly flammable at room temperature.
– Vapor Density: Lighter than air; vapors can travel and ignite at a distance.
– Regulatory Label: Flame pictogram, Health Hazard (methanol), Environmental Hazard (in some cases).
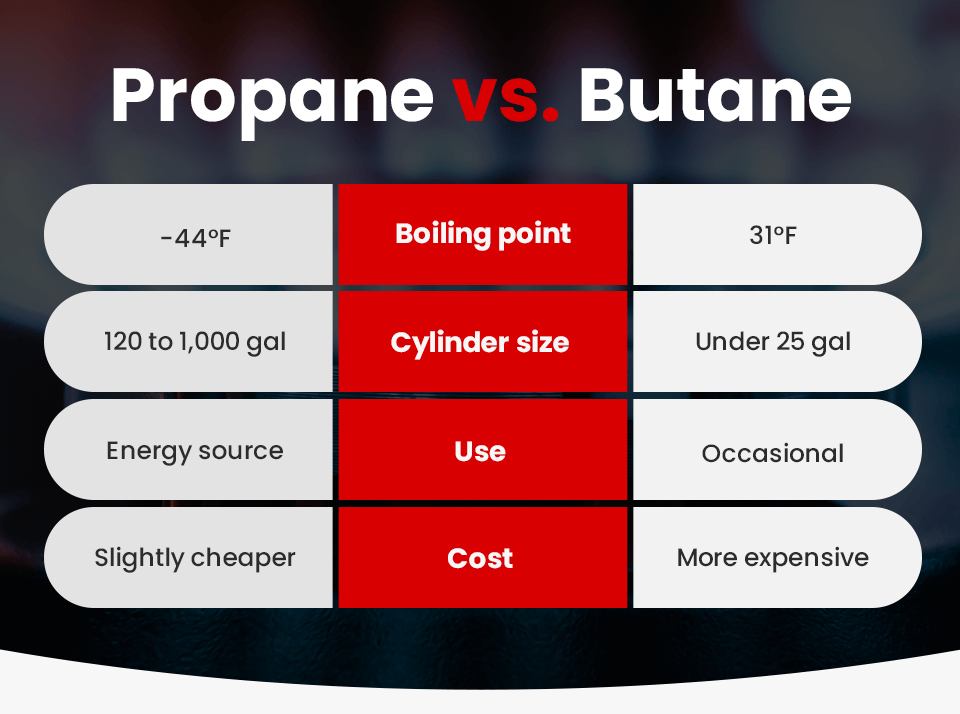
Butane:
– Classification: Flammable gas (Class 2.1 under UN GHS).
– State: Compressed liquefied gas.
– Flash Point: -60°C — extremely flammable, ignites easily.
– Vapor Density: Heavier than air; accumulates in low-lying areas, increasing explosion risk.
– Regulatory Label: Flame pictogram, Gas under pressure.
H2: 2. Storage Requirements
Alcohol:
– Store in approved flammable liquid safety cabinets.
– Must be kept away from heat, sparks, and oxidizers.
– Containers should be grounded and bonded during transfer.
– Ventilation: Adequate to prevent vapor buildup.
– Maximum allowable quantities per storage area regulated by fire codes (e.g., NFPA 30).
Butane:
– Store in well-ventilated, cool, dry areas away from ignition sources.
– Cylinders must be upright and secured to prevent falling.
– No indoor storage of large quantities; outdoor or dedicated gas storage lockers may be required.
– Prohibited in basements or pits due to vapor accumulation risk (NFPA 58).
– Temperature control: Avoid exposure to temperatures >50°C (risk of rupture).
H2: 3. Transportation
Alcohol:
– Subject to DOT (USA), ADR (Europe), and IATA/IMDG (air/sea) regulations for flammable liquids.
– Quantity limits per package; requires UN-certified containers (e.g., metal or approved plastic cans).
– Proper labeling: UN 1170, Flammable Liquid, Ethanol.
– Safety data sheet (SDS) required.
Butane:
– Regulated as a dangerous good (UN 1011, Liquefied Petroleum Gas).
– Requires UN-approved pressure cylinders with safety valves.
– Transport in ventilated vehicles; no passenger compartments.
– Prohibited on commercial aircraft in most circumstances (except small consumer devices under exceptions).
– Quantity limits strictly enforced; full compliance with 49 CFR (USA) or ADR (Europe).
H2: 4. Handling & Use
Alcohol Bunsen Burners:
– Refill only when cool and away from ignition sources.
– Use in fume hoods or well-ventilated areas.
– Spill risks: High volatility — immediate cleanup with absorbent materials.
– Refilling frequency: More frequent due to lower energy density.
Butane Bunsen Burners:
– Check for leaks using soapy water before each use.
– Use only with compatible, certified burners.
– Avoid repeated on/off cycling — risk of gas buildup.
– Never refill in use area; use only pre-filled, sealed cartridges.
– Safer in environments where open liquid fuel is a contamination or spill risk.
H2: 5. Regulatory Compliance
Occupational Safety (e.g., OSHA, HSE):
– Both require hazard communication (GHS labeling, SDS access).
– Alcohol: Permissible exposure limits (PELs) for vapors; methanol requires additional PPE.
– Butane: Confined space entry protocols due to asphyxiation and explosion risks.
Fire Codes (e.g., NFPA 30, NFPA 58):
– Alcohol: NFPA 30 governs storage and handling of flammable liquids.
– Butane: NFPA 58 applies to LPG storage and use — stricter separation distances.
– Local fire marshal approval may be required for bulk storage.
Environmental Regulations:
– Alcohol spills: May require reporting if released to drains or soil (especially methanol).
– Butane: No persistent environmental contamination, but large releases contribute to VOCs and greenhouse gases.
H2: 6. Risk Mitigation & PPE
| Hazard Type | Alcohol Burner | Butane Burner |
|———————|————————————|—————————————-|
| Fire/Explosion | High (vapor ignition) | Very High (gas accumulation) |
| Inhalation Risk | Moderate (vapors); High (methanol) | Low (short-term), asphyxiation risk |
| Skin Contact | Drying, irritation | Frostbite (from liquid butane) |
| Required PPE | Lab coat, gloves, safety goggles | Same + flame-resistant gloves |
| Engineering Controls | Fume hood, spill tray | Ventilation, leak detectors |
H2: 7. Disposal & Waste Management
Alcohol:
– Waste ethanol/methanol is hazardous waste.
– Must be disposed via licensed hazardous waste handlers.
– Never pour down the drain (violates EPA and local regulations).
Butane:
– Empty cylinders: Must be depressurized and recycled as scrap metal.
– Full or partially full cylinders: Return to supplier or dispose via hazardous waste program.
– Never puncture or incinerate cylinders.
H2: 8. Summary: Key Decision Factors
| Factor | Alcohol Burner | Butane Burner |
|————————|————————————|—————————————-|
| Ease of Use | Simple, but frequent refills | Long runtime, no refilling needed |
| Safety in Labs | Moderate fire risk, spill concern | High explosion risk if leaked |
| Regulatory Burden | Moderate (flammable liquid rules) | High (gas storage, cylinder handling) |
| Transport Complexity | Medium (liquid packaging) | High (pressure vessel regulations) |
| Environmental Impact | Biodegradable, but toxic in bulk | GHG emissions, no residue |
| Ideal Use Case | Low-use, teaching labs | Field work, portable applications |
H2: Conclusion
Choosing between alcohol and butane Bunsen burners involves balancing operational needs with safety and compliance requirements. Alcohol is easier to source and dispose of in small quantities but poses spill and vapor risks. Butane offers convenience and portability but demands strict adherence to gas safety codes. Institutions must conduct a site-specific risk assessment, train personnel, and maintain documentation to ensure full compliance with HAZMAT, fire, and occupational safety regulations.
In conclusion, when comparing alcohol and butane as fuel sources for Bunsen burners, several factors must be considered: safety, availability, cost, environmental impact, and performance. Alcohol burners are generally safer, producing lower flame temperatures and being less prone to explosive leaks, making them suitable for educational or low-temperature applications. They are also easier to handle and store, with fewer regulatory restrictions. However, alcohol provides less consistent heat and lower efficiency.
Butane burners, on the other hand, deliver higher, more controllable temperatures and are ideal for laboratory and industrial applications requiring precise heating. Butane is more energy-dense and burns cleaner than many fuels, but it poses greater safety risks due to its flammability and pressurized storage. It also tends to be more expensive and subject to stricter handling regulations.
Ultimately, the choice depends on the specific use case. For controlled, low-risk environments such as school laboratories, alcohol may be the preferred option. For applications demanding higher heat and precision, butane is more effective. Balancing safety, performance, and cost will guide the optimal sourcing decision.